 Found 26 hits with Last Name = 'kim' and Initial = 'gh'
Found 26 hits with Last Name = 'kim' and Initial = 'gh' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Adenosine deaminase
(Bos taurus (bovine)) | BDBM50105934

(2-(6-Amino-purin-7-ylmethoxy)-ethanol | CHEMBL1260...)Show InChI InChI=1S/C8H11N5O2/c9-7-6-8(11-3-10-7)12-4-13(6)5-15-2-1-14/h3-4,14H,1-2,5H2,(H2,9,10,11) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| 8.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Chemistry
Curated by ChEMBL
| Assay Description
Inhibitory activity against adenosine deaminase |
J Med Chem 44: 3710-20 (2001)
BindingDB Entry DOI: 10.7270/Q2G44R0Z |
More data for this
Ligand-Target Pair | |
Adenosine deaminase
(Bos taurus (bovine)) | BDBM50029651

(CHEMBL143926 | [2-(6-Amino-purin-9-ylmethoxy)-ethy...)Show InChI InChI=1S/C8H12N5O4P/c9-7-6-8(11-3-10-7)13(4-12-6)5-17-1-2-18(14,15)16/h3-4H,1-2,5H2,(H2,9,10,11)(H2,14,15,16) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| 1.82E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Chemistry
Curated by ChEMBL
| Assay Description
Inhibitory potency was determined by competitive inhibition of Adenosine deaminase |
J Med Chem 38: 4648-59 (1995)
BindingDB Entry DOI: 10.7270/Q2V40T7T |
More data for this
Ligand-Target Pair | |
Adenosine deaminase
(Bos taurus (bovine)) | BDBM50369958
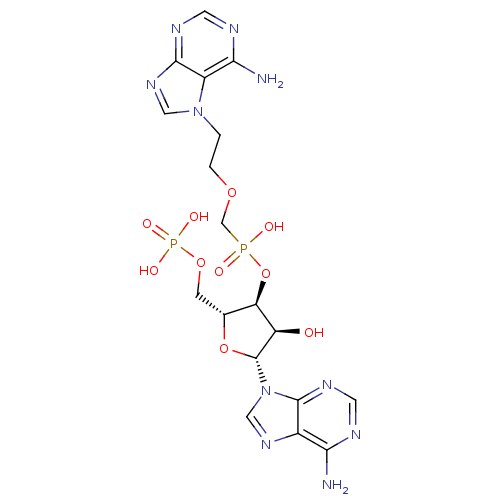
(CHEMBL1790862)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(O)=O)[C@@H](OP(O)(=O)COCCn2cnc3ncnc(N)c23)[C@H]1O Show InChI InChI=1S/C18H24N10O10P2/c19-14-10-17(24-5-21-14)28(7-25-10)18-12(29)13(9(37-18)3-36-40(32,33)34)38-39(30,31)8-35-2-1-27-6-26-16-11(27)15(20)22-4-23-16/h4-7,9,12-13,18,29H,1-3,8H2,(H,30,31)(H2,19,21,24)(H2,20,22,23)(H2,32,33,34)/t9-,12-,13-,18-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Chemistry
Curated by ChEMBL
| Assay Description
Inhibitory activity against adenosine deaminase |
J Med Chem 44: 3710-20 (2001)
BindingDB Entry DOI: 10.7270/Q2G44R0Z |
More data for this
Ligand-Target Pair | |
Adenosine deaminase
(Bos taurus (bovine)) | BDBM50029650

(2-(6-Amino-purin-9-ylmethoxy)-ethanol | CHEMBL3775...)Show InChI InChI=1S/C8H11N5O2/c9-7-6-8(11-3-10-7)13(4-12-6)5-15-2-1-14/h3-4,14H,1-2,5H2,(H2,9,10,11) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.40E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Chemistry
Curated by ChEMBL
| Assay Description
Inhibitory activity against adenosine deaminase |
J Med Chem 44: 3710-20 (2001)
BindingDB Entry DOI: 10.7270/Q2G44R0Z |
More data for this
Ligand-Target Pair | |
Adenosine deaminase
(Bos taurus (bovine)) | BDBM50029650

(2-(6-Amino-purin-9-ylmethoxy)-ethanol | CHEMBL3775...)Show InChI InChI=1S/C8H11N5O2/c9-7-6-8(11-3-10-7)13(4-12-6)5-15-2-1-14/h3-4,14H,1-2,5H2,(H2,9,10,11) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.43E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Chemistry
Curated by ChEMBL
| Assay Description
Inhibitory potency was determined by competitive inhibition of Adenosine deaminase |
J Med Chem 38: 4648-59 (1995)
BindingDB Entry DOI: 10.7270/Q2V40T7T |
More data for this
Ligand-Target Pair | |
Adenosine deaminase
(Bos taurus (bovine)) | BDBM50099681

(5-[2-(6-Amino-purin-9-yl)-ethylidene]-3,4-dimethox...)Show SMILES COC1=C(OC)\C(OC1=O)=C\Cn1cnc2c(N)ncnc12 |c:2| Show InChI InChI=1S/C13H13N5O4/c1-20-9-7(22-13(19)10(9)21-2)3-4-18-6-17-8-11(14)15-5-16-12(8)18/h3,5-6H,4H2,1-2H3,(H2,14,15,16)/b7-3- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 3.16E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Chemistry
Curated by ChEMBL
| Assay Description
Rate of deamination in the presence of calf mucosal adenosine deaminase (ADA) |
J Med Chem 44: 1749-57 (2001)
BindingDB Entry DOI: 10.7270/Q21V5D7J |
More data for this
Ligand-Target Pair | |
Phosphoribosyl pyrophosphate synthase-associated protein 2
(Homo sapiens (Human)) | BDBM50369958

(CHEMBL1790862)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(O)=O)[C@@H](OP(O)(=O)COCCn2cnc3ncnc(N)c23)[C@H]1O Show InChI InChI=1S/C18H24N10O10P2/c19-14-10-17(24-5-21-14)28(7-25-10)18-12(29)13(9(37-18)3-36-40(32,33)34)38-39(30,31)8-35-2-1-27-6-26-16-11(27)15(20)22-4-23-16/h4-7,9,12-13,18,29H,1-3,8H2,(H,30,31)(H2,19,21,24)(H2,20,22,23)(H2,32,33,34)/t9-,12-,13-,18-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 7.90E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Chemistry
Curated by ChEMBL
| Assay Description
Inhibitory activity against PRPP synthetase |
J Med Chem 44: 3710-20 (2001)
BindingDB Entry DOI: 10.7270/Q2G44R0Z |
More data for this
Ligand-Target Pair | |
Adenosine deaminase
(Bos taurus (bovine)) | BDBM50369957
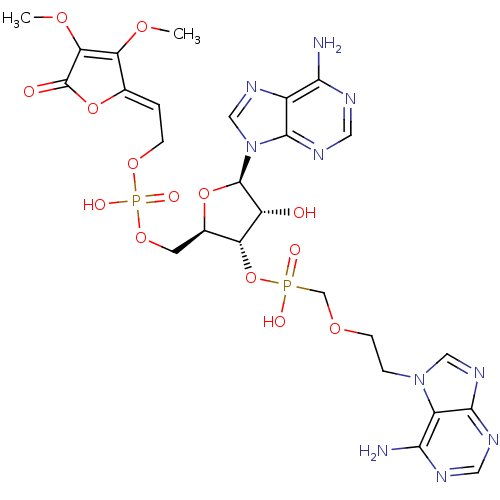
(CHEMBL1790864)Show SMILES COC1=C(OC)\C(OC1=O)=C\COP(O)(=O)OC[C@H]1O[C@H]([C@H](O)[C@@H]1OP(O)(=O)COCCn1cnc2ncnc(N)c12)n1cnc2c(N)ncnc12 |c:2| Show InChI InChI=1S/C26H32N10O14P2/c1-43-19-13(49-26(38)20(19)44-2)3-5-46-52(41,42)47-7-14-18(17(37)25(48-14)36-11-33-15-21(27)29-9-32-24(15)36)50-51(39,40)12-45-6-4-35-10-34-23-16(35)22(28)30-8-31-23/h3,8-11,14,17-18,25,37H,4-7,12H2,1-2H3,(H,39,40)(H,41,42)(H2,27,29,32)(H2,28,30,31)/b13-3-/t14-,17-,18-,25-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| >8.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Chemistry
Curated by ChEMBL
| Assay Description
Inhibitory activity against adenosine deaminase |
J Med Chem 44: 3710-20 (2001)
BindingDB Entry DOI: 10.7270/Q2G44R0Z |
More data for this
Ligand-Target Pair | |
Adenosine deaminase
(Bos taurus (bovine)) | BDBM50105931
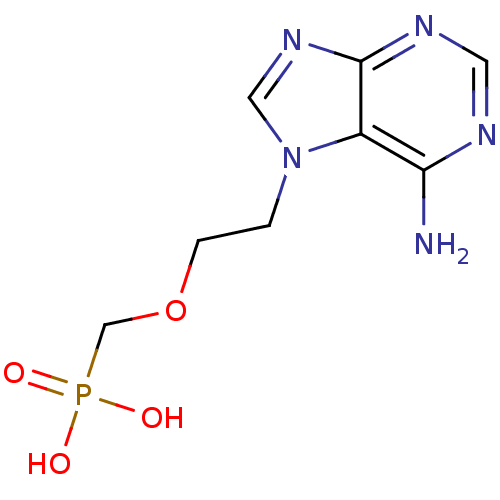
(CHEMBL123655 | [2-(6-Amino-purin-7-yl)-ethoxymethy...)Show InChI InChI=1S/C8H12N5O4P/c9-7-6-8(11-3-10-7)12-4-13(6)1-2-17-5-18(14,15)16/h3-4H,1-2,5H2,(H2,9,10,11)(H2,14,15,16) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >8.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Chemistry
Curated by ChEMBL
| Assay Description
Inhibitory activity against adenosine deaminase |
J Med Chem 44: 3710-20 (2001)
BindingDB Entry DOI: 10.7270/Q2G44R0Z |
More data for this
Ligand-Target Pair | |
Adenosine deaminase
(Bos taurus (bovine)) | BDBM50099682
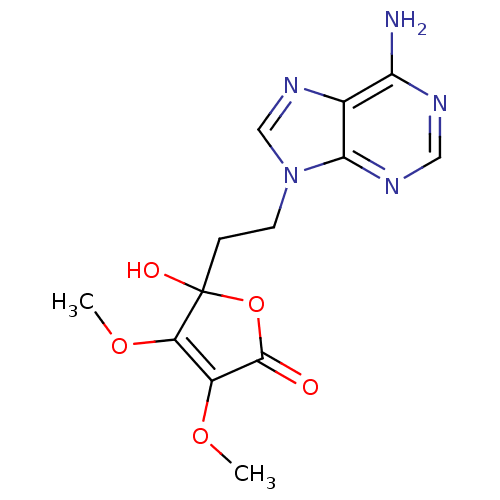
(5-(2-(6-amino-9H-purin-9-yl)ethyl)-5-hydroxy-3,4-d...)Show SMILES COC1=C(OC)C(O)(CCn2cnc3c(N)ncnc23)OC1=O |c:2| Show InChI InChI=1S/C13H15N5O5/c1-21-8-9(22-2)13(20,23-12(8)19)3-4-18-6-17-7-10(14)15-5-16-11(7)18/h5-6,20H,3-4H2,1-2H3,(H2,14,15,16) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| >8.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Chemistry
Curated by ChEMBL
| Assay Description
Rate of deamination in the presence of calf mucosal adenosine deaminase (ADA) |
J Med Chem 44: 1749-57 (2001)
BindingDB Entry DOI: 10.7270/Q21V5D7J |
More data for this
Ligand-Target Pair | |
Adenosine deaminase
(Bos taurus (bovine)) | BDBM50105935

(CHEMBL121723 | [2-(6-Amino-purin-9-yl)-ethoxymethy...)Show SMILES COC1=C(OC)\C(OC1=O)=C\COP(O)(=O)COCCn1cnc2c(N)ncnc12 |c:2| Show InChI InChI=1S/C16H20N5O8P/c1-25-12-10(29-16(22)13(12)26-2)3-5-28-30(23,24)9-27-6-4-21-8-20-11-14(17)18-7-19-15(11)21/h3,7-8H,4-6,9H2,1-2H3,(H,23,24)(H2,17,18,19)/b10-3- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| >8.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Chemistry
Curated by ChEMBL
| Assay Description
Inhibitory activity against adenosine deaminase |
J Med Chem 44: 3710-20 (2001)
BindingDB Entry DOI: 10.7270/Q2G44R0Z |
More data for this
Ligand-Target Pair | |
Adenosine deaminase
(Bos taurus (bovine)) | BDBM50001103
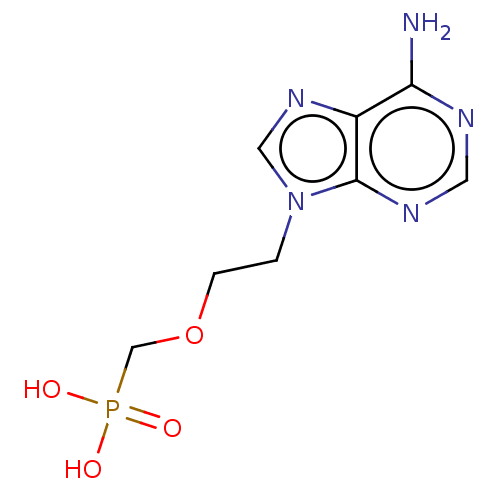
((2-(6-amino-9H-purin-9-yl)ethoxy)methylphosphonic ...)Show InChI InChI=1S/C8H12N5O4P/c9-7-6-8(11-3-10-7)13(4-12-6)1-2-17-5-18(14,15)16/h3-4H,1-2,5H2,(H2,9,10,11)(H2,14,15,16) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| >8.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Chemistry
Curated by ChEMBL
| Assay Description
Inhibitory activity against adenosine deaminase |
J Med Chem 44: 3710-20 (2001)
BindingDB Entry DOI: 10.7270/Q2G44R0Z |
More data for this
Ligand-Target Pair | |
Phosphoribosyl pyrophosphate synthase-associated protein 2
(Homo sapiens (Human)) | BDBM50001103

((2-(6-amino-9H-purin-9-yl)ethoxy)methylphosphonic ...)Show InChI InChI=1S/C8H12N5O4P/c9-7-6-8(11-3-10-7)13(4-12-6)1-2-17-5-18(14,15)16/h3-4H,1-2,5H2,(H2,9,10,11)(H2,14,15,16) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 3.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Chemistry
Curated by ChEMBL
| Assay Description
Inhibitory activity against PRPP synthetase |
J Med Chem 44: 3710-20 (2001)
BindingDB Entry DOI: 10.7270/Q2G44R0Z |
More data for this
Ligand-Target Pair | |
Phosphoribosyl pyrophosphate synthase-associated protein 2
(Homo sapiens (Human)) | BDBM50105931

(CHEMBL123655 | [2-(6-Amino-purin-7-yl)-ethoxymethy...)Show InChI InChI=1S/C8H12N5O4P/c9-7-6-8(11-3-10-7)12-4-13(6)1-2-17-5-18(14,15)16/h3-4H,1-2,5H2,(H2,9,10,11)(H2,14,15,16) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.70E+7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Chemistry
Curated by ChEMBL
| Assay Description
Inhibitory activity against PRPP synthetase |
J Med Chem 44: 3710-20 (2001)
BindingDB Entry DOI: 10.7270/Q2G44R0Z |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50559579
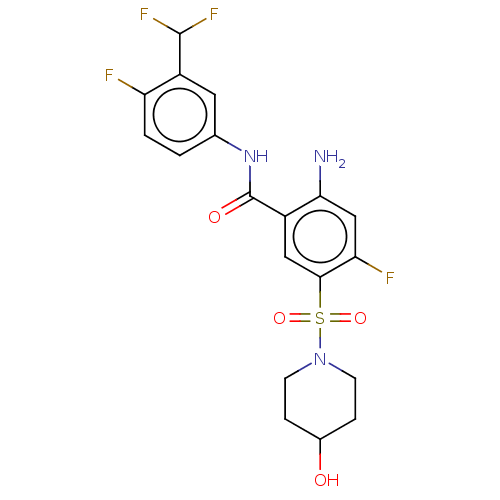
(CHEMBL4754334)Show SMILES Nc1cc(F)c(cc1C(=O)Nc1ccc(F)c(c1)C(F)F)S(=O)(=O)N1CCC(O)CC1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human ERG by fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.0c00606
BindingDB Entry DOI: 10.7270/Q2SJ1Q9C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50559580
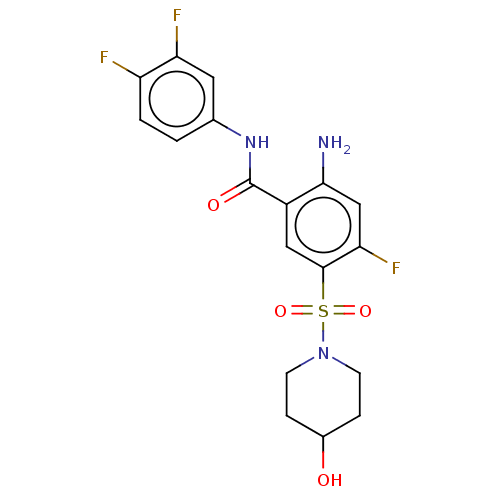
(CHEMBL4761323)Show SMILES Nc1cc(F)c(cc1C(=O)Nc1ccc(F)c(F)c1)S(=O)(=O)N1CCC(O)CC1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.83E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human liver microsome CYP2C19 using S-mephenytoin as substrate incubated for 20 mins by LC-MS/MS with HPLC analysis |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.0c00606
BindingDB Entry DOI: 10.7270/Q2SJ1Q9C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50559579

(CHEMBL4754334)Show SMILES Nc1cc(F)c(cc1C(=O)Nc1ccc(F)c(c1)C(F)F)S(=O)(=O)N1CCC(O)CC1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human liver microsome CYP2C19 using S-mephenytoin as substrate incubated for 20 mins by LC-MS/MS with HPLC analysis |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.0c00606
BindingDB Entry DOI: 10.7270/Q2SJ1Q9C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50559579

(CHEMBL4754334)Show SMILES Nc1cc(F)c(cc1C(=O)Nc1ccc(F)c(c1)C(F)F)S(=O)(=O)N1CCC(O)CC1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.08E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human liver microsome CYP3A4 using sorafenib as substrate incubated for 20 mins by LC-MS/MS with HPLC analysis |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.0c00606
BindingDB Entry DOI: 10.7270/Q2SJ1Q9C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50559580

(CHEMBL4761323)Show SMILES Nc1cc(F)c(cc1C(=O)Nc1ccc(F)c(F)c1)S(=O)(=O)N1CCC(O)CC1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human liver microsome CYP2C9 using tolbutamide as substrate incubated for 20 mins by LC-MS/MS with HPLC analysis |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.0c00606
BindingDB Entry DOI: 10.7270/Q2SJ1Q9C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50559579

(CHEMBL4754334)Show SMILES Nc1cc(F)c(cc1C(=O)Nc1ccc(F)c(c1)C(F)F)S(=O)(=O)N1CCC(O)CC1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.52E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human liver microsome CYP2C9 using tolbutamide as substrate incubated for 20 mins by LC-MS/MS with HPLC analysis |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.0c00606
BindingDB Entry DOI: 10.7270/Q2SJ1Q9C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50559579

(CHEMBL4754334)Show SMILES Nc1cc(F)c(cc1C(=O)Nc1ccc(F)c(c1)C(F)F)S(=O)(=O)N1CCC(O)CC1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.67E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human liver microsome CYP2D6 using dextromethorphan as substrate incubated for 20 mins by LC-MS/MS with HPLC analysis |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.0c00606
BindingDB Entry DOI: 10.7270/Q2SJ1Q9C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50559580

(CHEMBL4761323)Show SMILES Nc1cc(F)c(cc1C(=O)Nc1ccc(F)c(F)c1)S(=O)(=O)N1CCC(O)CC1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.93E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human liver microsome CYP2D6 using dextromethorphan as substrate incubated for 20 mins by LC-MS/MS with HPLC analysis |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.0c00606
BindingDB Entry DOI: 10.7270/Q2SJ1Q9C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50559580

(CHEMBL4761323)Show SMILES Nc1cc(F)c(cc1C(=O)Nc1ccc(F)c(F)c1)S(=O)(=O)N1CCC(O)CC1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human liver microsome CYP3A4 using sorafenib as substrate incubated for 20 mins by LC-MS/MS with HPLC analysis |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.0c00606
BindingDB Entry DOI: 10.7270/Q2SJ1Q9C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50559579

(CHEMBL4754334)Show SMILES Nc1cc(F)c(cc1C(=O)Nc1ccc(F)c(c1)C(F)F)S(=O)(=O)N1CCC(O)CC1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4.55E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human liver microsome CYP1A2 using phenacetin as substrate incubated for 20 mins by LC-MS/MS with HPLC analysis |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.0c00606
BindingDB Entry DOI: 10.7270/Q2SJ1Q9C |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50559580

(CHEMBL4761323)Show SMILES Nc1cc(F)c(cc1C(=O)Nc1ccc(F)c(F)c1)S(=O)(=O)N1CCC(O)CC1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human ERG by fluorescence polarization assay |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.0c00606
BindingDB Entry DOI: 10.7270/Q2SJ1Q9C |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50559580

(CHEMBL4761323)Show SMILES Nc1cc(F)c(cc1C(=O)Nc1ccc(F)c(F)c1)S(=O)(=O)N1CCC(O)CC1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human liver microsome CYP1A2 using phenacetin as substrate incubated for 20 mins by LC-MS/MS with HPLC analysis |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.0c00606
BindingDB Entry DOI: 10.7270/Q2SJ1Q9C |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data