 Found 241 hits with Last Name = 'suarez' and Initial = 'j'
Found 241 hits with Last Name = 'suarez' and Initial = 'j' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
5'-methylthioadenosine/S-adenosylhomocysteine nucleosidase
(Escherichia coli) | BDBM22113
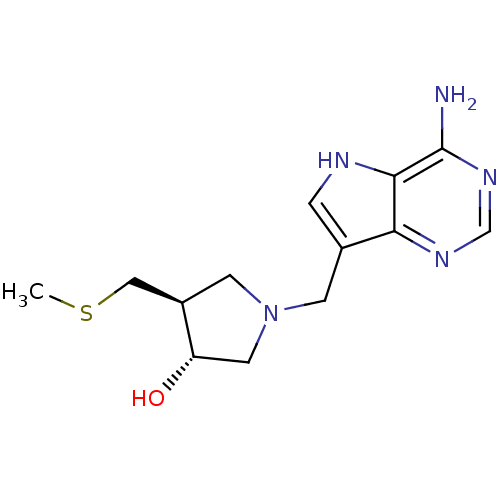
((3R,4S)-1-({4-amino-5H-pyrrolo[3,2-d]pyrimidin-7-y...)Show SMILES CSC[C@H]1CN(Cc2c[nH]c3c(N)ncnc23)C[C@@H]1O |r| Show InChI InChI=1S/C13H19N5OS/c1-20-6-9-4-18(5-10(9)19)3-8-2-15-12-11(8)16-7-17-13(12)14/h2,7,9-10,15,19H,3-6H2,1H3,(H2,14,16,17)/t9-,10+/m1/s1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.00200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Victoria University of Wellington
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Escherichia coli MTAN expressed in Escherichia coli BL-21 DE3 using methylthioadenosine as substrate assessed as inhibition... |
Bioorg Med Chem 23: 5326-33 (2015)
Article DOI: 10.1016/j.bmc.2015.07.059
BindingDB Entry DOI: 10.7270/Q2HX1FF5 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
5'-methylthioadenosine/S-adenosylhomocysteine nucleosidase
(Escherichia coli) | BDBM22113

((3R,4S)-1-({4-amino-5H-pyrrolo[3,2-d]pyrimidin-7-y...)Show SMILES CSC[C@H]1CN(Cc2c[nH]c3c(N)ncnc23)C[C@@H]1O |r| Show InChI InChI=1S/C13H19N5OS/c1-20-6-9-4-18(5-10(9)19)3-8-2-15-12-11(8)16-7-17-13(12)14/h2,7,9-10,15,19H,3-6H2,1H3,(H2,14,16,17)/t9-,10+/m1/s1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.0480 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Victoria University of Wellington
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Escherichia coli MTAN expressed in Escherichia coli BL-21 DE3 using methylthioadenosine as substrate by xanthine oxidase co... |
Bioorg Med Chem 23: 5326-33 (2015)
Article DOI: 10.1016/j.bmc.2015.07.059
BindingDB Entry DOI: 10.7270/Q2HX1FF5 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Purine nucleoside phosphorylase
(Homo sapiens (Human)) | BDBM50247151
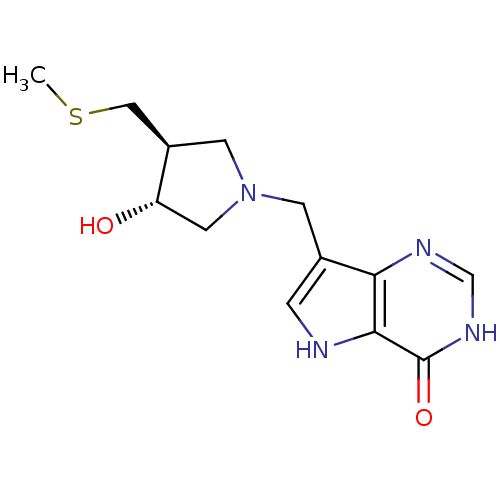
(7-(((3R,4S)-3-hydroxy-4-(methylthiomethyl)pyrrolid...)Show SMILES CSC[C@H]1CN(Cc2c[nH]c3c2nc[nH]c3=O)C[C@@H]1O |r| Show InChI InChI=1S/C13H18N4O2S/c1-20-6-9-4-17(5-10(9)18)3-8-2-14-12-11(8)15-7-16-13(12)19/h2,7,9-10,14,18H,3-6H2,1H3,(H,15,16,19)/t9-,10+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Victoria University of Wellington
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PNP using inosine as substrate assessed as inhibition constant for slow onset inhibition of enzyme-inhibitor complex ... |
Bioorg Med Chem 23: 5326-33 (2015)
Article DOI: 10.1016/j.bmc.2015.07.059
BindingDB Entry DOI: 10.7270/Q2HX1FF5 |
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM22113

((3R,4S)-1-({4-amino-5H-pyrrolo[3,2-d]pyrimidin-7-y...)Show SMILES CSC[C@H]1CN(Cc2c[nH]c3c(N)ncnc23)C[C@@H]1O |r| Show InChI InChI=1S/C13H19N5OS/c1-20-6-9-4-18(5-10(9)19)3-8-2-15-12-11(8)16-7-17-13(12)14/h2,7,9-10,15,19H,3-6H2,1H3,(H2,14,16,17)/t9-,10+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.0780 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Victoria University of Wellington
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP using methylthioadenosine as substrate assessed as inhibition constant for slow onset inhibition of enzyme-inhibitor complex... |
Bioorg Med Chem 23: 5326-33 (2015)
Article DOI: 10.1016/j.bmc.2015.07.059
BindingDB Entry DOI: 10.7270/Q2HX1FF5 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Purine nucleoside phosphorylase
(Homo sapiens (Human)) | BDBM50247151

(7-(((3R,4S)-3-hydroxy-4-(methylthiomethyl)pyrrolid...)Show SMILES CSC[C@H]1CN(Cc2c[nH]c3c2nc[nH]c3=O)C[C@@H]1O |r| Show InChI InChI=1S/C13H18N4O2S/c1-20-6-9-4-17(5-10(9)18)3-8-2-14-12-11(8)15-7-16-13(12)19/h2,7,9-10,14,18H,3-6H2,1H3,(H,15,16,19)/t9-,10+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Victoria University of Wellington
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PNP using inosine as substrate by xanthine oxidase coupling enzyme assay |
Bioorg Med Chem 23: 5326-33 (2015)
Article DOI: 10.1016/j.bmc.2015.07.059
BindingDB Entry DOI: 10.7270/Q2HX1FF5 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM11639

(4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyraz...)Show SMILES Cc1ccc(cc1)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C17H14F3N3O2S/c1-11-2-4-12(5-3-11)15-10-16(17(18,19)20)22-23(15)13-6-8-14(9-7-13)26(21,24)25/h2-10H,1H3,(H2,21,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| 0.470 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human recombinant COX-2 using arachidonic acid as substrate preincubated for 60 mins followed by substrate addition for 2 secs by ADPH ... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00890
BindingDB Entry DOI: 10.7270/Q24B352D |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM22113

((3R,4S)-1-({4-amino-5H-pyrrolo[3,2-d]pyrimidin-7-y...)Show SMILES CSC[C@H]1CN(Cc2c[nH]c3c(N)ncnc23)C[C@@H]1O |r| Show InChI InChI=1S/C13H19N5OS/c1-20-6-9-4-18(5-10(9)19)3-8-2-15-12-11(8)16-7-17-13(12)14/h2,7,9-10,15,19H,3-6H2,1H3,(H2,14,16,17)/t9-,10+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.530 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Victoria University of Wellington
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP using methylthioadenosine as substrate by xanthine oxidase coupling enzyme assay |
Bioorg Med Chem 23: 5326-33 (2015)
Article DOI: 10.1016/j.bmc.2015.07.059
BindingDB Entry DOI: 10.7270/Q2HX1FF5 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Purine nucleoside phosphorylase
(Plasmodium falciparum) | BDBM50247151

(7-(((3R,4S)-3-hydroxy-4-(methylthiomethyl)pyrrolid...)Show SMILES CSC[C@H]1CN(Cc2c[nH]c3c2nc[nH]c3=O)C[C@@H]1O |r| Show InChI InChI=1S/C13H18N4O2S/c1-20-6-9-4-17(5-10(9)18)3-8-2-14-12-11(8)15-7-16-13(12)19/h2,7,9-10,14,18H,3-6H2,1H3,(H,15,16,19)/t9-,10+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Victoria University of Wellington
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Plasmodium falciparum PNP using inosine as substrate assessed as inhibition constant for slow onset inhibition of enzyme-in... |
Bioorg Med Chem 23: 5326-33 (2015)
Article DOI: 10.1016/j.bmc.2015.07.059
BindingDB Entry DOI: 10.7270/Q2HX1FF5 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50566910
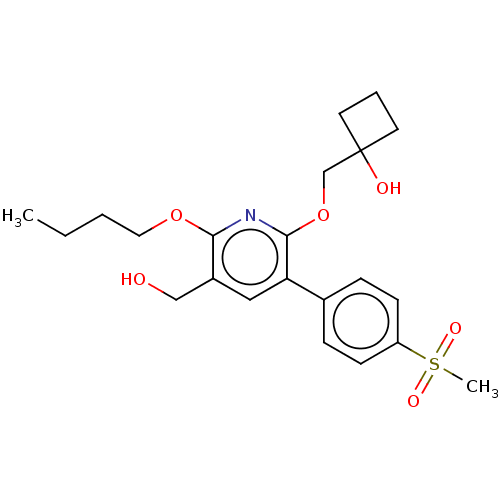
(CHEMBL4870569)Show SMILES CCCCOc1nc(OCC2(O)CCC2)c(cc1CO)-c1ccc(cc1)S(C)(=O)=O | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human recombinant COX-2 using arachidonic acid as substrate preincubated for 60 mins followed by substrate addition for 2 secs by ADPH ... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00890
BindingDB Entry DOI: 10.7270/Q24B352D |
More data for this
Ligand-Target Pair | |
5'-methylthioadenosine/S-adenosylhomocysteine nucleosidase
(Escherichia coli) | BDBM50116357
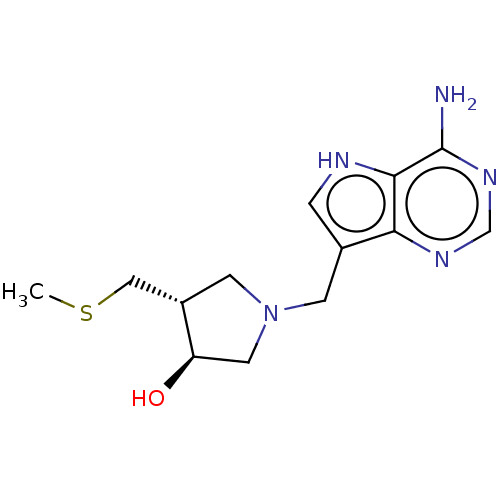
(CHEMBL3604360)Show SMILES CSC[C@@H]1CN(Cc2c[nH]c3c(N)ncnc23)C[C@H]1O |r| Show InChI InChI=1S/C13H19N5OS/c1-20-6-9-4-18(5-10(9)19)3-8-2-15-12-11(8)16-7-17-13(12)14/h2,7,9-10,15,19H,3-6H2,1H3,(H2,14,16,17)/t9-,10+/m0/s1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 4.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Victoria University of Wellington
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Escherichia coli MTAN expressed in Escherichia coli BL-21 DE3 using methylthioadenosine as substrate by xanthine oxidase co... |
Bioorg Med Chem 23: 5326-33 (2015)
Article DOI: 10.1016/j.bmc.2015.07.059
BindingDB Entry DOI: 10.7270/Q2HX1FF5 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Monocarboxylate transporter 4
(Homo sapiens (Human)) | BDBM50610833

(CHEMBL5276884)Show SMILES COc1ccc(c2ncccc12)S(=O)(=O)Nc1ccccc1C#Cc1cnc(C(O)=O)c(C)c1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 4
(Homo sapiens (Human)) | BDBM50610836

(MSC-4381)Show SMILES CCOc1ccc(c2ncccc12)S(=O)(=O)Nc1ccc(Cl)cc1C#Cc1cnc(cc1OC)C(O)=O | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Purine nucleoside phosphorylase
(Plasmodium falciparum) | BDBM50247151

(7-(((3R,4S)-3-hydroxy-4-(methylthiomethyl)pyrrolid...)Show SMILES CSC[C@H]1CN(Cc2c[nH]c3c2nc[nH]c3=O)C[C@@H]1O |r| Show InChI InChI=1S/C13H18N4O2S/c1-20-6-9-4-17(5-10(9)18)3-8-2-14-12-11(8)15-7-16-13(12)19/h2,7,9-10,14,18H,3-6H2,1H3,(H,15,16,19)/t9-,10+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Victoria University of Wellington
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Plasmodium falciparum PNP using inosine as substrate by xanthine oxidase coupling enzyme assay |
Bioorg Med Chem 23: 5326-33 (2015)
Article DOI: 10.1016/j.bmc.2015.07.059
BindingDB Entry DOI: 10.7270/Q2HX1FF5 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50566901
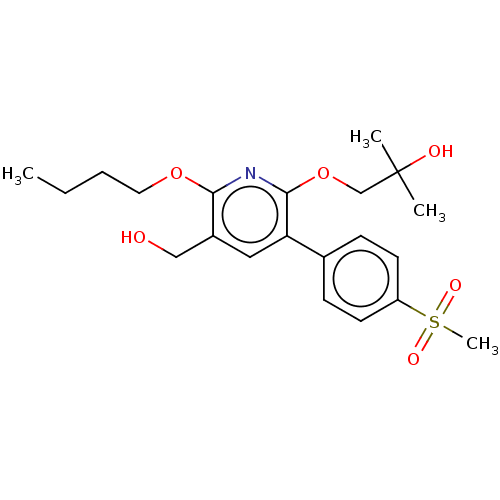
(CHEMBL4865464)Show SMILES CCCCOc1nc(OCC(C)(C)O)c(cc1CO)-c1ccc(cc1)S(C)(=O)=O | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human recombinant COX-2 using arachidonic acid as substrate preincubated for 60 mins followed by substrate addition for 2 secs by ADPH ... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00890
BindingDB Entry DOI: 10.7270/Q24B352D |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50566912
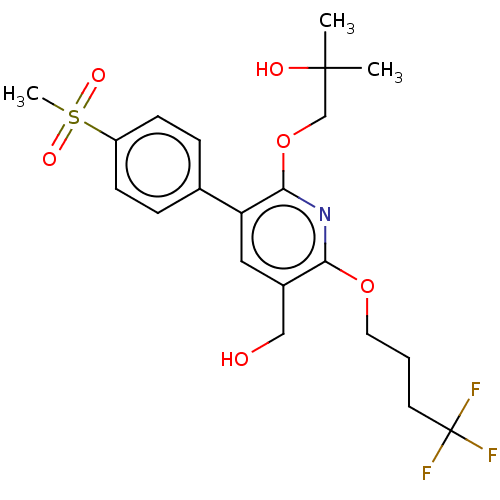
(CHEMBL4859571)Show SMILES CC(C)(O)COc1nc(OCCCC(F)(F)F)c(CO)cc1-c1ccc(cc1)S(C)(=O)=O | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human recombinant COX-2 using arachidonic acid as substrate preincubated for 60 mins followed by substrate addition for 2 secs by ADPH ... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00890
BindingDB Entry DOI: 10.7270/Q24B352D |
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 4
(Homo sapiens (Human)) | BDBM50610834
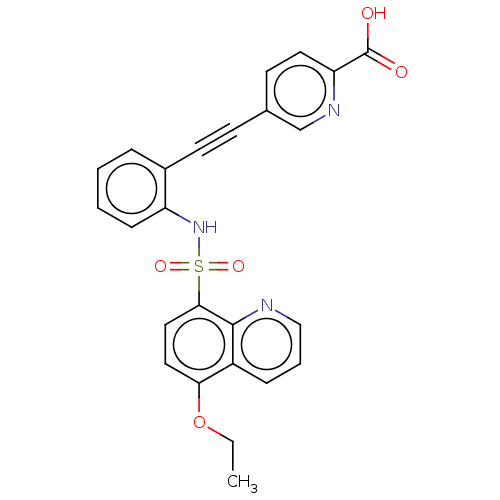
(CHEMBL5279064)Show SMILES CCOc1ccc(c2ncccc12)S(=O)(=O)Nc1ccccc1C#Cc1ccc(nc1)C(O)=O | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 4
(Homo sapiens (Human)) | BDBM50610835

(CHEMBL5267349)Show SMILES CCOc1ccc(c2ncccc12)S(=O)(=O)Nc1ccccc1C#Cc1cnc(cc1OC)C(O)=O | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 17 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 4
(Homo sapiens (Human)) | BDBM50610837
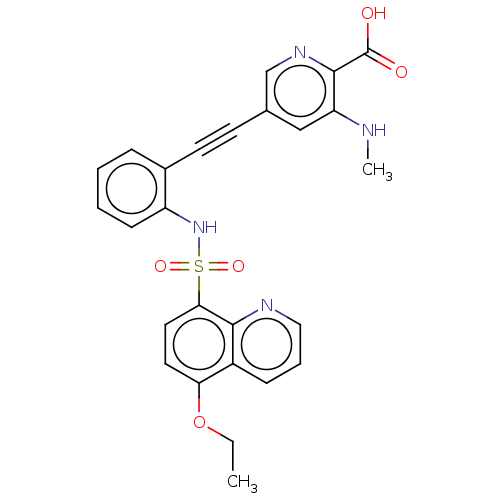
(CHEMBL5281492)Show SMILES CCOc1ccc(c2ncccc12)S(=O)(=O)Nc1ccccc1C#Cc1cnc(C(O)=O)c(NC)c1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 17 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 4
(Homo sapiens (Human)) | BDBM50610830
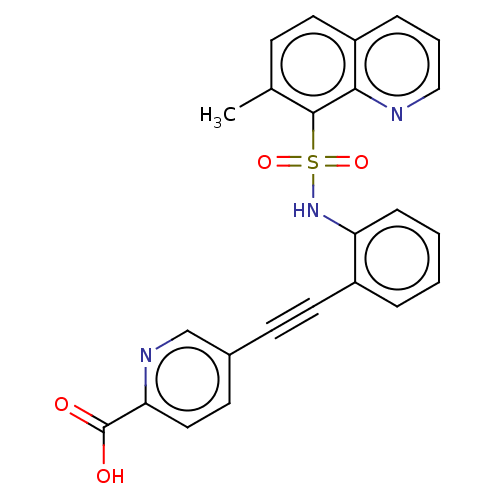
(CHEMBL5267752)Show SMILES Cc1ccc2cccnc2c1S(=O)(=O)Nc1ccccc1C#Cc1ccc(nc1)C(O)=O | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 17 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50566913

(CHEMBL4857474)Show SMILES CCCCOc1nc(OCC(C)(C)O)c(cc1CO)-c1ccc(cn1)S(N)(=O)=O | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 19 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human recombinant COX-2 using arachidonic acid as substrate preincubated for 60 mins followed by substrate addition for 2 secs by ADPH ... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00890
BindingDB Entry DOI: 10.7270/Q24B352D |
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 4
(Homo sapiens (Human)) | BDBM50610826

(CHEMBL5287351)Show SMILES Cc1cnc2c(cccc2c1)S(=O)(=O)Nc1ccccc1C#Cc1ccc(nc1)C(O)=O | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 29 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50566911
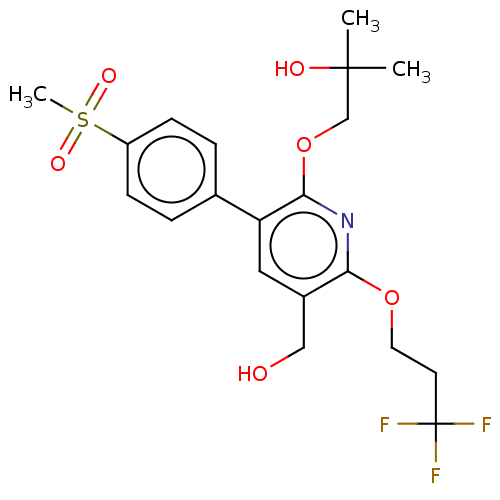
(CHEMBL4868446)Show SMILES CC(C)(O)COc1nc(OCCC(F)(F)F)c(CO)cc1-c1ccc(cc1)S(C)(=O)=O | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 32 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human recombinant COX-2 using arachidonic acid as substrate preincubated for 60 mins followed by substrate addition for 2 secs by ADPH ... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00890
BindingDB Entry DOI: 10.7270/Q24B352D |
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 4
(Homo sapiens (Human)) | BDBM50610827
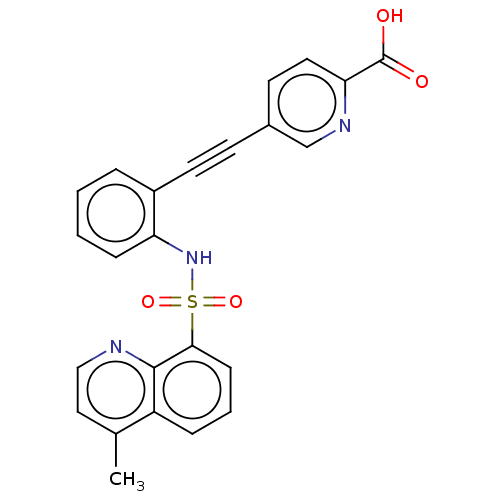
(CHEMBL5287780)Show SMILES Cc1ccnc2c(cccc12)S(=O)(=O)Nc1ccccc1C#Cc1ccc(nc1)C(O)=O | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 32 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 4
(Homo sapiens (Human)) | BDBM50610824

(CHEMBL5268966)Show SMILES OC(=O)c1ccc(cn1)C#Cc1ccccc1NS(=O)(=O)c1cccc2cccnc12 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 37 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 4
(Homo sapiens (Human)) | BDBM50610829
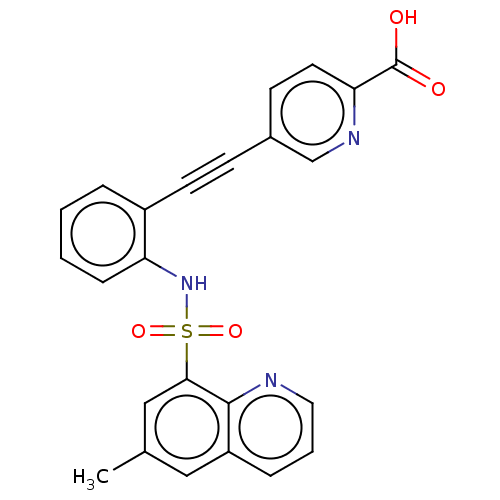
(CHEMBL5288904)Show SMILES Cc1cc(c2ncccc2c1)S(=O)(=O)Nc1ccccc1C#Cc1ccc(nc1)C(O)=O | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 51 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 4
(Homo sapiens (Human)) | BDBM50610831
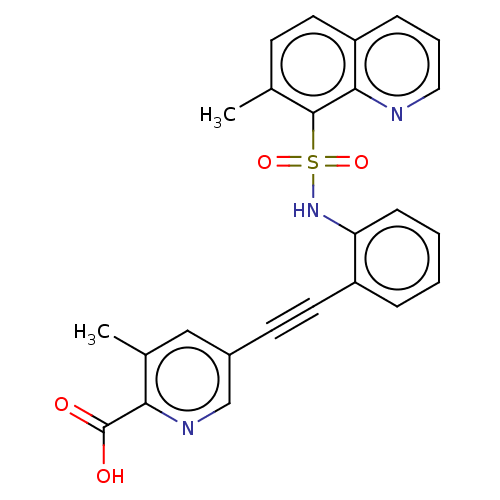
(CHEMBL5265956)Show SMILES Cc1ccc2cccnc2c1S(=O)(=O)Nc1ccccc1C#Cc1cnc(C(O)=O)c(C)c1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 56 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50072064
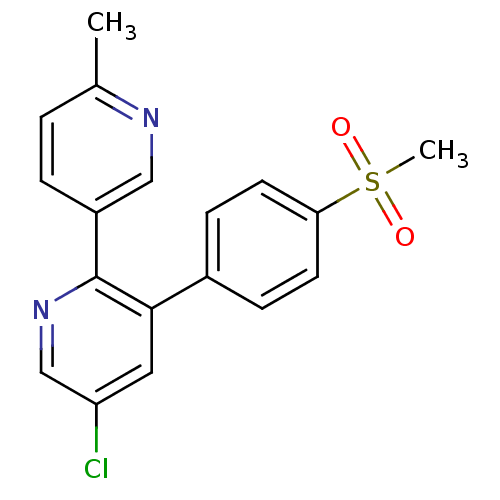
(5-Chloro-3-(4-methanesulfonyl-phenyl)-6''-methyl-[...)Show SMILES Cc1ccc(cn1)-c1ncc(Cl)cc1-c1ccc(cc1)S(C)(=O)=O Show InChI InChI=1S/C18H15ClN2O2S/c1-12-3-4-14(10-20-12)18-17(9-15(19)11-21-18)13-5-7-16(8-6-13)24(2,22)23/h3-11H,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 69 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human recombinant COX-2 using arachidonic acid as substrate preincubated for 60 mins followed by substrate addition for 2 secs by ADPH ... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00890
BindingDB Entry DOI: 10.7270/Q24B352D |
More data for this
Ligand-Target Pair | |
Purine nucleoside phosphorylase
(Homo sapiens (Human)) | BDBM50116358
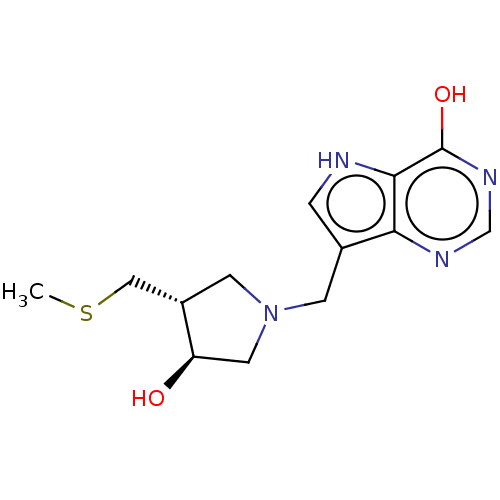
(CHEMBL3604359)Show SMILES CSC[C@@H]1CN(Cc2c[nH]c3c(O)ncnc23)C[C@H]1O |r| Show InChI InChI=1S/C13H18N4O2S/c1-20-6-9-4-17(5-10(9)18)3-8-2-14-12-11(8)15-7-16-13(12)19/h2,7,9-10,14,18H,3-6H2,1H3,(H,15,16,19)/t9-,10+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 84 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Victoria University of Wellington
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PNP using inosine as substrate assessed as inhibition constant for slow onset inhibition of enzyme-inhibitor complex ... |
Bioorg Med Chem 23: 5326-33 (2015)
Article DOI: 10.1016/j.bmc.2015.07.059
BindingDB Entry DOI: 10.7270/Q2HX1FF5 |
More data for this
Ligand-Target Pair | |
Purine nucleoside phosphorylase
(Plasmodium falciparum) | BDBM50116358

(CHEMBL3604359)Show SMILES CSC[C@@H]1CN(Cc2c[nH]c3c(O)ncnc23)C[C@H]1O |r| Show InChI InChI=1S/C13H18N4O2S/c1-20-6-9-4-17(5-10(9)18)3-8-2-14-12-11(8)15-7-16-13(12)19/h2,7,9-10,14,18H,3-6H2,1H3,(H,15,16,19)/t9-,10+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 93 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Victoria University of Wellington
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Plasmodium falciparum PNP using inosine as substrate assessed as inhibition constant for slow onset inhibition of enzyme-in... |
Bioorg Med Chem 23: 5326-33 (2015)
Article DOI: 10.1016/j.bmc.2015.07.059
BindingDB Entry DOI: 10.7270/Q2HX1FF5 |
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 4
(Homo sapiens (Human)) | BDBM50610825

(CHEMBL5266104)Show SMILES Cc1ccc2cccc(c2n1)S(=O)(=O)Nc1ccccc1C#Cc1ccc(nc1)C(O)=O | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 99 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Purine nucleoside phosphorylase
(Homo sapiens (Human)) | BDBM50116358

(CHEMBL3604359)Show SMILES CSC[C@@H]1CN(Cc2c[nH]c3c(O)ncnc23)C[C@H]1O |r| Show InChI InChI=1S/C13H18N4O2S/c1-20-6-9-4-17(5-10(9)18)3-8-2-14-12-11(8)15-7-16-13(12)19/h2,7,9-10,14,18H,3-6H2,1H3,(H,15,16,19)/t9-,10+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Victoria University of Wellington
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PNP using inosine as substrate by xanthine oxidase coupling enzyme assay |
Bioorg Med Chem 23: 5326-33 (2015)
Article DOI: 10.1016/j.bmc.2015.07.059
BindingDB Entry DOI: 10.7270/Q2HX1FF5 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50566900
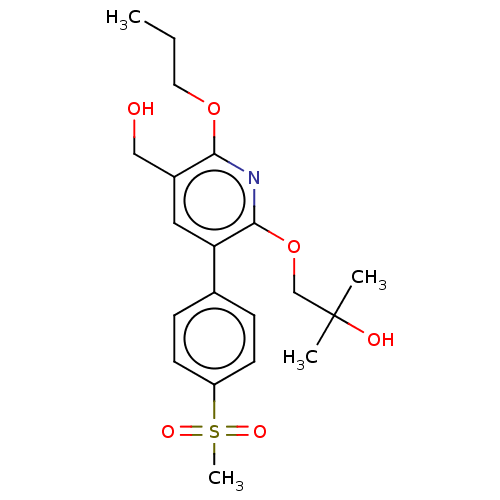
(CHEMBL4862802)Show SMILES CCCOc1nc(OCC(C)(C)O)c(cc1CO)-c1ccc(cc1)S(C)(=O)=O | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 114 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human recombinant COX-2 using arachidonic acid as substrate preincubated for 60 mins followed by substrate addition for 2 secs by ADPH ... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00890
BindingDB Entry DOI: 10.7270/Q24B352D |
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 4
(Homo sapiens (Human)) | BDBM50610828
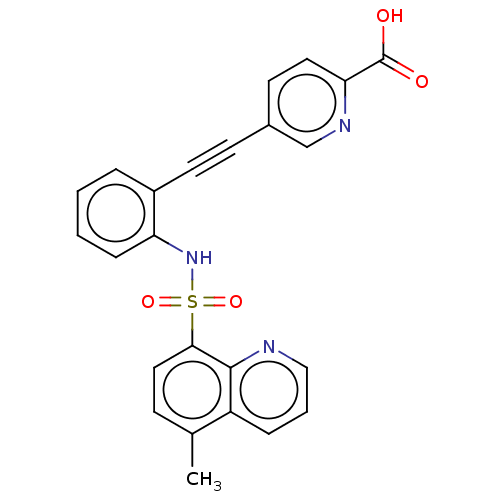
(CHEMBL5271816)Show SMILES Cc1ccc(c2ncccc12)S(=O)(=O)Nc1ccccc1C#Cc1ccc(nc1)C(O)=O | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 222 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Purine nucleoside phosphorylase
(Plasmodium falciparum) | BDBM50116358

(CHEMBL3604359)Show SMILES CSC[C@@H]1CN(Cc2c[nH]c3c(O)ncnc23)C[C@H]1O |r| Show InChI InChI=1S/C13H18N4O2S/c1-20-6-9-4-17(5-10(9)18)3-8-2-14-12-11(8)15-7-16-13(12)19/h2,7,9-10,14,18H,3-6H2,1H3,(H,15,16,19)/t9-,10+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 298 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Victoria University of Wellington
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Plasmodium falciparum PNP using inosine as substrate by xanthine oxidase coupling enzyme assay |
Bioorg Med Chem 23: 5326-33 (2015)
Article DOI: 10.1016/j.bmc.2015.07.059
BindingDB Entry DOI: 10.7270/Q2HX1FF5 |
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 4
(Mus musculus) | BDBM50610868
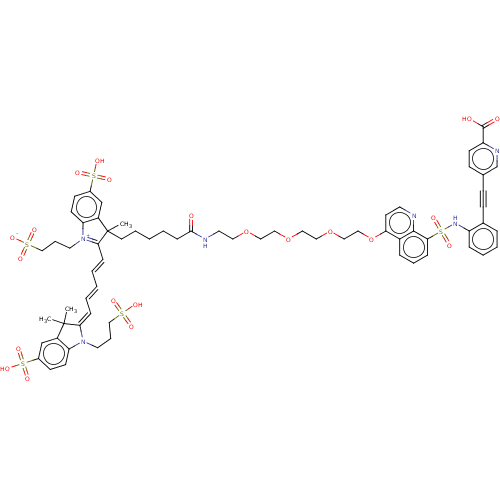
(CHEMBL5282978)Show SMILES CC1(C)\C(=C\C=C\C=C\C2=[N+](CCCS([O-])(=O)=O)c3ccc(cc3C2(C)CCCCCC(=O)NCCOCCOCCOCCOc2ccnc3c(cccc23)S(=O)(=O)Nc2ccccc2C#Cc2ccc(nc2)C(O)=O)S(O)(=O)=O)N(CCCS(O)(=O)=O)c2ccc(cc12)S(O)(=O)=O |c:9| | MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 301 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
S-methyl-5'-thioadenosine phosphorylase
(Homo sapiens (Human)) | BDBM50116357

(CHEMBL3604360)Show SMILES CSC[C@@H]1CN(Cc2c[nH]c3c(N)ncnc23)C[C@H]1O |r| Show InChI InChI=1S/C13H19N5OS/c1-20-6-9-4-18(5-10(9)19)3-8-2-15-12-11(8)16-7-17-13(12)14/h2,7,9-10,15,19H,3-6H2,1H3,(H2,14,16,17)/t9-,10+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 750 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Victoria University of Wellington
Curated by ChEMBL
| Assay Description
Inhibition of human MTAP using methylthioadenosine as substrate by xanthine oxidase coupling enzyme assay |
Bioorg Med Chem 23: 5326-33 (2015)
Article DOI: 10.1016/j.bmc.2015.07.059
BindingDB Entry DOI: 10.7270/Q2HX1FF5 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Mus musculus (Mouse)) | BDBM50566911

(CHEMBL4868446)Show SMILES CC(C)(O)COc1nc(OCCC(F)(F)F)c(CO)cc1-c1ccc(cc1)S(C)(=O)=O | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
In vivo inhibition of COX2 in po dosed C57BL/6J mouse model of spontaneous GI-tract tumor Apc-min assessed as chemopreventive effect by measuring red... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00890
BindingDB Entry DOI: 10.7270/Q24B352D |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Mus musculus (Mouse)) | BDBM50566912

(CHEMBL4859571)Show SMILES CC(C)(O)COc1nc(OCCCC(F)(F)F)c(CO)cc1-c1ccc(cc1)S(C)(=O)=O | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
In vivo inhibition of COX2 in po dosed C57BL/6J mouse model of spontaneous GI-tract tumor Apc-min assessed as chemopreventive effect by measuring red... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00890
BindingDB Entry DOI: 10.7270/Q24B352D |
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 4
(Homo sapiens (Human)) | BDBM21998
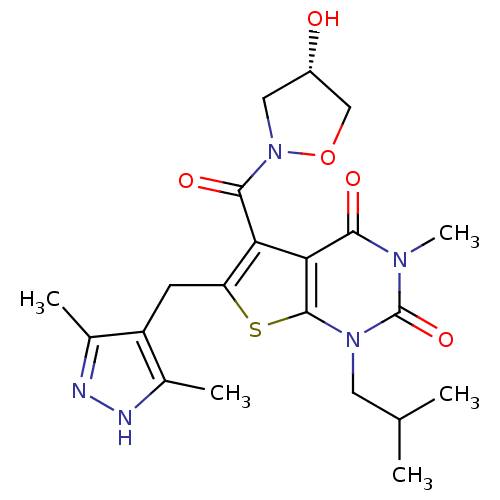
(6-[(3,5-dimethyl-1H-pyrazol-4-yl)methyl]-5-{[(4S)-...)Show SMILES CC(C)Cn1c2sc(Cc3c(C)n[nH]c3C)c(C(=O)N3C[C@H](O)CO3)c2c(=O)n(C)c1=O |r| Show InChI InChI=1S/C21H27N5O5S/c1-10(2)7-25-20-17(18(28)24(5)21(25)30)16(19(29)26-8-13(27)9-31-26)15(32-20)6-14-11(3)22-23-12(14)4/h10,13,27H,6-9H2,1-5H3,(H,22,23)/t13-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| | n/a | n/a | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Mus musculus (Mouse)) | BDBM11639

(4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyraz...)Show SMILES Cc1ccc(cc1)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C17H14F3N3O2S/c1-11-2-4-12(5-3-11)15-10-16(17(18,19)20)22-23(15)13-6-8-14(9-7-13)26(21,24)25/h2-10H,1H3,(H2,21,24,25) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
In vivo inhibition of COX2 in po dosed C57BL/6J mouse model of spontaneous GI-tract tumor Apc-min assessed as chemopreventive effect by measuring red... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00890
BindingDB Entry DOI: 10.7270/Q24B352D |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prostaglandin G/H synthase 2
(Mus musculus (Mouse)) | BDBM11639

(4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyraz...)Show SMILES Cc1ccc(cc1)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C17H14F3N3O2S/c1-11-2-4-12(5-3-11)15-10-16(17(18,19)20)22-23(15)13-6-8-14(9-7-13)26(21,24)25/h2-10H,1H3,(H2,21,24,25) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
In vivo inhibition of COX2 in po dosed C57BL/6J mouse model of spontaneous GI-tract tumor Apc-min assessed as chemopreventive effect by measuring red... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00890
BindingDB Entry DOI: 10.7270/Q24B352D |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Monocarboxylate transporter 4
(Homo sapiens (Human)) | BDBM21998

(6-[(3,5-dimethyl-1H-pyrazol-4-yl)methyl]-5-{[(4S)-...)Show SMILES CC(C)Cn1c2sc(Cc3c(C)n[nH]c3C)c(C(=O)N3C[C@H](O)CO3)c2c(=O)n(C)c1=O |r| Show InChI InChI=1S/C21H27N5O5S/c1-10(2)7-25-20-17(18(28)24(5)21(25)30)16(19(29)26-8-13(27)9-31-26)15(32-20)6-14-11(3)22-23-12(14)4/h10,13,27H,6-9H2,1-5H3,(H,22,23)/t13-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| | n/a | n/a | 6.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 4
(Homo sapiens (Human)) | BDBM50103571
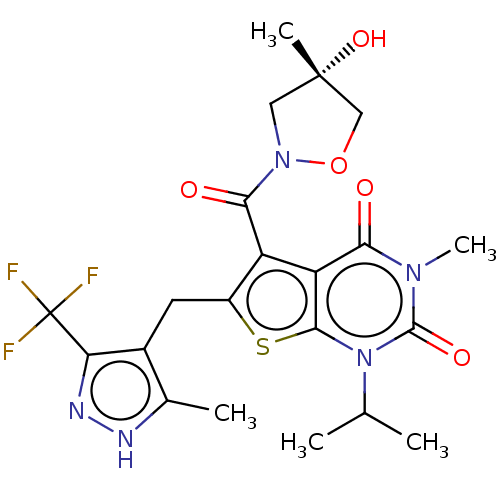
(CHEMBL3335793)Show SMILES CC(C)n1c2sc(Cc3c(C)[nH]nc3C(F)(F)F)c(C(=O)N3C[C@](C)(O)CO3)c2c(=O)n(C)c1=O |r| | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| | n/a | n/a | 7.10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 4
(Homo sapiens (Human)) | BDBM50610832
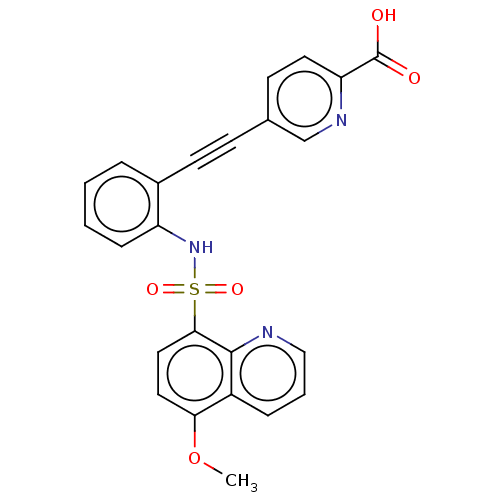
(CHEMBL5282741)Show SMILES COc1ccc(c2ncccc12)S(=O)(=O)Nc1ccccc1C#Cc1ccc(nc1)C(O)=O | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 4
(Homo sapiens (Human)) | BDBM50610830

(CHEMBL5267752)Show SMILES Cc1ccc2cccnc2c1S(=O)(=O)Nc1ccccc1C#Cc1ccc(nc1)C(O)=O | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 4
(Homo sapiens (Human)) | BDBM50610852
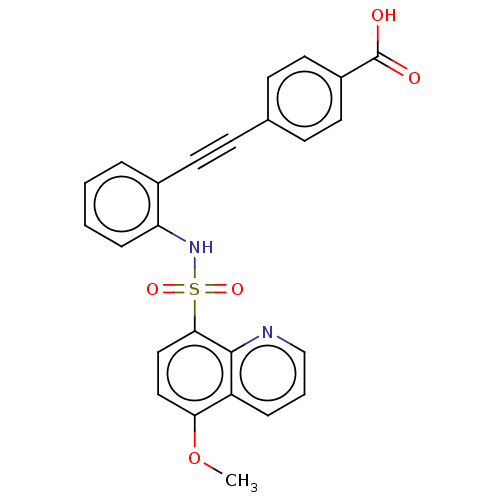
(CHEMBL5287835)Show SMILES COc1ccc(c2ncccc12)S(=O)(=O)Nc1ccccc1C#Cc1ccc(cc1)C(O)=O | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 4
(Homo sapiens (Human)) | BDBM50103571

(CHEMBL3335793)Show SMILES CC(C)n1c2sc(Cc3c(C)[nH]nc3C(F)(F)F)c(C(=O)N3C[C@](C)(O)CO3)c2c(=O)n(C)c1=O |r| | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| | n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM11639

(4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyraz...)Show SMILES Cc1ccc(cc1)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C17H14F3N3O2S/c1-11-2-4-12(5-3-11)15-10-16(17(18,19)20)22-23(15)13-6-8-14(9-7-13)26(21,24)25/h2-10H,1H3,(H2,21,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human recombinant COX-2 assessed as reduction in peroxidase activity using arachidonic acid as substrate preincubated for 60 mins follo... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00890
BindingDB Entry DOI: 10.7270/Q24B352D |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prostaglandin G/H synthase 2
(Mus musculus (Mouse)) | BDBM50566900

(CHEMBL4862802)Show SMILES CCCOc1nc(OCC(C)(C)O)c(cc1CO)-c1ccc(cc1)S(C)(=O)=O | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 35 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
In vivo inhibition of COX2 in po dosed C57BL/6J mouse model of spontaneous GI-tract tumor Apc-min assessed as chemopreventive effect by measuring red... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00890
BindingDB Entry DOI: 10.7270/Q24B352D |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50566907
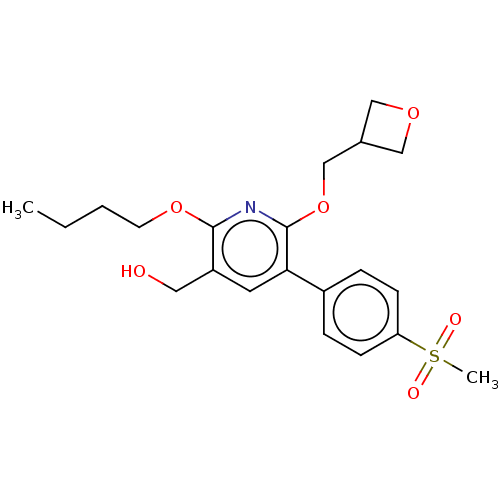
(CHEMBL4851236)Show SMILES CCCCOc1nc(OCC2COC2)c(cc1CO)-c1ccc(cc1)S(C)(=O)=O | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 36 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of COX2 (unknown origin) expressed in HEK293 TRex cells assessed as reduction in PGE2 production using arachidonic acid as substrate prein... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00890
BindingDB Entry DOI: 10.7270/Q24B352D |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data