 Found 386 hits with Last Name = 'frormann' and Initial = 's'
Found 386 hits with Last Name = 'frormann' and Initial = 's' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50398494
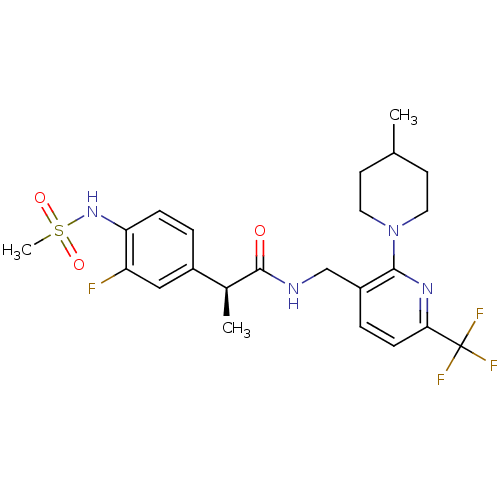
(CHEMBL2177429)Show SMILES C[C@H](C(=O)NCc1ccc(nc1N1CCC(C)CC1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 |r| Show InChI InChI=1S/C23H28F4N4O3S/c1-14-8-10-31(11-9-14)21-17(5-7-20(29-21)23(25,26)27)13-28-22(32)15(2)16-4-6-19(18(24)12-16)30-35(3,33)34/h4-7,12,14-15,30H,8-11,13H2,1-3H3,(H,28,32)/t15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHOK1 cells assessed as inhibition of N-acetyldopamine-induced activity after 5 mins by FLIPR assay |
Bioorg Med Chem 21: 6657-64 (2013)
Article DOI: 10.1016/j.bmc.2013.08.015
BindingDB Entry DOI: 10.7270/Q26Q1ZPN |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50049553
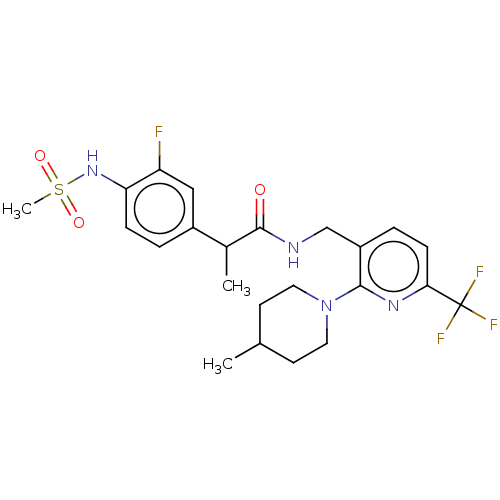
(CHEMBL2177428)Show SMILES CC(C(=O)NCc1ccc(nc1N1CCC(C)CC1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 Show InChI InChI=1S/C23H28F4N4O3S/c1-14-8-10-31(11-9-14)21-17(5-7-20(29-21)23(25,26)27)13-28-22(32)15(2)16-4-6-19(18(24)12-16)30-35(3,33)34/h4-7,12,14-15,30H,8-11,13H2,1-3H3,(H,28,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of N-arachidonoyl dopamine-induced activity by FLIPR assay |
Eur J Med Chem 93: 101-8 (2015)
Article DOI: 10.1016/j.ejmech.2015.02.001
BindingDB Entry DOI: 10.7270/Q2N0188S |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50061298

(CHEMBL3393837)Show SMILES CC1CCN(CC1)c1nc(ccc1CNC(=O)Nc1cccc2cnccc12)C(F)(F)F Show InChI InChI=1S/C23H24F3N5O/c1-15-8-11-31(12-9-15)21-17(5-6-20(30-21)23(24,25)26)14-28-22(32)29-19-4-2-3-16-13-27-10-7-18(16)19/h2-7,10,13,15H,8-9,11-12,14H2,1H3,(H2,28,29,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 assessed as inhibition of NADA-induced effect at 1 uM by FLIPR assay |
Bioorg Med Chem Lett 25: 803-6 (2015)
Article DOI: 10.1016/j.bmcl.2014.12.086
BindingDB Entry DOI: 10.7270/Q2JD4ZG3 |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50073160
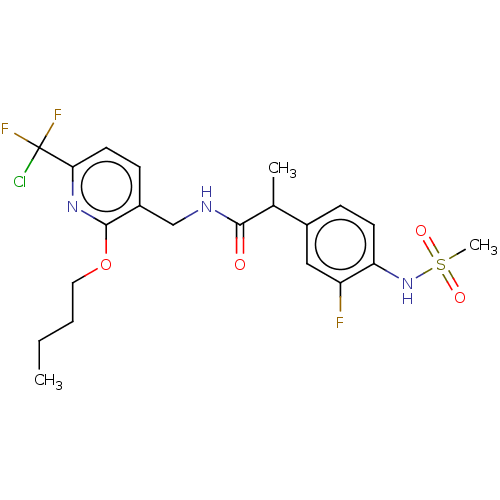
(CHEMBL3407762)Show SMILES CCCCOc1nc(ccc1CNC(=O)C(C)c1ccc(NS(C)(=O)=O)c(F)c1)C(F)(F)Cl Show InChI InChI=1S/C21H25ClF3N3O4S/c1-4-5-10-32-20-15(7-9-18(27-20)21(22,24)25)12-26-19(29)13(2)14-6-8-17(16(23)11-14)28-33(3,30)31/h6-9,11,13,28H,4-5,10,12H2,1-3H3,(H,26,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of N-arachidonoyl dopamine-induced activity by FLIPR assay |
Eur J Med Chem 93: 101-8 (2015)
Article DOI: 10.1016/j.ejmech.2015.02.001
BindingDB Entry DOI: 10.7270/Q2N0188S |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50073159
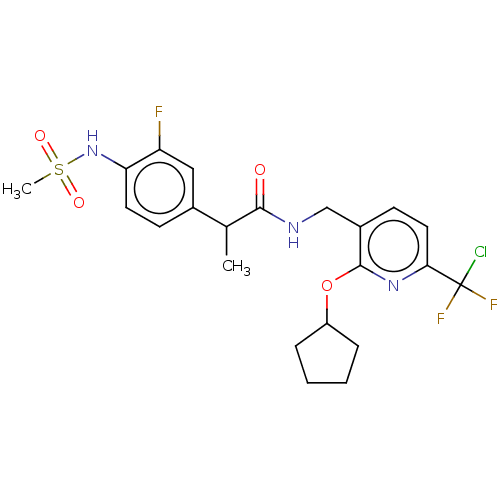
(CHEMBL3407765)Show SMILES CC(C(=O)NCc1ccc(nc1OC1CCCC1)C(F)(F)Cl)c1ccc(NS(C)(=O)=O)c(F)c1 Show InChI InChI=1S/C22H25ClF3N3O4S/c1-13(14-7-9-18(17(24)11-14)29-34(2,31)32)20(30)27-12-15-8-10-19(22(23,25)26)28-21(15)33-16-5-3-4-6-16/h7-11,13,16,29H,3-6,12H2,1-2H3,(H,27,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of N-arachidonoyl dopamine-induced activity by FLIPR assay |
Eur J Med Chem 93: 101-8 (2015)
Article DOI: 10.1016/j.ejmech.2015.02.001
BindingDB Entry DOI: 10.7270/Q2N0188S |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50442379
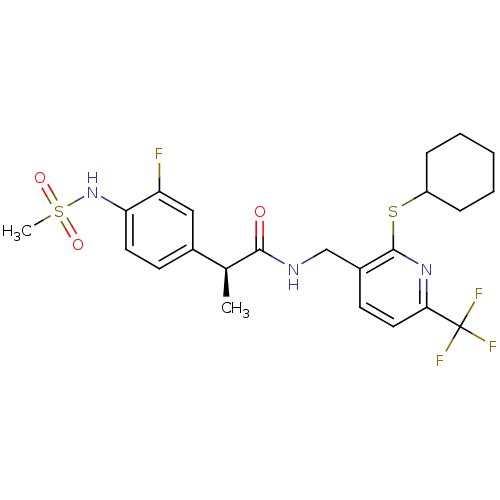
(CHEMBL2442912)Show SMILES C[C@H](C(=O)NCc1ccc(nc1SC1CCCCC1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 |r| Show InChI InChI=1S/C23H27F4N3O3S2/c1-14(15-8-10-19(18(24)12-15)30-35(2,32)33)21(31)28-13-16-9-11-20(23(25,26)27)29-22(16)34-17-6-4-3-5-7-17/h8-12,14,17,30H,3-7,13H2,1-2H3,(H,28,31)/t14-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHOK1 cells assessed as inhibition of N-acetyldopamine-induced activity after 5 mins by FLIPR assay |
Bioorg Med Chem 21: 6657-64 (2013)
Article DOI: 10.1016/j.bmc.2013.08.015
BindingDB Entry DOI: 10.7270/Q26Q1ZPN |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM176555
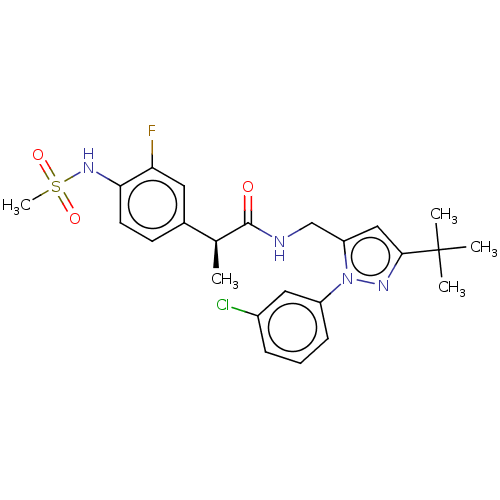
(US9120756, 17)Show SMILES C[C@H](C(=O)NCc1cc(nn1-c1cccc(Cl)c1)C(C)(C)C)c1ccc(NS(C)(=O)=O)c(F)c1 Show InChI InChI=1S/C24H28ClFN4O3S/c1-15(16-9-10-21(20(26)11-16)29-34(5,32)33)23(31)27-14-19-13-22(24(2,3)4)28-30(19)18-8-6-7-17(25)12-18/h6-13,15,29H,14H2,1-5H3,(H,27,31)/t15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 0.100 | -59.4 | n/a | n/a | n/a | n/a | n/a | n/a | 37 |
Gruenenthal GmbH
US Patent
| Assay Description
The agonistic or antagonistic effect of the substances to be tested on the vanilloid receptor 1 (VR1) can also be determined using the following assa... |
US Patent US9120756 (2015)
BindingDB Entry DOI: 10.7270/Q26D5RRG |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Rattus norvegicus (rat)) | BDBM176555

(US9120756, 17)Show SMILES C[C@H](C(=O)NCc1cc(nn1-c1cccc(Cl)c1)C(C)(C)C)c1ccc(NS(C)(=O)=O)c(F)c1 Show InChI InChI=1S/C24H28ClFN4O3S/c1-15(16-9-10-21(20(26)11-16)29-34(5,32)33)23(31)27-14-19-13-22(24(2,3)4)28-30(19)18-8-6-7-17(25)12-18/h6-13,15,29H,14H2,1-5H3,(H,27,31)/t15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Gruenenthal GmbH
US Patent
| Assay Description
The agonistic or antagonistic effect of the substances to be tested on the rat-species vanilloid receptor 1 (VR1/TRPV1) can be determined using the f... |
US Patent US9120756 (2015)
BindingDB Entry DOI: 10.7270/Q26D5RRG |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM176564
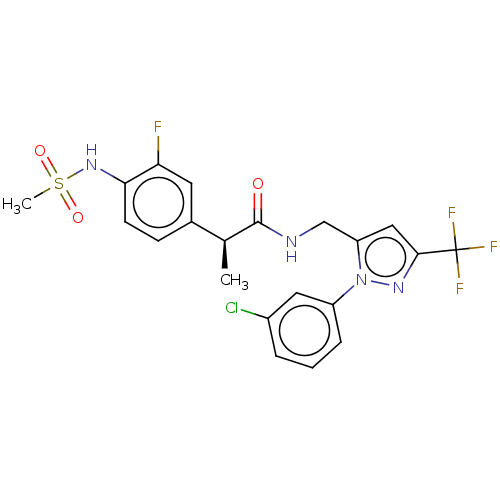
(US9120756, 26)Show SMILES C[C@H](C(=O)NCc1cc(nn1-c1cccc(Cl)c1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 Show InChI InChI=1S/C21H19ClF4N4O3S/c1-12(13-6-7-18(17(23)8-13)29-34(2,32)33)20(31)27-11-16-10-19(21(24,25)26)28-30(16)15-5-3-4-14(22)9-15/h3-10,12,29H,11H2,1-2H3,(H,27,31)/t12-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 0.100 | -59.4 | n/a | n/a | n/a | n/a | n/a | n/a | 37 |
Gruenenthal GmbH
US Patent
| Assay Description
The agonistic or antagonistic effect of the substances to be tested on the vanilloid receptor 1 (VR1) can also be determined using the following assa... |
US Patent US9120756 (2015)
BindingDB Entry DOI: 10.7270/Q26D5RRG |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Rattus norvegicus (rat)) | BDBM176564

(US9120756, 26)Show SMILES C[C@H](C(=O)NCc1cc(nn1-c1cccc(Cl)c1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 Show InChI InChI=1S/C21H19ClF4N4O3S/c1-12(13-6-7-18(17(23)8-13)29-34(2,32)33)20(31)27-11-16-10-19(21(24,25)26)28-30(16)15-5-3-4-14(22)9-15/h3-10,12,29H,11H2,1-2H3,(H,27,31)/t12-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Gruenenthal GmbH
US Patent
| Assay Description
The agonistic or antagonistic effect of the substances to be tested on the rat-species vanilloid receptor 1 (VR1/TRPV1) can be determined using the f... |
US Patent US9120756 (2015)
BindingDB Entry DOI: 10.7270/Q26D5RRG |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50046394
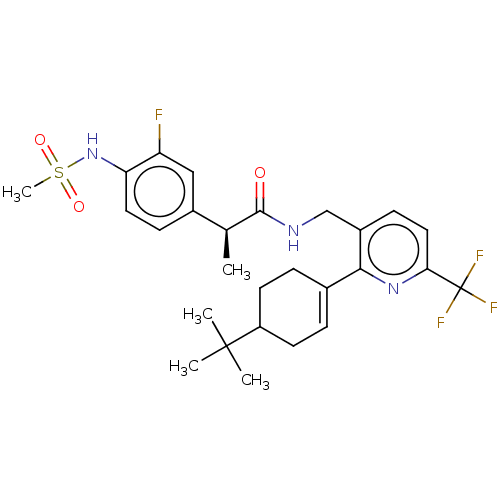
(CHEMBL3314409)Show SMILES C[C@H](C(=O)NCc1ccc(nc1C1=CCC(CC1)C(C)(C)C)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 |r,t:13| Show InChI InChI=1S/C27H33F4N3O3S/c1-16(18-8-12-22(21(28)14-18)34-38(5,36)37)25(35)32-15-19-9-13-23(27(29,30)31)33-24(19)17-6-10-20(11-7-17)26(2,3)4/h6,8-9,12-14,16,20,34H,7,10-11,15H2,1-5H3,(H,32,35)/t16-,20?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of capsaicin-induced increase in intracellular Ca2+ level by FLIPR a... |
Bioorg Med Chem Lett 24: 4039-43 (2014)
Article DOI: 10.1016/j.bmcl.2014.05.074
BindingDB Entry DOI: 10.7270/Q2B859RZ |
More data for this
Ligand-Target Pair | |
Nociceptin receptor
(Homo sapiens (Human)) | BDBM50100983
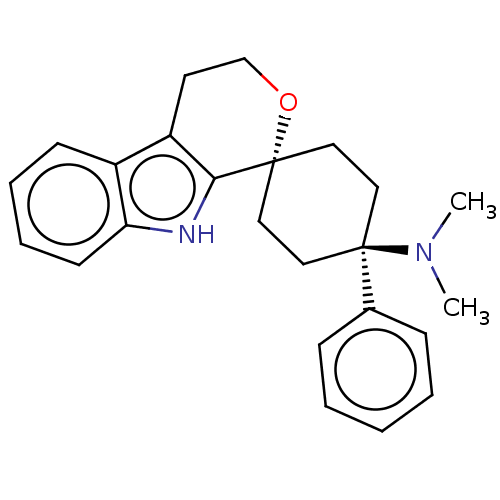
(CHEMBL3326224)Show SMILES Cl.CN(C)[C@]1(CC[C@@]2(CC1)OCCc1c2[nH]c2ccccc12)c1ccccc1 |r,wU:4.2,wD:7.9,(22.71,-7.97,;18.3,-7.93,;16.96,-7.16,;16.96,-5.62,;15.63,-7.93,;14.86,-9.26,;13.32,-9.27,;12.55,-7.94,;13.32,-6.61,;14.86,-6.6,;11.79,-9.28,;10.25,-9.27,;9.47,-7.94,;10.25,-6.6,;11.79,-6.61,;12.26,-5.14,;11.03,-4.24,;10.87,-2.7,;9.47,-2.07,;8.21,-2.98,;8.37,-4.51,;9.77,-5.14,;16.43,-9.25,;15.68,-10.6,;16.48,-11.91,;18.02,-11.88,;18.76,-10.52,;17.96,-9.21,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmacokinetics
Curated by ChEMBL
| Assay Description
Displacement of [3H]nociceptin from human NOP receptor expressed in CHO-K1 cells by scintillation proximity assay |
ACS Med Chem Lett 5: 857-62 (2014)
Article DOI: 10.1021/ml500117c
BindingDB Entry DOI: 10.7270/Q25140ZK |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50046396
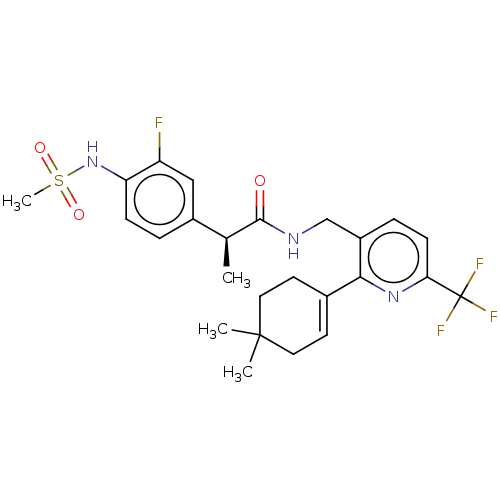
(CHEMBL3314411)Show SMILES C[C@H](C(=O)NCc1ccc(nc1C1=CCC(C)(C)CC1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 |r,t:13| Show InChI InChI=1S/C25H29F4N3O3S/c1-15(17-5-7-20(19(26)13-17)32-36(4,34)35)23(33)30-14-18-6-8-21(25(27,28)29)31-22(18)16-9-11-24(2,3)12-10-16/h5-9,13,15,32H,10-12,14H2,1-4H3,(H,30,33)/t15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of capsaicin-induced increase in intracellular Ca2+ level by FLIPR a... |
Bioorg Med Chem Lett 24: 4039-43 (2014)
Article DOI: 10.1016/j.bmcl.2014.05.074
BindingDB Entry DOI: 10.7270/Q2B859RZ |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50046379
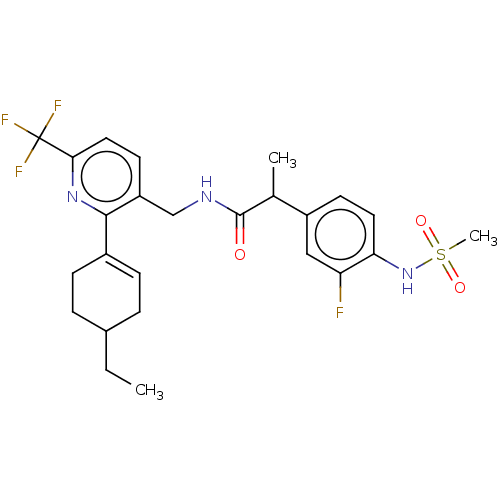
(CHEMBL3314407)Show SMILES CCC1CCC(=CC1)c1nc(ccc1CNC(=O)C(C)c1ccc(NS(C)(=O)=O)c(F)c1)C(F)(F)F |c:5| Show InChI InChI=1S/C25H29F4N3O3S/c1-4-16-5-7-17(8-6-16)23-19(10-12-22(31-23)25(27,28)29)14-30-24(33)15(2)18-9-11-21(20(26)13-18)32-36(3,34)35/h7,9-13,15-16,32H,4-6,8,14H2,1-3H3,(H,30,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of capsaicin-induced increase in intracellular Ca2+ level by FLIPR a... |
Bioorg Med Chem Lett 24: 4039-43 (2014)
Article DOI: 10.1016/j.bmcl.2014.05.074
BindingDB Entry DOI: 10.7270/Q2B859RZ |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50046377
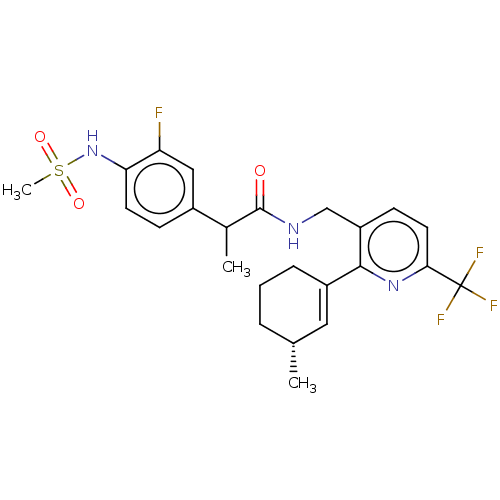
(CHEMBL3314406)Show SMILES CC(C(=O)NCc1ccc(nc1C1=C[C@H](C)CCC1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 |r,t:13| Show InChI InChI=1S/C24H27F4N3O3S/c1-14-5-4-6-17(11-14)22-18(8-10-21(30-22)24(26,27)28)13-29-23(32)15(2)16-7-9-20(19(25)12-16)31-35(3,33)34/h7-12,14-15,31H,4-6,13H2,1-3H3,(H,29,32)/t14-,15?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of capsaicin-induced increase in intracellular Ca2+ level by FLIPR a... |
Bioorg Med Chem Lett 24: 4039-43 (2014)
Article DOI: 10.1016/j.bmcl.2014.05.074
BindingDB Entry DOI: 10.7270/Q2B859RZ |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM342149

(1-((2-(tert-butyl)-4-(3-chlorophenyl)thiazol-5-yl)...)Show SMILES CC(C)(C)c1nc(c(CNC(=O)Nc2cccc3cnccc23)s1)-c1cccc(Cl)c1 Show InChI InChI=1S/C24H23ClN4OS/c1-24(2,3)22-29-21(15-6-4-8-17(25)12-15)20(31-22)14-27-23(30)28-19-9-5-7-16-13-26-11-10-18(16)19/h4-13H,14H2,1-3H3,(H2,27,28,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medifron DBT Inc.
US Patent
| Assay Description
The FLIPR protocol consists of 2 substance additions during a kinetic measurement. First the compounds to be tested (5 μM) are pipetted onto the... |
US Patent US9771359 (2017)
BindingDB Entry DOI: 10.7270/Q2CZ3984 |
More data for this
Ligand-Target Pair | |
Nociceptin receptor
(Homo sapiens (Human)) | BDBM50101099

(CHEMBL3326228)Show SMILES Cl.CN(C)[C@]1(CC[C@@]2(CC1)NCCc1c2[nH]c2ccccc12)c1ccccc1 |r,wU:4.2,wD:7.9,(19.24,-9.37,;10.32,-12.36,;8.84,-12.77,;7.74,-11.68,;8.44,-14.27,;7.67,-15.59,;6.14,-15.59,;5.38,-14.26,;6.14,-12.93,;7.68,-12.94,;4.47,-15.52,;2.93,-15.35,;2.3,-13.94,;3.21,-12.69,;4.75,-12.85,;5.38,-11.44,;4.23,-10.41,;4.23,-8.85,;2.89,-8.09,;1.56,-8.86,;1.56,-10.41,;2.89,-11.18,;9.98,-14.28,;10.74,-15.62,;12.28,-15.63,;13.06,-14.29,;12.29,-12.95,;10.75,-12.95,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmacokinetics
Curated by ChEMBL
| Assay Description
Displacement of [3H]nociceptin from human NOP receptor expressed in CHO-K1 cells by scintillation proximity assay |
ACS Med Chem Lett 5: 857-62 (2014)
Article DOI: 10.1021/ml500117c
BindingDB Entry DOI: 10.7270/Q25140ZK |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50049529
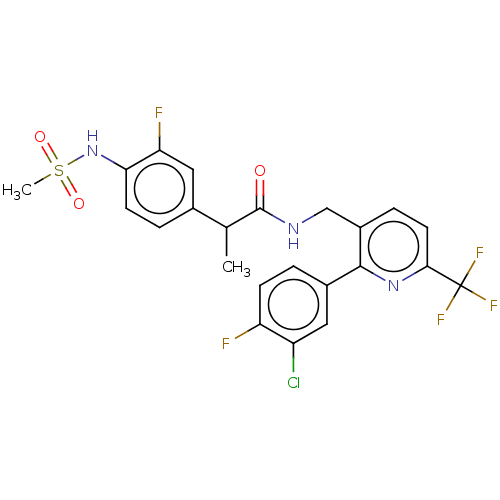
(CHEMBL3317476)Show SMILES CC(C(=O)NCc1ccc(nc1-c1ccc(F)c(Cl)c1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 Show InChI InChI=1S/C23H19ClF5N3O3S/c1-12(13-4-7-19(18(26)10-13)32-36(2,34)35)22(33)30-11-15-5-8-20(23(27,28)29)31-21(15)14-3-6-17(25)16(24)9-14/h3-10,12,32H,11H2,1-2H3,(H,30,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity against human TRPV1 expressed in CHO cells assessed as inhibition of capsaicin-induced channel activation by FLIPR assay |
Bioorg Med Chem Lett 24: 4044-7 (2014)
Article DOI: 10.1016/j.bmcl.2014.05.072
BindingDB Entry DOI: 10.7270/Q2JH3NTH |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50061298

(CHEMBL3393837)Show SMILES CC1CCN(CC1)c1nc(ccc1CNC(=O)Nc1cccc2cnccc12)C(F)(F)F Show InChI InChI=1S/C23H24F3N5O/c1-15-8-11-31(12-9-15)21-17(5-6-20(30-21)23(24,25)26)14-28-22(32)29-19-4-2-3-16-13-27-10-7-18(16)19/h2-7,10,13,15H,8-9,11-12,14H2,1H3,(H2,28,29,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 assessed as inhibition of capsaicin-induced effect by FLIPR assay |
Bioorg Med Chem Lett 25: 803-6 (2015)
Article DOI: 10.1016/j.bmcl.2014.12.086
BindingDB Entry DOI: 10.7270/Q2JD4ZG3 |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50046299

(CHEMBL3314393)Show SMILES CCC1CCC(CC1)c1nc(ccc1CNC(=O)[C@@H](C)c1ccc(NS(C)(=O)=O)c(F)c1)C(F)(F)F |r,wU:18.20,(10.24,-14.41,;8.9,-13.63,;8.91,-12.08,;7.58,-11.3,;7.58,-9.77,;8.92,-9,;10.25,-9.76,;10.24,-11.31,;8.92,-7.46,;7.59,-6.69,;7.59,-5.14,;8.92,-4.37,;10.25,-5.13,;10.26,-6.68,;11.59,-7.45,;12.92,-6.68,;14.26,-7.45,;14.26,-8.99,;15.59,-6.68,;15.59,-5.14,;16.93,-7.44,;16.92,-8.98,;18.25,-9.75,;19.59,-8.98,;20.92,-9.75,;22.26,-8.97,;23.59,-9.74,;23.02,-7.64,;21.48,-7.64,;19.58,-7.43,;20.91,-6.66,;18.25,-6.67,;6.25,-4.37,;6.25,-2.83,;4.92,-5.14,;4.92,-3.6,)| Show InChI InChI=1S/C25H31F4N3O3S/c1-4-16-5-7-17(8-6-16)23-19(10-12-22(31-23)25(27,28)29)14-30-24(33)15(2)18-9-11-21(20(26)13-18)32-36(3,34)35/h9-13,15-17,32H,4-8,14H2,1-3H3,(H,30,33)/t15-,16?,17?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of capsaicin-induced increase in intracellular Ca2+ level by FLIPR a... |
Bioorg Med Chem Lett 24: 4039-43 (2014)
Article DOI: 10.1016/j.bmcl.2014.05.074
BindingDB Entry DOI: 10.7270/Q2B859RZ |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50046295
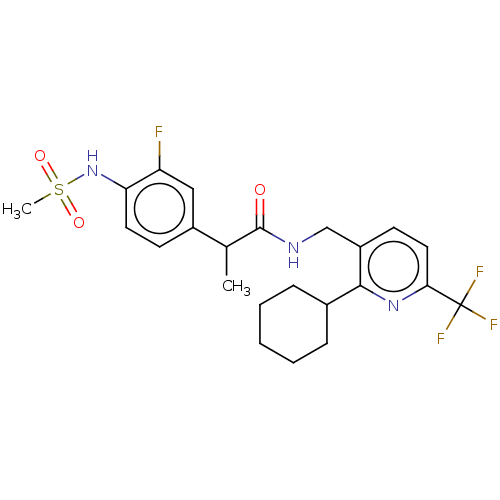
(CHEMBL3314389)Show SMILES CC(C(=O)NCc1ccc(nc1C1CCCCC1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 Show InChI InChI=1S/C23H27F4N3O3S/c1-14(16-8-10-19(18(24)12-16)30-34(2,32)33)22(31)28-13-17-9-11-20(23(25,26)27)29-21(17)15-6-4-3-5-7-15/h8-12,14-15,30H,3-7,13H2,1-2H3,(H,28,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of N-arachidonoyl dopamine-induced increase in intracellular Ca2+ le... |
Bioorg Med Chem Lett 24: 4039-43 (2014)
Article DOI: 10.1016/j.bmcl.2014.05.074
BindingDB Entry DOI: 10.7270/Q2B859RZ |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Rattus norvegicus (rat)) | BDBM176554
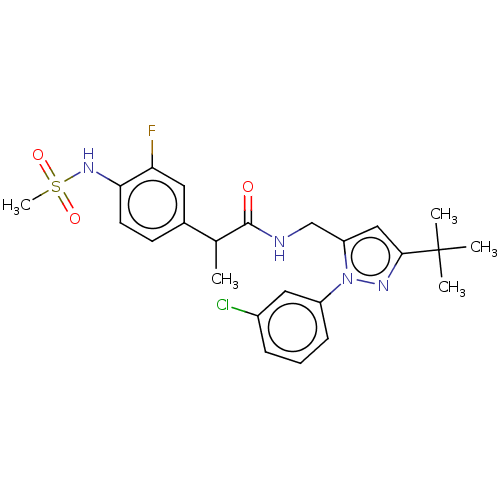
(US9120756, 16)Show SMILES CC(C(=O)NCc1cc(nn1-c1cccc(Cl)c1)C(C)(C)C)c1ccc(NS(C)(=O)=O)c(F)c1 Show InChI InChI=1S/C24H28ClFN4O3S/c1-15(16-9-10-21(20(26)11-16)29-34(5,32)33)23(31)27-14-19-13-22(24(2,3)4)28-30(19)18-8-6-7-17(25)12-18/h6-13,15,29H,14H2,1-5H3,(H,27,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Gruenenthal GmbH
US Patent
| Assay Description
The agonistic or antagonistic effect of the substances to be tested on the rat-species vanilloid receptor 1 (VR1/TRPV1) can be determined using the f... |
US Patent US9120756 (2015)
BindingDB Entry DOI: 10.7270/Q26D5RRG |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50046366
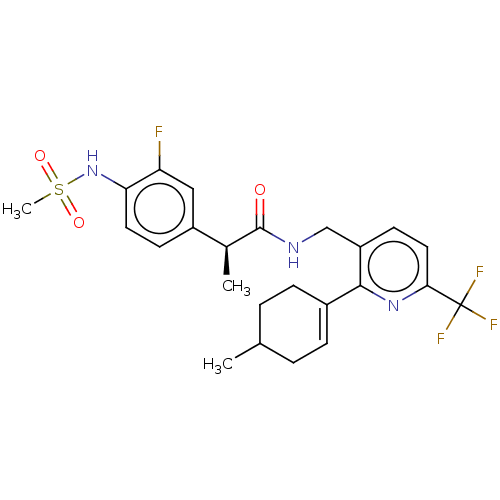
(CHEMBL3314404)Show SMILES C[C@H](C(=O)NCc1ccc(nc1C1=CCC(C)CC1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 |r,t:13| Show InChI InChI=1S/C24H27F4N3O3S/c1-14-4-6-16(7-5-14)22-18(9-11-21(30-22)24(26,27)28)13-29-23(32)15(2)17-8-10-20(19(25)12-17)31-35(3,33)34/h6,8-12,14-15,31H,4-5,7,13H2,1-3H3,(H,29,32)/t14?,15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of capsaicin-induced increase in intracellular Ca2+ level by FLIPR a... |
Bioorg Med Chem Lett 24: 4039-43 (2014)
Article DOI: 10.1016/j.bmcl.2014.05.074
BindingDB Entry DOI: 10.7270/Q2B859RZ |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50046375

(CHEMBL3314405)Show SMILES CC(C(=O)NCc1ccc(nc1C1=CC(C)CCC1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 |t:13| Show InChI InChI=1S/C24H27F4N3O3S/c1-14-5-4-6-17(11-14)22-18(8-10-21(30-22)24(26,27)28)13-29-23(32)15(2)16-7-9-20(19(25)12-16)31-35(3,33)34/h7-12,14-15,31H,4-6,13H2,1-3H3,(H,29,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of capsaicin-induced increase in intracellular Ca2+ level by FLIPR a... |
Bioorg Med Chem Lett 24: 4039-43 (2014)
Article DOI: 10.1016/j.bmcl.2014.05.074
BindingDB Entry DOI: 10.7270/Q2B859RZ |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50046388

(CHEMBL3314408)Show SMILES CC(C(=O)NCc1ccc(nc1C1=CCC(CC1)C(C)(C)C)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 |t:13| Show InChI InChI=1S/C27H33F4N3O3S/c1-16(18-8-12-22(21(28)14-18)34-38(5,36)37)25(35)32-15-19-9-13-23(27(29,30)31)33-24(19)17-6-10-20(11-7-17)26(2,3)4/h6,8-9,12-14,16,20,34H,7,10-11,15H2,1-5H3,(H,32,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of capsaicin-induced increase in intracellular Ca2+ level by FLIPR a... |
Bioorg Med Chem Lett 24: 4039-43 (2014)
Article DOI: 10.1016/j.bmcl.2014.05.074
BindingDB Entry DOI: 10.7270/Q2B859RZ |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50046282

(CHEMBL3314377)Show SMILES CC(C(=O)NCc1ccc(nc1Cl)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 Show InChI InChI=1S/C17H16ClF4N3O3S/c1-9(10-3-5-13(12(19)7-10)25-29(2,27)28)16(26)23-8-11-4-6-14(17(20,21)22)24-15(11)18/h3-7,9,25H,8H2,1-2H3,(H,23,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of capsaicin-induced increase in intracellular Ca2+ level by FLIPR a... |
Bioorg Med Chem Lett 24: 4039-43 (2014)
Article DOI: 10.1016/j.bmcl.2014.05.074
BindingDB Entry DOI: 10.7270/Q2B859RZ |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50046395

(CHEMBL3314410)Show SMILES CC(C(=O)NCc1ccc(nc1C1=CCC(C)(C)CC1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 |t:13| Show InChI InChI=1S/C25H29F4N3O3S/c1-15(17-5-7-20(19(26)13-17)32-36(4,34)35)23(33)30-14-18-6-8-21(25(27,28)29)31-22(18)16-9-11-24(2,3)12-10-16/h5-9,13,15,32H,10-12,14H2,1-4H3,(H,30,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of capsaicin-induced increase in intracellular Ca2+ level by FLIPR a... |
Bioorg Med Chem Lett 24: 4039-43 (2014)
Article DOI: 10.1016/j.bmcl.2014.05.074
BindingDB Entry DOI: 10.7270/Q2B859RZ |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50398494

(CHEMBL2177429)Show SMILES C[C@H](C(=O)NCc1ccc(nc1N1CCC(C)CC1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 |r| Show InChI InChI=1S/C23H28F4N4O3S/c1-14-8-10-31(11-9-14)21-17(5-7-20(29-21)23(25,26)27)13-28-22(32)15(2)16-4-6-19(18(24)12-16)30-35(3,33)34/h4-7,12,14-15,30H,8-11,13H2,1-3H3,(H,28,32)/t15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of capsaicin-induced increase in intracellular Ca2+ level by FLIPR a... |
Bioorg Med Chem Lett 24: 4039-43 (2014)
Article DOI: 10.1016/j.bmcl.2014.05.074
BindingDB Entry DOI: 10.7270/Q2B859RZ |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50398494

(CHEMBL2177429)Show SMILES C[C@H](C(=O)NCc1ccc(nc1N1CCC(C)CC1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 |r| Show InChI InChI=1S/C23H28F4N4O3S/c1-14-8-10-31(11-9-14)21-17(5-7-20(29-21)23(25,26)27)13-28-22(32)15(2)16-4-6-19(18(24)12-16)30-35(3,33)34/h4-7,12,14-15,30H,8-11,13H2,1-3H3,(H,28,32)/t15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHOK1 cells assessed as inhibition of capsaicin-induced activity by FLIPR assay |
Bioorg Med Chem 21: 6657-64 (2013)
Article DOI: 10.1016/j.bmc.2013.08.015
BindingDB Entry DOI: 10.7270/Q26Q1ZPN |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50101306
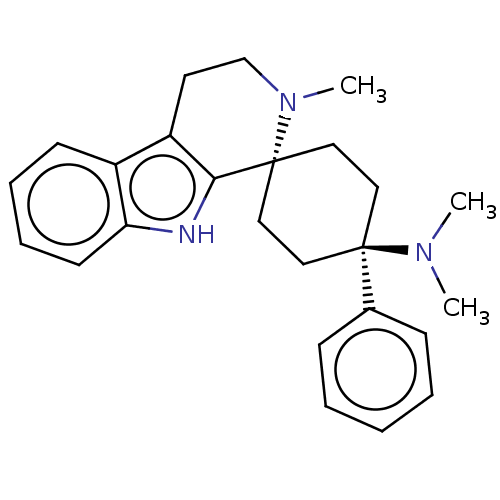
(CHEMBL3326229)Show SMILES Cl.CN(C)[C@]1(CC[C@@]2(CC1)N(C)CCc1c2[nH]c2ccccc12)c1ccccc1 |r,wU:4.2,wD:7.9,(35,-11.75,;25.44,-13.91,;23.96,-14.32,;22.86,-13.23,;23.56,-15.82,;22.79,-17.14,;21.26,-17.14,;20.5,-15.8,;21.26,-14.48,;22.8,-14.49,;19.59,-17.06,;20.22,-18.47,;18.05,-16.9,;17.42,-15.49,;18.33,-14.24,;19.87,-14.4,;20.5,-12.99,;19.35,-11.95,;19.35,-10.4,;18.01,-9.64,;16.68,-10.41,;16.68,-11.95,;18.01,-12.72,;25.1,-15.82,;25.86,-17.16,;27.4,-17.17,;28.18,-15.84,;27.4,-14.5,;25.87,-14.5,)| | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmacokinetics
Curated by ChEMBL
| Assay Description
Displacement of [3H]naloxone from human MOP receptor expressed in CHO-K1 cells by scintillation proximity assay |
ACS Med Chem Lett 5: 857-62 (2014)
Article DOI: 10.1021/ml500117c
BindingDB Entry DOI: 10.7270/Q25140ZK |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50100983

(CHEMBL3326224)Show SMILES Cl.CN(C)[C@]1(CC[C@@]2(CC1)OCCc1c2[nH]c2ccccc12)c1ccccc1 |r,wU:4.2,wD:7.9,(22.71,-7.97,;18.3,-7.93,;16.96,-7.16,;16.96,-5.62,;15.63,-7.93,;14.86,-9.26,;13.32,-9.27,;12.55,-7.94,;13.32,-6.61,;14.86,-6.6,;11.79,-9.28,;10.25,-9.27,;9.47,-7.94,;10.25,-6.6,;11.79,-6.61,;12.26,-5.14,;11.03,-4.24,;10.87,-2.7,;9.47,-2.07,;8.21,-2.98,;8.37,-4.51,;9.77,-5.14,;16.43,-9.25,;15.68,-10.6,;16.48,-11.91,;18.02,-11.88,;18.76,-10.52,;17.96,-9.21,)| | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmacokinetics
Curated by ChEMBL
| Assay Description
Displacement of [3H]naloxone from human MOP receptor expressed in CHO-K1 cells by scintillation proximity assay |
ACS Med Chem Lett 5: 857-62 (2014)
Article DOI: 10.1021/ml500117c
BindingDB Entry DOI: 10.7270/Q25140ZK |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50046341
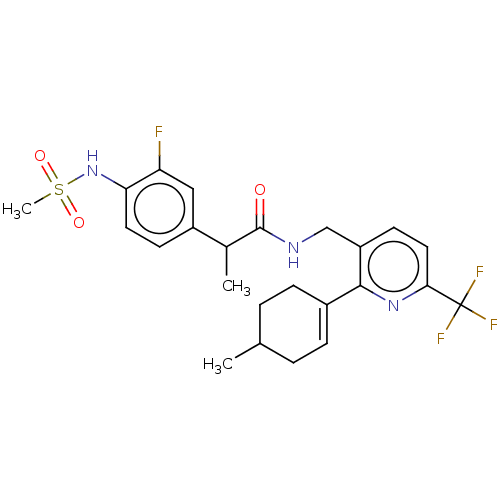
(CHEMBL3314403)Show SMILES CC(C(=O)NCc1ccc(nc1C1=CCC(C)CC1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 |t:13| Show InChI InChI=1S/C24H27F4N3O3S/c1-14-4-6-16(7-5-14)22-18(9-11-21(30-22)24(26,27)28)13-29-23(32)15(2)17-8-10-20(19(25)12-17)31-35(3,33)34/h6,8-12,14-15,31H,4-5,7,13H2,1-3H3,(H,29,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of capsaicin-induced increase in intracellular Ca2+ level by FLIPR a... |
Bioorg Med Chem Lett 24: 4039-43 (2014)
Article DOI: 10.1016/j.bmcl.2014.05.074
BindingDB Entry DOI: 10.7270/Q2B859RZ |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50046304
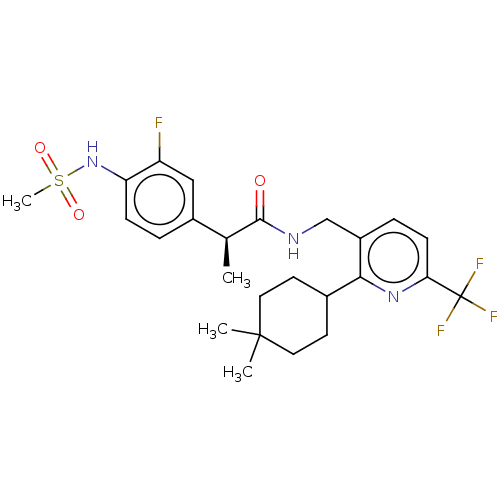
(CHEMBL3314398)Show SMILES C[C@H](C(=O)NCc1ccc(nc1C1CCC(C)(C)CC1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 |r| Show InChI InChI=1S/C25H31F4N3O3S/c1-15(17-5-7-20(19(26)13-17)32-36(4,34)35)23(33)30-14-18-6-8-21(25(27,28)29)31-22(18)16-9-11-24(2,3)12-10-16/h5-8,13,15-16,32H,9-12,14H2,1-4H3,(H,30,33)/t15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of capsaicin-induced increase in intracellular Ca2+ level by FLIPR a... |
Bioorg Med Chem Lett 24: 4039-43 (2014)
Article DOI: 10.1016/j.bmcl.2014.05.074
BindingDB Entry DOI: 10.7270/Q2B859RZ |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50046302

(CHEMBL3314396)Show SMILES CC(C(=O)NCc1ccc(nc1[C@H]1CC[C@@H](CC1)C(C)(C)C)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 |r,wU:12.12,wD:15.19,(15.59,-5.14,;15.59,-6.68,;14.26,-7.45,;14.26,-8.99,;12.92,-6.68,;11.59,-7.45,;10.26,-6.68,;10.25,-5.13,;8.92,-4.37,;7.59,-5.14,;7.59,-6.68,;8.92,-7.46,;8.92,-8.99,;10.25,-9.76,;10.24,-11.31,;8.91,-12.07,;7.58,-11.3,;7.58,-9.76,;8.9,-13.63,;10.24,-14.41,;7.55,-14.4,;8.9,-15.18,;6.25,-4.37,;6.25,-2.83,;4.92,-5.14,;4.91,-3.6,;16.93,-7.44,;16.92,-8.98,;18.25,-9.75,;19.59,-8.98,;20.92,-9.75,;22.25,-8.97,;23.59,-9.74,;23.02,-7.64,;21.48,-7.64,;19.58,-7.43,;20.91,-6.66,;18.24,-6.67,)| Show InChI InChI=1S/C27H35F4N3O3S/c1-16(18-8-12-22(21(28)14-18)34-38(5,36)37)25(35)32-15-19-9-13-23(27(29,30)31)33-24(19)17-6-10-20(11-7-17)26(2,3)4/h8-9,12-14,16-17,20,34H,6-7,10-11,15H2,1-5H3,(H,32,35)/t16?,17-,20- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of capsaicin-induced increase in intracellular Ca2+ level by FLIPR a... |
Bioorg Med Chem Lett 24: 4039-43 (2014)
Article DOI: 10.1016/j.bmcl.2014.05.074
BindingDB Entry DOI: 10.7270/Q2B859RZ |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50046300
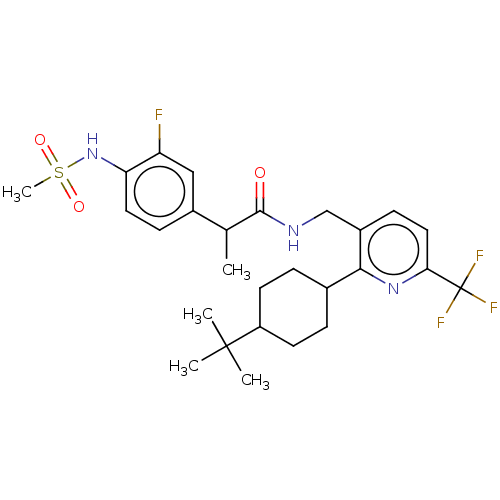
(CHEMBL3314394)Show SMILES CC(C(=O)NCc1ccc(nc1C1CCC(CC1)C(C)(C)C)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 |(15.59,-5.14,;15.59,-6.68,;14.26,-7.45,;14.26,-8.99,;12.92,-6.68,;11.59,-7.45,;10.26,-6.68,;10.25,-5.13,;8.92,-4.37,;7.59,-5.14,;7.59,-6.68,;8.92,-7.46,;8.92,-8.99,;7.58,-9.76,;7.58,-11.3,;8.91,-12.07,;10.24,-11.31,;10.25,-9.76,;8.9,-13.63,;10.24,-14.41,;7.55,-14.4,;8.9,-15.18,;6.25,-4.37,;6.25,-2.83,;4.92,-5.14,;4.91,-3.6,;16.93,-7.44,;16.92,-8.98,;18.25,-9.75,;19.59,-8.98,;20.92,-9.75,;22.25,-8.97,;23.59,-9.74,;23.02,-7.64,;21.48,-7.64,;19.58,-7.43,;20.91,-6.66,;18.24,-6.67,)| Show InChI InChI=1S/C27H35F4N3O3S/c1-16(18-8-12-22(21(28)14-18)34-38(5,36)37)25(35)32-15-19-9-13-23(27(29,30)31)33-24(19)17-6-10-20(11-7-17)26(2,3)4/h8-9,12-14,16-17,20,34H,6-7,10-11,15H2,1-5H3,(H,32,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of capsaicin-induced increase in intracellular Ca2+ level by FLIPR a... |
Bioorg Med Chem Lett 24: 4039-43 (2014)
Article DOI: 10.1016/j.bmcl.2014.05.074
BindingDB Entry DOI: 10.7270/Q2B859RZ |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50046298
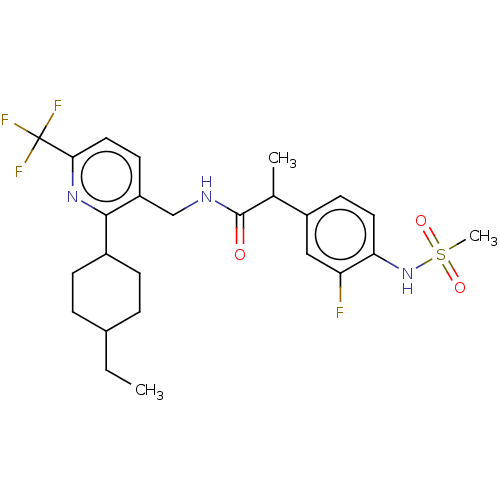
(CHEMBL3314392)Show SMILES CCC1CCC(CC1)c1nc(ccc1CNC(=O)C(C)c1ccc(NS(C)(=O)=O)c(F)c1)C(F)(F)F |(10.24,-14.41,;8.9,-13.63,;8.91,-12.08,;7.58,-11.3,;7.58,-9.77,;8.92,-9,;10.25,-9.76,;10.24,-11.31,;8.92,-7.46,;7.59,-6.69,;7.59,-5.14,;8.92,-4.37,;10.25,-5.13,;10.26,-6.68,;11.59,-7.45,;12.92,-6.68,;14.26,-7.45,;14.26,-8.99,;15.59,-6.68,;15.59,-5.14,;16.93,-7.44,;16.92,-8.98,;18.25,-9.75,;19.59,-8.98,;20.92,-9.75,;22.26,-8.97,;23.59,-9.74,;23.02,-7.64,;21.48,-7.64,;19.58,-7.43,;20.91,-6.66,;18.25,-6.67,;6.25,-4.37,;6.25,-2.83,;4.92,-5.14,;4.92,-3.6,)| Show InChI InChI=1S/C25H31F4N3O3S/c1-4-16-5-7-17(8-6-16)23-19(10-12-22(31-23)25(27,28)29)14-30-24(33)15(2)18-9-11-21(20(26)13-18)32-36(3,34)35/h9-13,15-17,32H,4-8,14H2,1-3H3,(H,30,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of capsaicin-induced increase in intracellular Ca2+ level by FLIPR a... |
Bioorg Med Chem Lett 24: 4039-43 (2014)
Article DOI: 10.1016/j.bmcl.2014.05.074
BindingDB Entry DOI: 10.7270/Q2B859RZ |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50046296
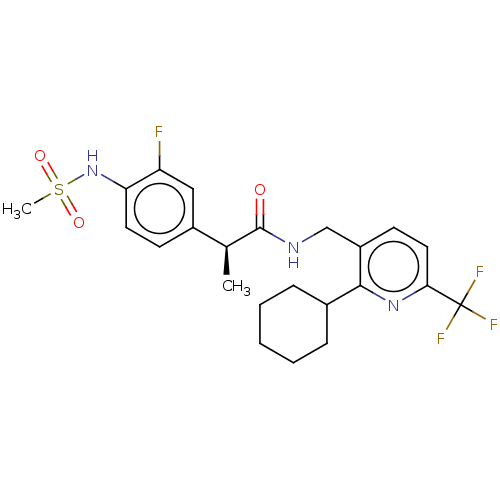
(CHEMBL3314390)Show SMILES C[C@H](C(=O)NCc1ccc(nc1C1CCCCC1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 |r| Show InChI InChI=1S/C23H27F4N3O3S/c1-14(16-8-10-19(18(24)12-16)30-34(2,32)33)22(31)28-13-17-9-11-20(23(25,26)27)29-21(17)15-6-4-3-5-7-15/h8-12,14-15,30H,3-7,13H2,1-2H3,(H,28,31)/t14-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of capsaicin-induced increase in intracellular Ca2+ level by FLIPR a... |
Bioorg Med Chem Lett 24: 4039-43 (2014)
Article DOI: 10.1016/j.bmcl.2014.05.074
BindingDB Entry DOI: 10.7270/Q2B859RZ |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50073156
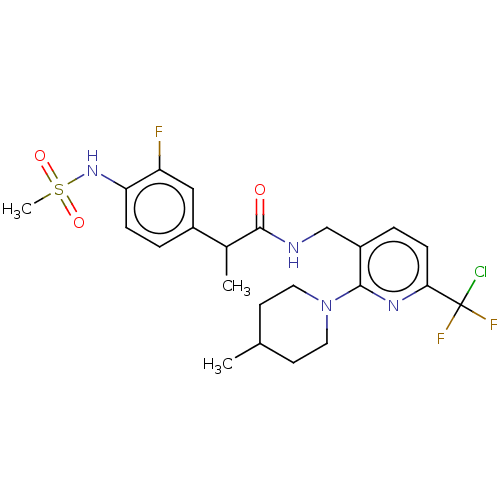
(CHEMBL3407753)Show SMILES CC(C(=O)NCc1ccc(nc1N1CCC(C)CC1)C(F)(F)Cl)c1ccc(NS(C)(=O)=O)c(F)c1 Show InChI InChI=1S/C23H28ClF3N4O3S/c1-14-8-10-31(11-9-14)21-17(5-7-20(29-21)23(24,26)27)13-28-22(32)15(2)16-4-6-19(18(25)12-16)30-35(3,33)34/h4-7,12,14-15,30H,8-11,13H2,1-3H3,(H,28,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of capsaicin-induced activity by FLIPR assay |
Eur J Med Chem 93: 101-8 (2015)
Article DOI: 10.1016/j.ejmech.2015.02.001
BindingDB Entry DOI: 10.7270/Q2N0188S |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM176562
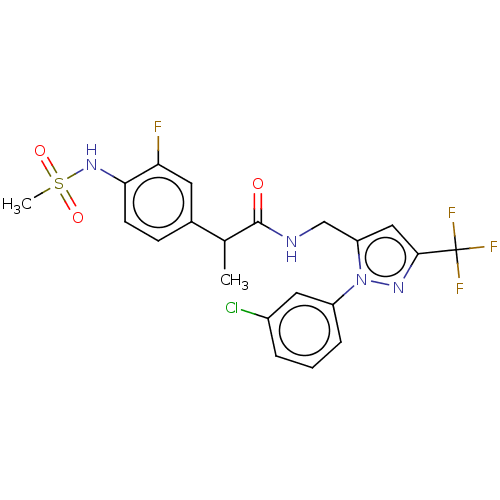
(US9120756, 24)Show SMILES CC(C(=O)NCc1cc(nn1-c1cccc(Cl)c1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 Show InChI InChI=1S/C21H19ClF4N4O3S/c1-12(13-6-7-18(17(23)8-13)29-34(2,32)33)20(31)27-11-16-10-19(21(24,25)26)28-30(16)15-5-3-4-14(22)9-15/h3-10,12,29H,11H2,1-2H3,(H,27,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 0.300 | -56.5 | n/a | n/a | n/a | n/a | n/a | n/a | 37 |
Gruenenthal GmbH
US Patent
| Assay Description
The agonistic or antagonistic effect of the substances to be tested on the vanilloid receptor 1 (VR1) can also be determined using the following assa... |
US Patent US9120756 (2015)
BindingDB Entry DOI: 10.7270/Q26D5RRG |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM176561

(US9120756, 23)Show SMILES C[C@H](C(=O)NCc1cc(nn1-c1ccc(Cl)cc1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 Show InChI InChI=1S/C21H19ClF4N4O3S/c1-12(13-3-8-18(17(23)9-13)29-34(2,32)33)20(31)27-11-16-10-19(21(24,25)26)28-30(16)15-6-4-14(22)5-7-15/h3-10,12,29H,11H2,1-2H3,(H,27,31)/t12-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 0.300 | -56.5 | n/a | n/a | n/a | n/a | n/a | n/a | 37 |
Gruenenthal GmbH
US Patent
| Assay Description
The agonistic or antagonistic effect of the substances to be tested on the vanilloid receptor 1 (VR1) can also be determined using the following assa... |
US Patent US9120756 (2015)
BindingDB Entry DOI: 10.7270/Q26D5RRG |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM176559
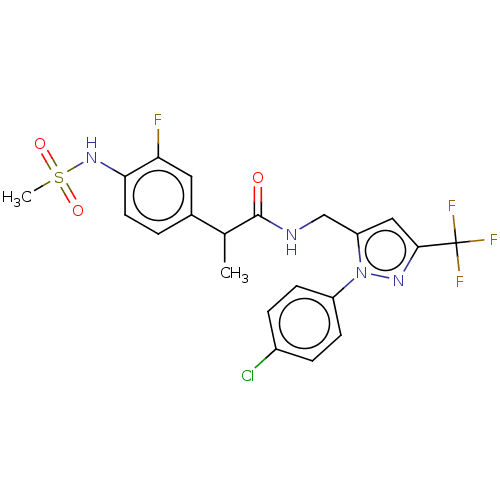
(US9120756, 21)Show SMILES CC(C(=O)NCc1cc(nn1-c1ccc(Cl)cc1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 Show InChI InChI=1S/C21H19ClF4N4O3S/c1-12(13-3-8-18(17(23)9-13)29-34(2,32)33)20(31)27-11-16-10-19(21(24,25)26)28-30(16)15-6-4-14(22)5-7-15/h3-10,12,29H,11H2,1-2H3,(H,27,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 0.300 | -56.5 | n/a | n/a | n/a | n/a | n/a | n/a | 37 |
Gruenenthal GmbH
US Patent
| Assay Description
The agonistic or antagonistic effect of the substances to be tested on the vanilloid receptor 1 (VR1) can also be determined using the following assa... |
US Patent US9120756 (2015)
BindingDB Entry DOI: 10.7270/Q26D5RRG |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM176554

(US9120756, 16)Show SMILES CC(C(=O)NCc1cc(nn1-c1cccc(Cl)c1)C(C)(C)C)c1ccc(NS(C)(=O)=O)c(F)c1 Show InChI InChI=1S/C24H28ClFN4O3S/c1-15(16-9-10-21(20(26)11-16)29-34(5,32)33)23(31)27-14-19-13-22(24(2,3)4)28-30(19)18-8-6-7-17(25)12-18/h6-13,15,29H,14H2,1-5H3,(H,27,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 0.300 | -56.5 | n/a | n/a | n/a | n/a | n/a | n/a | 37 |
Gruenenthal GmbH
US Patent
| Assay Description
The agonistic or antagonistic effect of the substances to be tested on the vanilloid receptor 1 (VR1) can also be determined using the following assa... |
US Patent US9120756 (2015)
BindingDB Entry DOI: 10.7270/Q26D5RRG |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM342128
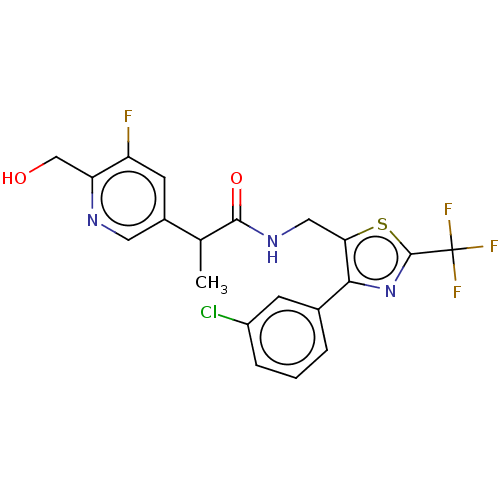
(N-((4-(3-chlorophenyl)-2-(trifluoromethyl)thiazol-...)Show SMILES CC(C(=O)NCc1sc(nc1-c1cccc(Cl)c1)C(F)(F)F)c1cnc(CO)c(F)c1 Show InChI InChI=1S/C20H16ClF4N3O2S/c1-10(12-6-14(22)15(9-29)26-7-12)18(30)27-8-16-17(11-3-2-4-13(21)5-11)28-19(31-16)20(23,24)25/h2-7,10,29H,8-9H2,1H3,(H,27,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medifron DBT Inc.
US Patent
| Assay Description
The FLIPR protocol consists of 2 substance additions during a kinetic measurement. First the compounds to be tested (5 μM) are pipetted onto the... |
US Patent US9771359 (2017)
BindingDB Entry DOI: 10.7270/Q2CZ3984 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50101088
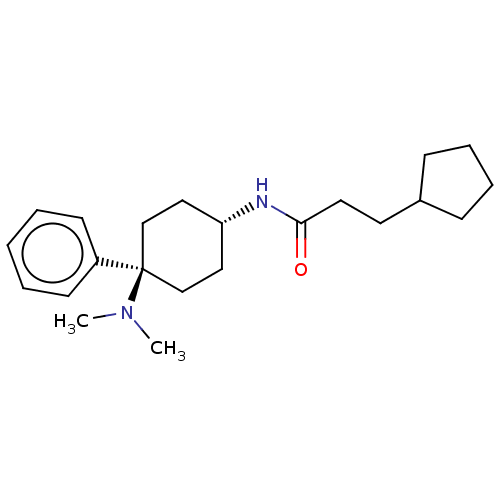
(CHEMBL3326220)Show SMILES Cl.CN(C)[C@]1(CC[C@@H](CC1)NC(=O)CCC1CCCC1)c1ccccc1 |r,wU:4.2,wD:7.9,(15.72,-29.86,;12.69,-28.26,;11.36,-27.5,;11.36,-25.96,;10.03,-28.27,;9.26,-29.6,;7.72,-29.6,;6.95,-28.28,;7.71,-26.95,;9.25,-26.94,;5.41,-28.28,;4.63,-26.95,;5.4,-25.61,;3.1,-26.95,;2.33,-25.61,;.79,-25.61,;-.11,-24.36,;-1.58,-24.83,;-1.58,-26.37,;-.12,-26.85,;10.82,-29.59,;10.08,-30.93,;10.87,-32.25,;12.41,-32.22,;13.16,-30.86,;12.36,-29.55,)| Show InChI InChI=1S/C22H34N2O/c1-24(2)22(19-10-4-3-5-11-19)16-14-20(15-17-22)23-21(25)13-12-18-8-6-7-9-18/h3-5,10-11,18,20H,6-9,12-17H2,1-2H3,(H,23,25)/t20-,22- | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmacokinetics
Curated by ChEMBL
| Assay Description
Displacement of [3H]Naloxone from human mu opioid receptor receptor expressed in CHO-K1 cells after 90 mins |
ACS Med Chem Lett 5: 851-6 (2014)
Article DOI: 10.1021/ml500116x
BindingDB Entry DOI: 10.7270/Q28S4RPM |
More data for this
Ligand-Target Pair | |
Nociceptin receptor
(Homo sapiens (Human)) | BDBM50100983

(CHEMBL3326224)Show SMILES Cl.CN(C)[C@]1(CC[C@@]2(CC1)OCCc1c2[nH]c2ccccc12)c1ccccc1 |r,wU:4.2,wD:7.9,(22.71,-7.97,;18.3,-7.93,;16.96,-7.16,;16.96,-5.62,;15.63,-7.93,;14.86,-9.26,;13.32,-9.27,;12.55,-7.94,;13.32,-6.61,;14.86,-6.6,;11.79,-9.28,;10.25,-9.27,;9.47,-7.94,;10.25,-6.6,;11.79,-6.61,;12.26,-5.14,;11.03,-4.24,;10.87,-2.7,;9.47,-2.07,;8.21,-2.98,;8.37,-4.51,;9.77,-5.14,;16.43,-9.25,;15.68,-10.6,;16.48,-11.91,;18.02,-11.88,;18.76,-10.52,;17.96,-9.21,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmacokinetics
Curated by ChEMBL
| Assay Description
Displacement of [3H]nociceptin from human NOP receptor expressed in CHO-K1 cells after 90 mins |
ACS Med Chem Lett 5: 851-6 (2014)
Article DOI: 10.1021/ml500116x
BindingDB Entry DOI: 10.7270/Q28S4RPM |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50049553

(CHEMBL2177428)Show SMILES CC(C(=O)NCc1ccc(nc1N1CCC(C)CC1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 Show InChI InChI=1S/C23H28F4N4O3S/c1-14-8-10-31(11-9-14)21-17(5-7-20(29-21)23(25,26)27)13-28-22(32)15(2)16-4-6-19(18(24)12-16)30-35(3,33)34/h4-7,12,14-15,30H,8-11,13H2,1-3H3,(H,28,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity against human TRPV1 expressed in CHO cells assessed as inhibition of capsaicin-induced channel activation by FLIPR assay |
Bioorg Med Chem Lett 24: 4044-7 (2014)
Article DOI: 10.1016/j.bmcl.2014.05.072
BindingDB Entry DOI: 10.7270/Q2JH3NTH |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50073160

(CHEMBL3407762)Show SMILES CCCCOc1nc(ccc1CNC(=O)C(C)c1ccc(NS(C)(=O)=O)c(F)c1)C(F)(F)Cl Show InChI InChI=1S/C21H25ClF3N3O4S/c1-4-5-10-32-20-15(7-9-18(27-20)21(22,24)25)12-26-19(29)13(2)14-6-8-17(16(23)11-14)28-33(3,30)31/h6-9,11,13,28H,4-5,10,12H2,1-3H3,(H,26,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of capsaicin-induced activity by FLIPR assay |
Eur J Med Chem 93: 101-8 (2015)
Article DOI: 10.1016/j.ejmech.2015.02.001
BindingDB Entry DOI: 10.7270/Q2N0188S |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50049553

(CHEMBL2177428)Show SMILES CC(C(=O)NCc1ccc(nc1N1CCC(C)CC1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 Show InChI InChI=1S/C23H28F4N4O3S/c1-14-8-10-31(11-9-14)21-17(5-7-20(29-21)23(25,26)27)13-28-22(32)15(2)16-4-6-19(18(24)12-16)30-35(3,33)34/h4-7,12,14-15,30H,8-11,13H2,1-3H3,(H,28,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of capsaicin-induced activity by FLIPR assay |
Eur J Med Chem 93: 101-8 (2015)
Article DOI: 10.1016/j.ejmech.2015.02.001
BindingDB Entry DOI: 10.7270/Q2N0188S |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50049547
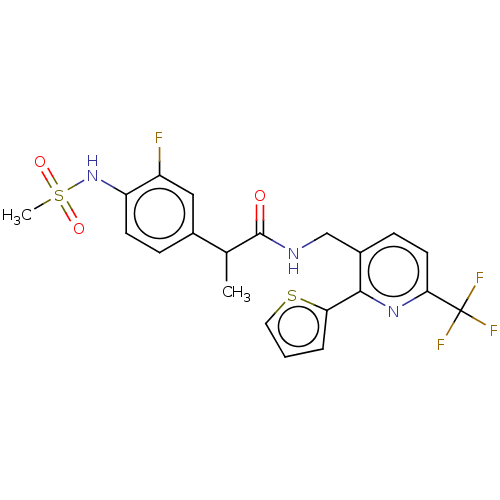
(CHEMBL3317484)Show SMILES CC(C(=O)NCc1ccc(nc1-c1cccs1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 Show InChI InChI=1S/C21H19F4N3O3S2/c1-12(13-5-7-16(15(22)10-13)28-33(2,30)31)20(29)26-11-14-6-8-18(21(23,24)25)27-19(14)17-4-3-9-32-17/h3-10,12,28H,11H2,1-2H3,(H,26,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity against human TRPV1 expressed in CHO cells assessed as inhibition of capsaicin-induced channel activation by FLIPR assay |
Bioorg Med Chem Lett 24: 4044-7 (2014)
Article DOI: 10.1016/j.bmcl.2014.05.072
BindingDB Entry DOI: 10.7270/Q2JH3NTH |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Rattus norvegicus (rat)) | BDBM176549

(US9120756, 7)Show SMILES CC(C(=O)NCc1cc(nn1C1CCCCC1)C(C)(C)C)c1ccc(NS(C)(=O)=O)c(F)c1 Show InChI InChI=1S/C24H35FN4O3S/c1-16(17-11-12-21(20(25)13-17)28-33(5,31)32)23(30)26-15-19-14-22(24(2,3)4)27-29(19)18-9-7-6-8-10-18/h11-14,16,18,28H,6-10,15H2,1-5H3,(H,26,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 0.350 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Gruenenthal GmbH
US Patent
| Assay Description
The agonistic or antagonistic effect of the substances to be tested on the rat-species vanilloid receptor 1 (VR1/TRPV1) can be determined using the f... |
US Patent US9120756 (2015)
BindingDB Entry DOI: 10.7270/Q26D5RRG |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data