 Found 49 hits with Last Name = 'eta' and Initial = 'je'
Found 49 hits with Last Name = 'eta' and Initial = 'je' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
DNA topoisomerase 1
(Homo sapiens (Human)) | BDBM50250903

((5Z,9Z)-5,9-heptacosadienoic acid | CHEMBL463437)Show InChI InChI=1S/C27H50O2/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18-19-20-21-22-23-24-25-26-27(28)29/h18-19,22-23H,2-17,20-21,24-26H2,1H3,(H,28,29)/b19-18-,23-22- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 900 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Puerto Rico
Curated by ChEMBL
| Assay Description
Inhibition of human DNA topoisomerase 1 |
J Nat Prod 65: 1715-8 (2002)
BindingDB Entry DOI: 10.7270/Q2WW7HFB |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50306714
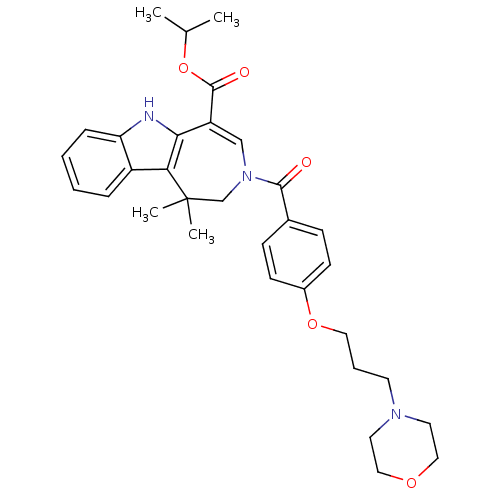
(CHEMBL601873 | Isopropyl 1,1-Dimethyl-3-[4-(3-morp...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1ccc(OCCCN2CCOCC2)cc1 |t:6| Show InChI InChI=1S/C32H39N3O5/c1-22(2)40-31(37)26-20-35(21-32(3,4)28-25-8-5-6-9-27(25)33-29(26)28)30(36)23-10-12-24(13-11-23)39-17-7-14-34-15-18-38-19-16-34/h5-6,8-13,20,22,33H,7,14-19,21H2,1-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.74E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50306744
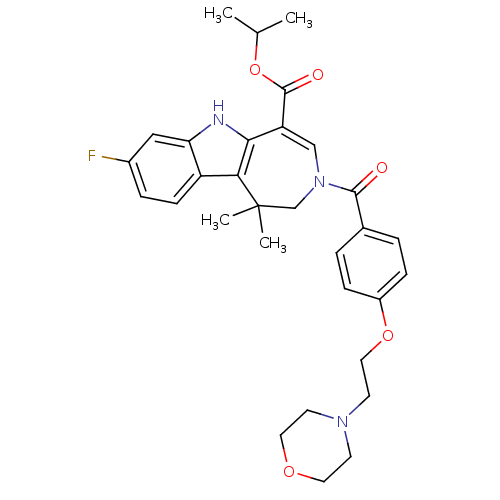
(CHEMBL601221 | Isopropyl 8-Fluoro-1,1-dimethyl-3-[...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1cc(F)ccc21)C(=O)c1ccc(OCCN2CCOCC2)cc1 |t:6| Show InChI InChI=1S/C31H36FN3O5/c1-20(2)40-30(37)25-18-35(19-31(3,4)27-24-10-7-22(32)17-26(24)33-28(25)27)29(36)21-5-8-23(9-6-21)39-16-13-34-11-14-38-15-12-34/h5-10,17-18,20,33H,11-16,19H2,1-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50306735

(1-methylethyl 8-fluoro-1,1-dimethyl-3-{[4-(3-morph...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1cc(F)ccc21)C(=O)c1ccc(OCCCN2CCOCC2)cc1 |t:6| Show InChI InChI=1S/C32H38FN3O5/c1-21(2)41-31(38)26-19-36(20-32(3,4)28-25-11-8-23(33)18-27(25)34-29(26)28)30(37)22-6-9-24(10-7-22)40-15-5-12-35-13-16-39-17-14-35/h6-11,18-19,21,34H,5,12-17,20H2,1-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
DNA topoisomerase 1
(Homo sapiens (Human)) | BDBM50150484
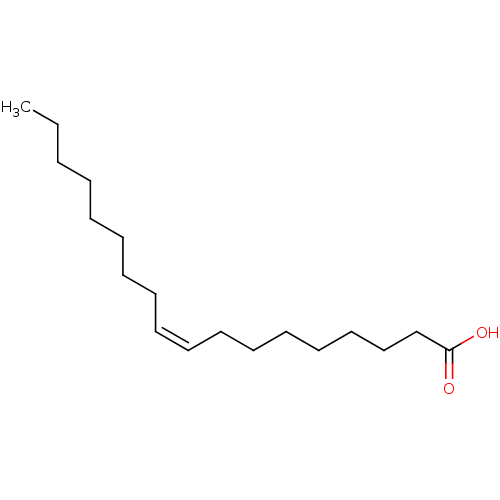
((Z)-9-octadecenoic acid | (Z)-Octadec-9-enoic acid...)Show InChI InChI=1S/C18H34O2/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18(19)20/h9-10H,2-8,11-17H2,1H3,(H,19,20)/b10-9- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 3.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Puerto Rico
Curated by ChEMBL
| Assay Description
Inhibition of DNA topoisomerase 1 (unknown origin) |
J Nat Prod 65: 1715-8 (2002)
BindingDB Entry DOI: 10.7270/Q2WW7HFB |
More data for this
Ligand-Target Pair | |
DNA topoisomerase 1
(Homo sapiens (Human)) | BDBM50250904

(CHEMBL460657 | Elaidinsaeure | elaidic acid | tran...)Show InChI InChI=1S/C18H34O2/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18(19)20/h9-10H,2-8,11-17H2,1H3,(H,19,20)/b10-9+ | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| n/a | n/a | >1.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Puerto Rico
Curated by ChEMBL
| Assay Description
Inhibition of DNA topoisomerase 1 (unknown origin) |
J Nat Prod 65: 1715-8 (2002)
BindingDB Entry DOI: 10.7270/Q2WW7HFB |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306720
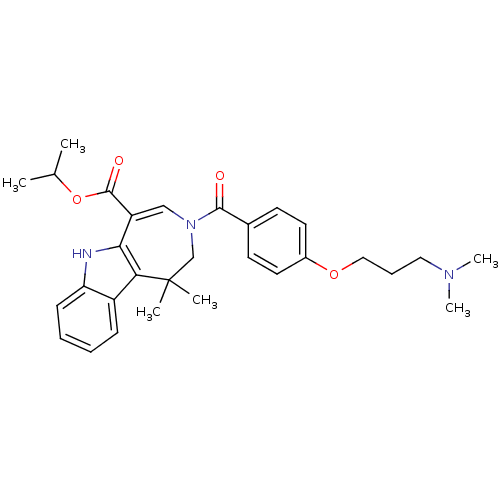
(CHEMBL603099 | Isopropyl 3-{4-[3-(Dimethylamino)pr...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1ccc(OCCCN(C)C)cc1 |t:6| Show InChI InChI=1S/C30H37N3O4/c1-20(2)37-29(35)24-18-33(19-30(3,4)26-23-10-7-8-11-25(23)31-27(24)26)28(34)21-12-14-22(15-13-21)36-17-9-16-32(5)6/h7-8,10-15,18,20,31H,9,16-17,19H2,1-6H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 271 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306721
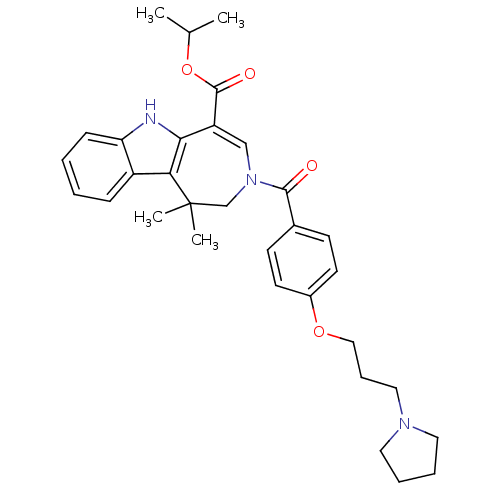
(CHEMBL602494 | Isopropyl 1,1-Dimethyl-3-[4-(3-pyrr...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1ccc(OCCCN2CCCC2)cc1 |t:6| Show InChI InChI=1S/C32H39N3O4/c1-22(2)39-31(37)26-20-35(21-32(3,4)28-25-10-5-6-11-27(25)33-29(26)28)30(36)23-12-14-24(15-13-23)38-19-9-18-34-16-7-8-17-34/h5-6,10-15,20,22,33H,7-9,16-19,21H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 362 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306722
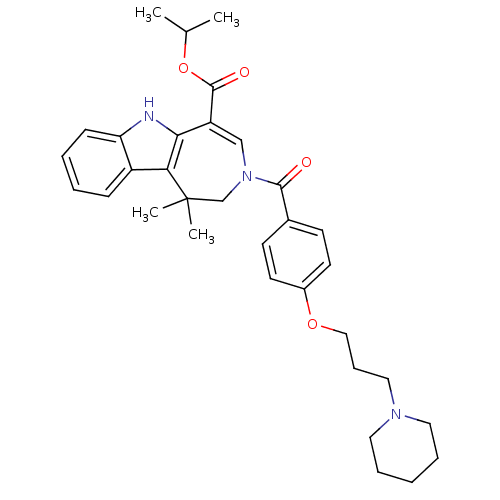
(CHEMBL602495 | Isopropyl 1,1-Dimethyl-3-[4-(3-pipe...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1ccc(OCCCN2CCCCC2)cc1 |t:6| Show InChI InChI=1S/C33H41N3O4/c1-23(2)40-32(38)27-21-36(22-33(3,4)29-26-11-6-7-12-28(26)34-30(27)29)31(37)24-13-15-25(16-14-24)39-20-10-19-35-17-8-5-9-18-35/h6-7,11-16,21,23,34H,5,8-10,17-20,22H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 424 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306723
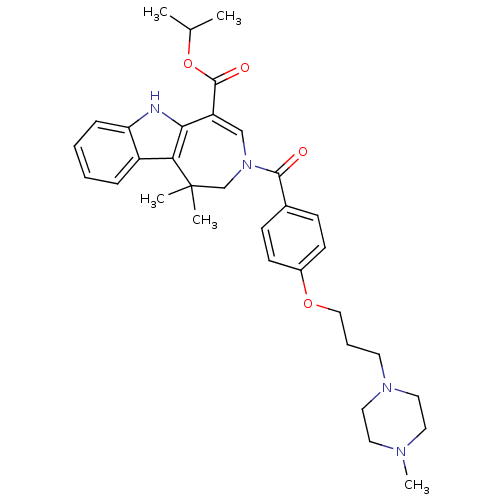
(CHEMBL592647 | Isopropyl 1,1-Dimethyl-3-{4-[3-(4-m...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1ccc(OCCCN2CCN(C)CC2)cc1 |t:6| Show InChI InChI=1S/C33H42N4O4/c1-23(2)41-32(39)27-21-37(22-33(3,4)29-26-9-6-7-10-28(26)34-30(27)29)31(38)24-11-13-25(14-12-24)40-20-8-15-36-18-16-35(5)17-19-36/h6-7,9-14,21,23,34H,8,15-20,22H2,1-5H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 194 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306724
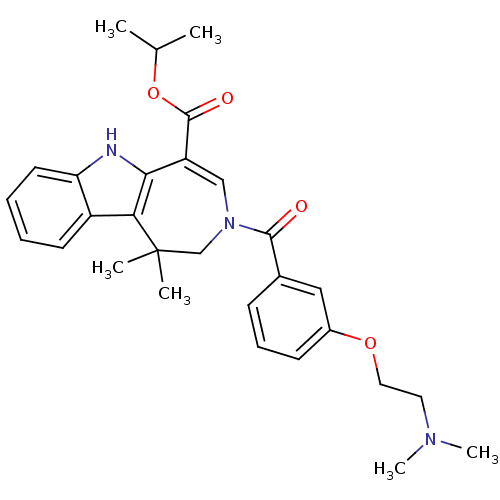
(CHEMBL602693 | Isopropyl 3-{3-[2-(Dimethylamino)et...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1cccc(OCCN(C)C)c1 |t:6| Show InChI InChI=1S/C29H35N3O4/c1-19(2)36-28(34)23-17-32(27(33)20-10-9-11-21(16-20)35-15-14-31(5)6)18-29(3,4)25-22-12-7-8-13-24(22)30-26(23)25/h7-13,16-17,19,30H,14-15,18H2,1-6H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.39E+3 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306725

(CHEMBL602287 | Isopropyl 1,1-Dimethyl-3-[3-(2-pyrr...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1cccc(OCCN2CCCC2)c1 |t:6| Show InChI InChI=1S/C31H37N3O4/c1-21(2)38-30(36)25-19-34(20-31(3,4)27-24-12-5-6-13-26(24)32-28(25)27)29(35)22-10-9-11-23(18-22)37-17-16-33-14-7-8-15-33/h5-6,9-13,18-19,21,32H,7-8,14-17,20H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.32E+3 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306726
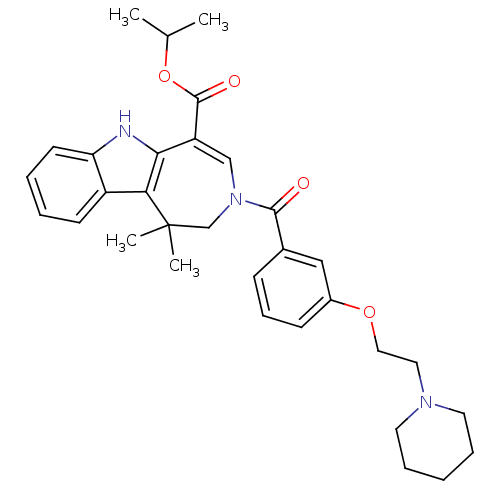
(CHEMBL602288 | Isopropyl 1,1-Dimethyl-3-[3-(2-pipe...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1cccc(OCCN2CCCCC2)c1 |t:6| Show InChI InChI=1S/C32H39N3O4/c1-22(2)39-31(37)26-20-35(21-32(3,4)28-25-13-6-7-14-27(25)33-29(26)28)30(36)23-11-10-12-24(19-23)38-18-17-34-15-8-5-9-16-34/h6-7,10-14,19-20,22,33H,5,8-9,15-18,21H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 894 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306727

(CHEMBL605996 | Isopropyl 1,1-Dimethyl-3-[3-(2-morp...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1cccc(OCCN2CCOCC2)c1 |t:6| Show InChI InChI=1S/C31H37N3O5/c1-21(2)39-30(36)25-19-34(20-31(3,4)27-24-10-5-6-11-26(24)32-28(25)27)29(35)22-8-7-9-23(18-22)38-17-14-33-12-15-37-16-13-33/h5-11,18-19,21,32H,12-17,20H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 855 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306728

(CHEMBL601251 | Isopropyl 1,1-Dimethyl-3-{3-[2-(4-m...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1cccc(OCCN2CCN(C)CC2)c1 |t:6| Show InChI InChI=1S/C32H40N4O4/c1-22(2)40-31(38)26-20-36(21-32(3,4)28-25-11-6-7-12-27(25)33-29(26)28)30(37)23-9-8-10-24(19-23)39-18-17-35-15-13-34(5)14-16-35/h6-12,19-20,22,33H,13-18,21H2,1-5H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.87E+3 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306729
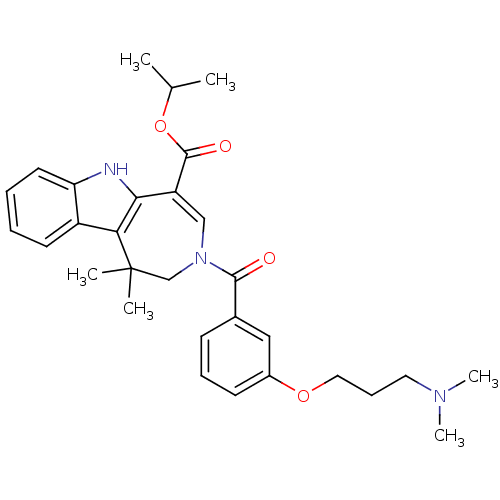
(CHEMBL601459 | Isopropyl 3-{3-[3-(Dimethylamino)pr...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1cccc(OCCCN(C)C)c1 |t:6| Show InChI InChI=1S/C30H37N3O4/c1-20(2)37-29(35)24-18-33(19-30(3,4)26-23-13-7-8-14-25(23)31-27(24)26)28(34)21-11-9-12-22(17-21)36-16-10-15-32(5)6/h7-9,11-14,17-18,20,31H,10,15-16,19H2,1-6H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 617 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306730
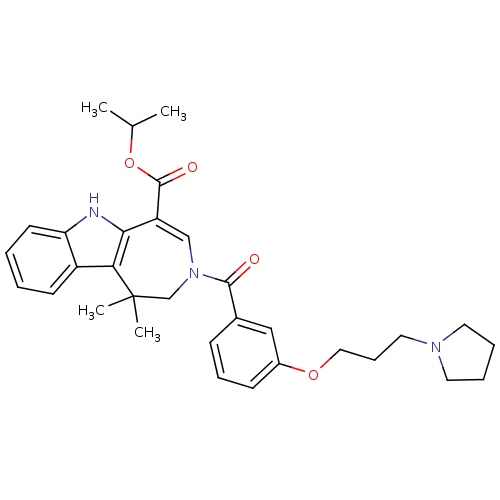
(CHEMBL602059 | Isopropyl 1,1-Dimethyl-3-[3-(3-pyrr...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1cccc(OCCCN2CCCC2)c1 |t:6| Show InChI InChI=1S/C32H39N3O4/c1-22(2)39-31(37)26-20-35(21-32(3,4)28-25-13-5-6-14-27(25)33-29(26)28)30(36)23-11-9-12-24(19-23)38-18-10-17-34-15-7-8-16-34/h5-6,9,11-14,19-20,22,33H,7-8,10,15-18,21H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306731
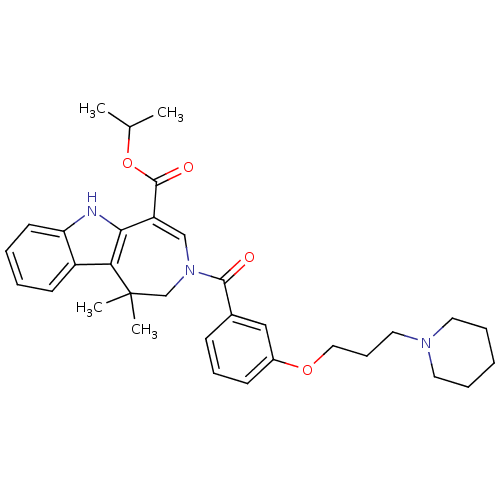
(CHEMBL604335 | Isopropyl 1,1-Dimethyl-3-[3-(3-pipe...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1cccc(OCCCN2CCCCC2)c1 |t:6| Show InChI InChI=1S/C33H41N3O4/c1-23(2)40-32(38)27-21-36(22-33(3,4)29-26-14-6-7-15-28(26)34-30(27)29)31(37)24-12-10-13-25(20-24)39-19-11-18-35-16-8-5-9-17-35/h6-7,10,12-15,20-21,23,34H,5,8-9,11,16-19,22H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.12E+3 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306732

(CHEMBL602061 | Isopropyl 1,1-Dimethyl-3-[3-(3-morp...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1cccc(OCCCN2CCOCC2)c1 |t:6| Show InChI InChI=1S/C32H39N3O5/c1-22(2)40-31(37)26-20-35(21-32(3,4)28-25-11-5-6-12-27(25)33-29(26)28)30(36)23-9-7-10-24(19-23)39-16-8-13-34-14-17-38-18-15-34/h5-7,9-12,19-20,22,33H,8,13-18,21H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 796 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306733
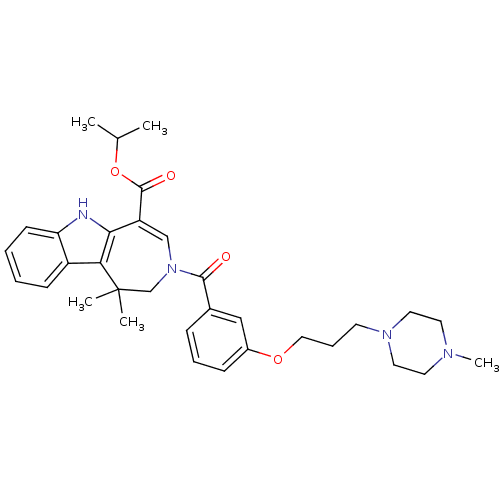
(CHEMBL602709 | Isopropyl 1,1-Dimethyl-3-{3-[3-(4-m...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1cccc(OCCCN2CCN(C)CC2)c1 |t:6| Show InChI InChI=1S/C33H42N4O4/c1-23(2)41-32(39)27-21-37(22-33(3,4)29-26-12-6-7-13-28(26)34-30(27)29)31(38)24-10-8-11-25(20-24)40-19-9-14-36-17-15-35(5)16-18-36/h6-8,10-13,20-21,23,34H,9,14-19,22H2,1-5H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.02E+3 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Mus musculus) | BDBM28542
(WAY-362450 | XL335 | propan-2-yl 3-[(3,4-difluorop...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1ccc(F)c(F)c1 |t:6| Show InChI InChI=1S/C25H24F2N2O3/c1-14(2)32-24(31)17-12-29(23(30)15-9-10-18(26)19(27)11-15)13-25(3,4)21-16-7-5-6-8-20(16)28-22(17)21/h5-12,14,28H,13H2,1-4H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 152 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at mouse FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cytochrome P450 2B1
(Rattus norvegicus) | BDBM50330387

(1H-pyrrolidine-1-(4'-hydroxyphenyl)-2,5-dione | CH...)Show InChI InChI=1S/C10H9NO3/c12-8-3-1-7(2-4-8)11-9(13)5-6-10(11)14/h1-6,12-14H | PDB
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 2.40E+4 | n/a | n/a | n/a | n/a | n/a |
Instituto Politécnico Nacional
Curated by ChEMBL
| Assay Description
Binding affinity to rat CYP2B1 |
Eur J Med Chem 45: 4845-55 (2010)
Article DOI: 10.1016/j.ejmech.2010.07.055
BindingDB Entry DOI: 10.7270/Q2862GPP |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306735

(1-methylethyl 8-fluoro-1,1-dimethyl-3-{[4-(3-morph...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1cc(F)ccc21)C(=O)c1ccc(OCCCN2CCOCC2)cc1 |t:6| Show InChI InChI=1S/C32H38FN3O5/c1-21(2)41-31(38)26-19-36(20-32(3,4)28-25-11-8-23(33)18-27(25)34-29(26)28)30(37)22-6-9-24(10-7-22)40-15-5-12-35-13-16-39-17-14-35/h6-11,18-19,21,34H,5,12-17,20H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 88 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Bile acid receptor
(Homo sapiens (Human)) | BDBM28542

(WAY-362450 | XL335 | propan-2-yl 3-[(3,4-difluorop...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1ccc(F)c(F)c1 |t:6| Show InChI InChI=1S/C25H24F2N2O3/c1-14(2)32-24(31)17-12-29(23(30)15-9-10-18(26)19(27)11-15)13-25(3,4)21-16-7-5-6-8-20(16)28-22(17)21/h5-12,14,28H,13H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 15 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
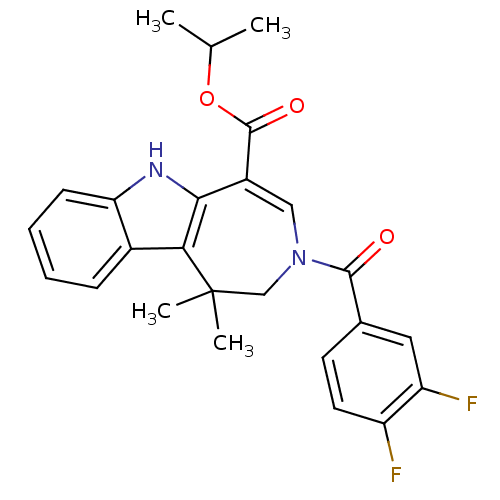
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306734

(CHEMBL603278 | isopropyl 3-(3,4-difluorobenzoyl)-9...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccc(F)cc21)C(=O)c1ccc(F)c(F)c1 |t:6| Show InChI InChI=1S/C25H23F3N2O3/c1-13(2)33-24(32)17-11-30(23(31)14-5-7-18(27)19(28)9-14)12-25(3,4)21-16-10-15(26)6-8-20(16)29-22(17)21/h5-11,13,29H,12H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 45 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306736

(CHEMBL604334 | Isopropyl 1,1-Dimethyl-3-[3-(morpho...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1cccc(CN2CCOCC2)c1 |t:6| Show InChI InChI=1S/C30H35N3O4/c1-20(2)37-29(35)24-18-33(19-30(3,4)26-23-10-5-6-11-25(23)31-27(24)26)28(34)22-9-7-8-21(16-22)17-32-12-14-36-15-13-32/h5-11,16,18,20,31H,12-15,17,19H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.58E+3 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306737

(CHEMBL601434 | Isopropyl 1,1-Dimethyl-3-{3-[(4-met...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1cccc(CN2CCN(C)CC2)c1 |t:6| Show InChI InChI=1S/C31H38N4O3/c1-21(2)38-30(37)25-19-35(20-31(3,4)27-24-11-6-7-12-26(24)32-28(25)27)29(36)23-10-8-9-22(17-23)18-34-15-13-33(5)14-16-34/h6-12,17,19,21,32H,13-16,18,20H2,1-5H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 368 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306738
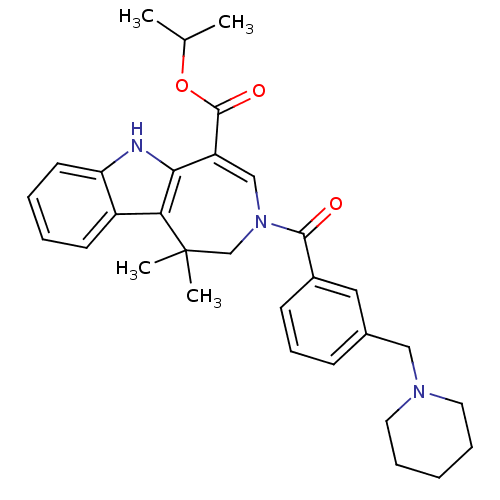
(CHEMBL601647 | Isopropyl 1,1-Dimethyl-3-[3-(piperi...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1cccc(CN2CCCCC2)c1 |t:6| Show InChI InChI=1S/C31H37N3O3/c1-21(2)37-30(36)25-19-34(20-31(3,4)27-24-13-6-7-14-26(24)32-28(25)27)29(35)23-12-10-11-22(17-23)18-33-15-8-5-9-16-33/h6-7,10-14,17,19,21,32H,5,8-9,15-16,18,20H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 534 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306739

(CHEMBL602883 | Isopropyl 1,1-Dimethyl-3-[3-(pyrrol...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1cccc(CN2CCCC2)c1 |t:6| Show InChI InChI=1S/C30H35N3O3/c1-20(2)36-29(35)24-18-33(19-30(3,4)26-23-12-5-6-13-25(23)31-27(24)26)28(34)22-11-9-10-21(16-22)17-32-14-7-8-15-32/h5-6,9-13,16,18,20,31H,7-8,14-15,17,19H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 340 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306740
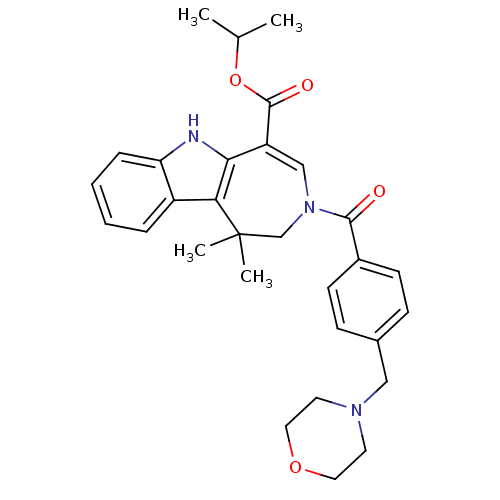
(CHEMBL603086 | Isopropyl 1,1-Dimethyl-3-[4-(morpho...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1ccc(CN2CCOCC2)cc1 |t:6| Show InChI InChI=1S/C30H35N3O4/c1-20(2)37-29(35)24-18-33(19-30(3,4)26-23-7-5-6-8-25(23)31-27(24)26)28(34)22-11-9-21(10-12-22)17-32-13-15-36-16-14-32/h5-12,18,20,31H,13-17,19H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 23 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306741

(CHEMBL603282 | Isopropyl 8-Fluoro-1,1-dimethyl-3-[...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1cc(F)ccc21)C(=O)c1ccc(CN2CCOCC2)cc1 |t:6| Show InChI InChI=1S/C30H34FN3O4/c1-19(2)38-29(36)24-17-34(18-30(3,4)26-23-10-9-22(31)15-25(23)32-27(24)26)28(35)21-7-5-20(6-8-21)16-33-11-13-37-14-12-33/h5-10,15,17,19,32H,11-14,16,18H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 447 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306742

(CHEMBL602681 | Isopropyl 1,1-Dimethyl-3-[4-(2-morp...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1ccc(CCN2CCOCC2)cc1 |t:6| Show InChI InChI=1S/C31H37N3O4/c1-21(2)38-30(36)25-19-34(20-31(3,4)27-24-7-5-6-8-26(24)32-28(25)27)29(35)23-11-9-22(10-12-23)13-14-33-15-17-37-18-16-33/h5-12,19,21,32H,13-18,20H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 602 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306743

(CHEMBL611221 | Isopropyl 8-Fluoro-1,1-dimethyl-3-[...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1cc(F)ccc21)C(=O)c1ccc(CCN2CCOCC2)cc1 |t:6| Show InChI InChI=1S/C31H36FN3O4/c1-20(2)39-30(37)25-18-35(19-31(3,4)27-24-10-9-23(32)17-26(24)33-28(25)27)29(36)22-7-5-21(6-8-22)11-12-34-13-15-38-16-14-34/h5-10,17-18,20,33H,11-16,19H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 342 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Mus musculus) | BDBM50306735

(1-methylethyl 8-fluoro-1,1-dimethyl-3-{[4-(3-morph...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1cc(F)ccc21)C(=O)c1ccc(OCCCN2CCOCC2)cc1 |t:6| Show InChI InChI=1S/C32H38FN3O5/c1-21(2)41-31(38)26-19-36(20-32(3,4)28-25-11-8-23(33)18-27(25)34-29(26)28)30(37)22-6-9-24(10-7-22)40-15-5-12-35-13-16-39-17-14-35/h6-11,18-19,21,34H,5,12-17,20H2,1-4H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 52 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at mouse FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306744

(CHEMBL601221 | Isopropyl 8-Fluoro-1,1-dimethyl-3-[...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1cc(F)ccc21)C(=O)c1ccc(OCCN2CCOCC2)cc1 |t:6| Show InChI InChI=1S/C31H36FN3O5/c1-20(2)40-30(37)25-18-35(19-31(3,4)27-24-10-7-22(32)17-26(24)33-28(25)27)29(36)21-5-8-23(9-6-21)39-16-13-34-11-14-38-15-12-34/h5-10,17-18,20,33H,11-16,19H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 99 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Mus musculus) | BDBM50306744

(CHEMBL601221 | Isopropyl 8-Fluoro-1,1-dimethyl-3-[...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1cc(F)ccc21)C(=O)c1ccc(OCCN2CCOCC2)cc1 |t:6| Show InChI InChI=1S/C31H36FN3O5/c1-20(2)40-30(37)25-18-35(19-31(3,4)27-24-10-7-22(32)17-26(24)33-28(25)27)29(36)21-5-8-23(9-6-21)39-16-13-34-11-14-38-15-12-34/h5-10,17-18,20,33H,11-16,19H2,1-4H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 188 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at mouse FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Mus musculus) | BDBM50306714

(CHEMBL601873 | Isopropyl 1,1-Dimethyl-3-[4-(3-morp...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1ccc(OCCCN2CCOCC2)cc1 |t:6| Show InChI InChI=1S/C32H39N3O5/c1-22(2)40-31(37)26-20-35(21-32(3,4)28-25-8-5-6-9-27(25)33-29(26)28)30(36)23-10-12-24(13-11-23)39-17-7-14-34-15-18-38-19-16-34/h5-6,8-13,20,22,33H,7,14-19,21H2,1-4H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 108 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at mouse FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Mus musculus) | BDBM50306718
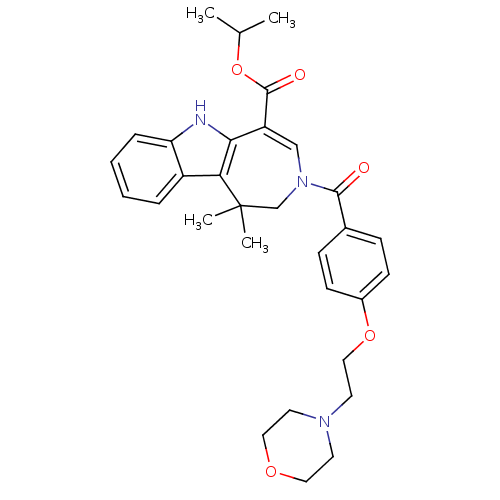
(CHEMBL602894 | Isopropyl 1,1-Dimethyl-3-[4-(2-morp...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1ccc(OCCN2CCOCC2)cc1 |t:6| Show InChI InChI=1S/C31H37N3O5/c1-21(2)39-30(36)25-19-34(20-31(3,4)27-24-7-5-6-8-26(24)32-28(25)27)29(35)22-9-11-23(12-10-22)38-18-15-33-13-16-37-17-14-33/h5-12,19,21,32H,13-18,20H2,1-4H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 190 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at mouse FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2B1
(Rattus norvegicus) | BDBM50330386
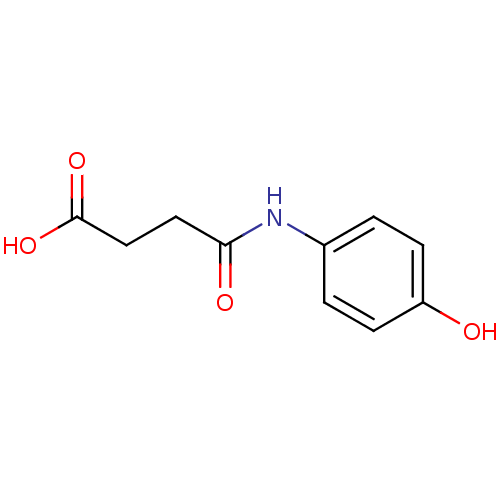
(4-(4'-hydroxy-phenylamino)-4-oxo propanoic acid | ...)Show InChI InChI=1S/C10H11NO4/c12-8-3-1-7(2-4-8)11-9(13)5-6-10(14)15/h1-4,12H,5-6H2,(H,11,13)(H,14,15) | PDB
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.12E+3 | n/a | n/a | n/a | n/a | n/a |
Instituto Politécnico Nacional
Curated by ChEMBL
| Assay Description
Binding affinity to rat CYP2B1 |
Eur J Med Chem 45: 4845-55 (2010)
Article DOI: 10.1016/j.ejmech.2010.07.055
BindingDB Entry DOI: 10.7270/Q2862GPP |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A1
(Homo sapiens (Human)) | BDBM50330387

(1H-pyrrolidine-1-(4'-hydroxyphenyl)-2,5-dione | CH...)Show InChI InChI=1S/C10H9NO3/c12-8-3-1-7(2-4-8)11-9(13)5-6-10(11)14/h1-6,12-14H | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.35E+4 | n/a | n/a | n/a | n/a | n/a |
Instituto Politécnico Nacional
Curated by ChEMBL
| Assay Description
Binding affinity to human CYP1A1 |
Eur J Med Chem 45: 4845-55 (2010)
Article DOI: 10.1016/j.ejmech.2010.07.055
BindingDB Entry DOI: 10.7270/Q2862GPP |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A1
(Homo sapiens (Human)) | BDBM50330386

(4-(4'-hydroxy-phenylamino)-4-oxo propanoic acid | ...)Show InChI InChI=1S/C10H11NO4/c12-8-3-1-7(2-4-8)11-9(13)5-6-10(14)15/h1-4,12H,5-6H2,(H,11,13)(H,14,15) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 2.25E+3 | n/a | n/a | n/a | n/a | n/a |
Instituto Politécnico Nacional
Curated by ChEMBL
| Assay Description
Binding affinity to human CYP1A1 |
Eur J Med Chem 45: 4845-55 (2010)
Article DOI: 10.1016/j.ejmech.2010.07.055
BindingDB Entry DOI: 10.7270/Q2862GPP |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A1
(Homo sapiens (Human)) | BDBM26197
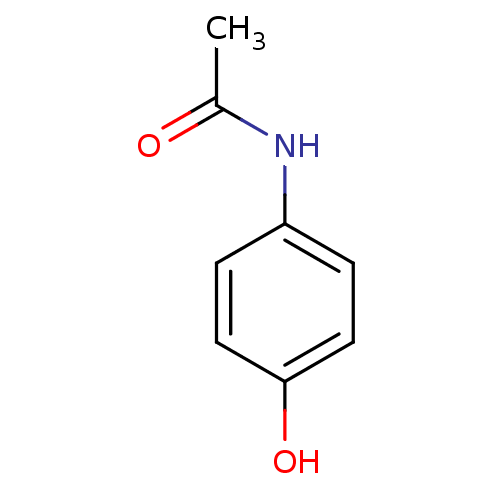
(CHEMBL112 | N-(4-hydroxyphenyl)acetamide | Norco |...)Show InChI InChI=1S/C8H9NO2/c1-6(10)9-7-2-4-8(11)5-3-7/h2-5,11H,1H3,(H,9,10) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
| DrugBank
Article
PubMed
| n/a | n/a | n/a | 1.91E+4 | n/a | n/a | n/a | n/a | n/a |
Instituto Politécnico Nacional
Curated by ChEMBL
| Assay Description
Binding affinity to human CYP1A1 |
Eur J Med Chem 45: 4845-55 (2010)
Article DOI: 10.1016/j.ejmech.2010.07.055
BindingDB Entry DOI: 10.7270/Q2862GPP |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306719
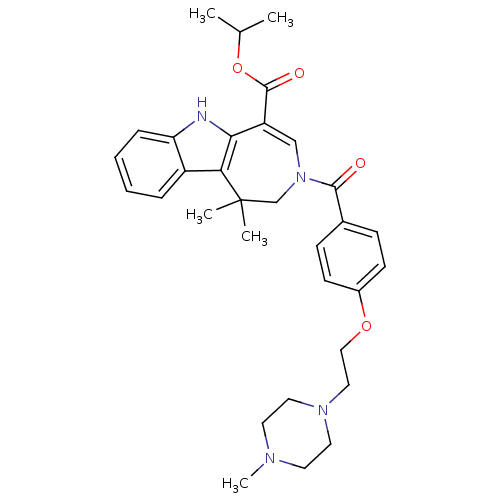
(CHEMBL602895 | Isopropyl 1,1-Dimethyl-3-{4-[2-(4-m...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1ccc(OCCN2CCN(C)CC2)cc1 |t:6| Show InChI InChI=1S/C32H40N4O4/c1-22(2)40-31(38)26-20-36(21-32(3,4)28-25-8-6-7-9-27(25)33-29(26)28)30(37)23-10-12-24(13-11-23)39-19-18-35-16-14-34(5)15-17-35/h6-13,20,22,33H,14-19,21H2,1-5H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 287 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306718

(CHEMBL602894 | Isopropyl 1,1-Dimethyl-3-[4-(2-morp...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1ccc(OCCN2CCOCC2)cc1 |t:6| Show InChI InChI=1S/C31H37N3O5/c1-21(2)39-30(36)25-19-34(20-31(3,4)27-24-7-5-6-8-26(24)32-28(25)27)29(35)22-9-11-23(12-10-22)38-18-15-33-13-16-37-17-14-33/h5-12,19,21,32H,13-18,20H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 249 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306717

(CHEMBL600415 | Isopropyl 1,1-Dimethyl-3-[4-(2-pyrr...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1ccc(OCCN2CCCC2)cc1 |t:6| Show InChI InChI=1S/C31H37N3O4/c1-21(2)38-30(36)25-19-34(20-31(3,4)27-24-9-5-6-10-26(24)32-28(25)27)29(35)22-11-13-23(14-12-22)37-18-17-33-15-7-8-16-33/h5-6,9-14,19,21,32H,7-8,15-18,20H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 698 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306716
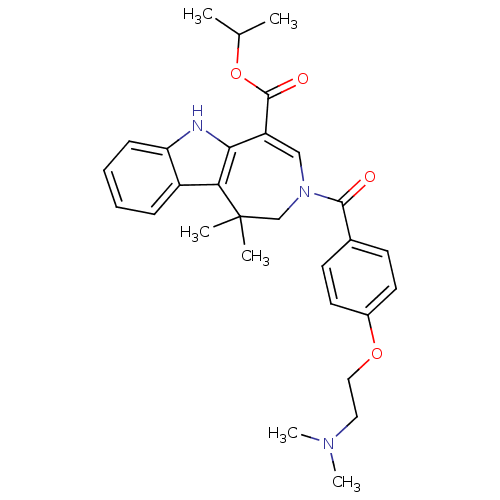
(CHEMBL604761 | Isopropyl 3-{4-[2-(Dimethylamino)et...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1ccc(OCCN(C)C)cc1 |t:6| Show InChI InChI=1S/C29H35N3O4/c1-19(2)36-28(34)23-17-32(27(33)20-11-13-21(14-12-20)35-16-15-31(5)6)18-29(3,4)25-22-9-7-8-10-24(22)30-26(23)25/h7-14,17,19,30H,15-16,18H2,1-6H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 439 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306715
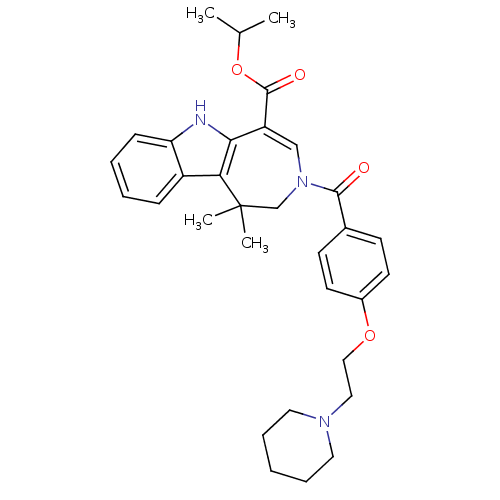
(CHEMBL603492 | Isopropyl 1,1-Dimethyl-3-[4-(2-pipe...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1ccc(OCCN2CCCCC2)cc1 |t:6| Show InChI InChI=1S/C32H39N3O4/c1-22(2)39-31(37)26-20-35(21-32(3,4)28-25-10-6-7-11-27(25)33-29(26)28)30(36)23-12-14-24(15-13-23)38-19-18-34-16-8-5-9-17-34/h6-7,10-15,20,22,33H,5,8-9,16-19,21H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.07E+3 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Homo sapiens (Human)) | BDBM50306714

(CHEMBL601873 | Isopropyl 1,1-Dimethyl-3-[4-(3-morp...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1ccc(OCCCN2CCOCC2)cc1 |t:6| Show InChI InChI=1S/C32H39N3O5/c1-22(2)40-31(37)26-20-35(21-32(3,4)28-25-8-5-6-9-27(25)33-29(26)28)30(36)23-10-12-24(13-11-23)39-17-7-14-34-15-18-38-19-16-34/h5-6,8-13,20,22,33H,7,14-19,21H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 101 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at human FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |
Bile acid receptor
(Mus musculus) | BDBM50306734

(CHEMBL603278 | isopropyl 3-(3,4-difluorobenzoyl)-9...)Show SMILES CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccc(F)cc21)C(=O)c1ccc(F)c(F)c1 |t:6| Show InChI InChI=1S/C25H23F3N2O3/c1-13(2)33-24(32)17-11-30(23(31)14-5-7-18(27)19(28)9-14)12-25(3,4)21-16-10-15(26)6-8-20(16)29-22(17)21/h5-11,13,29H,12H2,1-4H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 112 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonist activity at mouse FXR-LBD expressed in HEL293 cells by Gal4-luciferase assay |
J Med Chem 53: 1774-87 (2010)
Article DOI: 10.1021/jm901650u
BindingDB Entry DOI: 10.7270/Q24F1QT5 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data