| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Bile acid receptor |
|---|
| Ligand | BDBM50306723 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_611887 (CHEMBL1070842) |
|---|
| EC50 | 194±n/a nM |
|---|
| Citation |  Lundquist, JT; Harnish, DC; Kim, CY; Mehlmann, JF; Unwalla, RJ; Phipps, KM; Crawley, ML; Commons, T; Green, DM; Xu, W; Hum, WT; Eta, JE; Feingold, I; Patel, V; Evans, MJ; Lai, K; Borges-Marcucci, L; Mahaney, PE; Wrobel, JE Improvement of physiochemical properties of the tetrahydroazepinoindole series of farnesoid X receptor (FXR) agonists: beneficial modulation of lipids in primates. J Med Chem53:1774-87 (2010) [PubMed] Article Lundquist, JT; Harnish, DC; Kim, CY; Mehlmann, JF; Unwalla, RJ; Phipps, KM; Crawley, ML; Commons, T; Green, DM; Xu, W; Hum, WT; Eta, JE; Feingold, I; Patel, V; Evans, MJ; Lai, K; Borges-Marcucci, L; Mahaney, PE; Wrobel, JE Improvement of physiochemical properties of the tetrahydroazepinoindole series of farnesoid X receptor (FXR) agonists: beneficial modulation of lipids in primates. J Med Chem53:1774-87 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Bile acid receptor |
|---|
| Name: | Bile acid receptor |
|---|
| Synonyms: | BAR | Bile acid receptor FXR | FXR | Farnesol receptor HRR-1 | HRR1 | NR1H4 | NR1H4_HUMAN | Nuclear receptor subfamily 1 group H member 4 | RIP14 | RXR-interacting protein 14 | Retinoid X receptor-interacting protein 14 | farnesoid x receptor |
|---|
| Type: | Nuclear Receptor |
|---|
| Mol. Mass.: | 55916.24 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q96RI1 |
|---|
| Residue: | 486 |
|---|
| Sequence: | MVMQFQGLENPIQISPHCSCTPSGFFMEMMSMKPAKGVLTEQVAGPLGQNLEVEPYSQYS
NVQFPQVQPQISSSSYYSNLGFYPQQPEEWYSPGIYELRRMPAETLYQGETEVAEMPVTK
KPRMGASAGRIKGDELCVVCGDRASGYHYNALTCEGCKGFFRRSITKNAVYKCKNGGNCV
MDMYMRRKCQECRLRKCKEMGMLAECMYTGLLTEIQCKSKRLRKNVKQHADQTVNEDSEG
RDLRQVTSTTKSCREKTELTPDQQTLLHFIMDSYNKQRMPQEITNKILKEEFSAEENFLI
LTEMATNHVQVLVEFTKKLPGFQTLDHEDQIALLKGSAVEAMFLRSAEIFNKKLPSGHSD
LLEERIRNSGISDEYITPMFSFYKSIGELKMTQEEYALLTAIVILSPDRQYIKDREAVEK
LQEPLLDVLQKLCKIHQPENPQHFACLLGRLTELRTFNHHHAEMLMSWRVNDHKFTPLLC
EIWDVQ
|
|
|
|---|
| BDBM50306723 |
|---|
| n/a |
|---|
| Name | BDBM50306723 |
|---|
| Synonyms: | CHEMBL592647 | Isopropyl 1,1-Dimethyl-3-{4-[3-(4-methylpiperazin-1-yl)propoxy]benzoyl}-1,2,3,6-tetrahydroazepino[4,5-b]indole-5-carboxylate |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C33H42N4O4 |
|---|
| Mol. Mass. | 558.711 |
|---|
| SMILES | CC(C)OC(=O)C1=CN(CC(C)(C)c2c1[nH]c1ccccc21)C(=O)c1ccc(OCCCN2CCN(C)CC2)cc1 |t:6| |
|---|
| Structure | 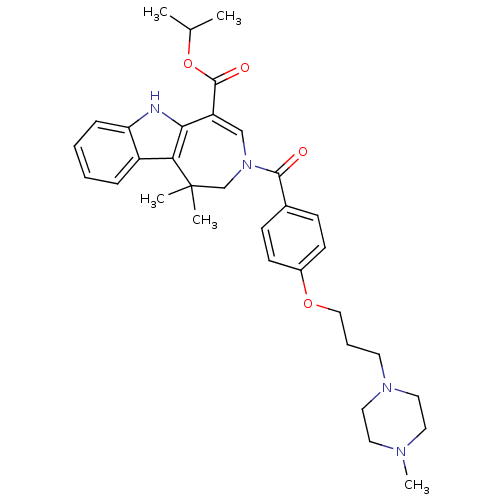
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Lundquist, JT; Harnish, DC; Kim, CY; Mehlmann, JF; Unwalla, RJ; Phipps, KM; Crawley, ML; Commons, T; Green, DM; Xu, W; Hum, WT; Eta, JE; Feingold, I; Patel, V; Evans, MJ; Lai, K; Borges-Marcucci, L; Mahaney, PE; Wrobel, JE Improvement of physiochemical properties of the tetrahydroazepinoindole series of farnesoid X receptor (FXR) agonists: beneficial modulation of lipids in primates. J Med Chem53:1774-87 (2010) [PubMed] Article
Lundquist, JT; Harnish, DC; Kim, CY; Mehlmann, JF; Unwalla, RJ; Phipps, KM; Crawley, ML; Commons, T; Green, DM; Xu, W; Hum, WT; Eta, JE; Feingold, I; Patel, V; Evans, MJ; Lai, K; Borges-Marcucci, L; Mahaney, PE; Wrobel, JE Improvement of physiochemical properties of the tetrahydroazepinoindole series of farnesoid X receptor (FXR) agonists: beneficial modulation of lipids in primates. J Med Chem53:1774-87 (2010) [PubMed] Article