

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14863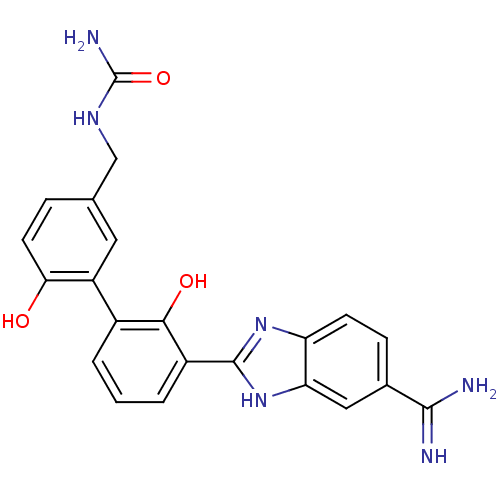 (({3-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-2...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 13 | -44.6 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tryptase delta (Homo sapiens (Human)) | BDBM50475863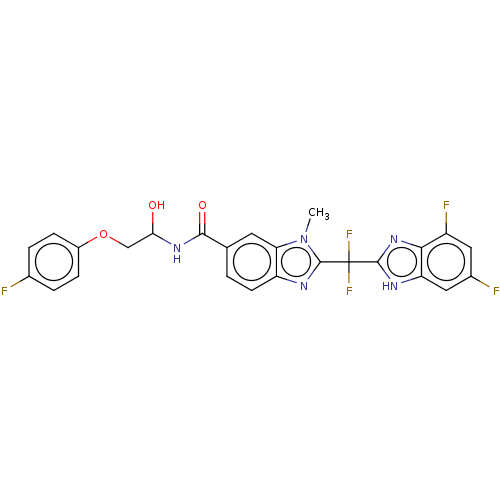 (CHEMBL209054) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 24 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics Curated by ChEMBL | Assay Description Inhibition of mast cell tryptase | Bioorg Med Chem Lett 16: 4053-8 (2006) Article DOI: 10.1016/j.bmcl.2006.05.003 BindingDB Entry DOI: 10.7270/Q26H4M69 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14880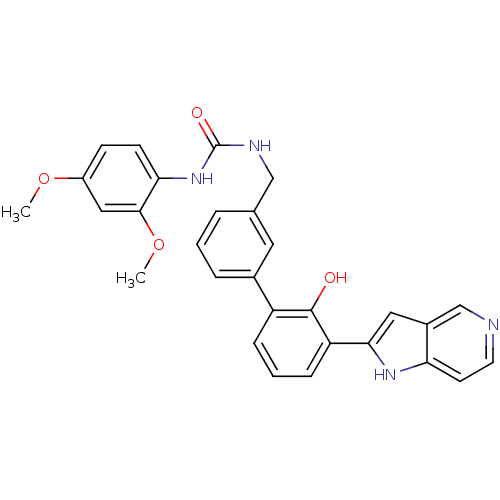 (1-(2,4-dimethoxyphenyl)-3-{[3-(2-hydroxy-3-{1H-pyr...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 28 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14872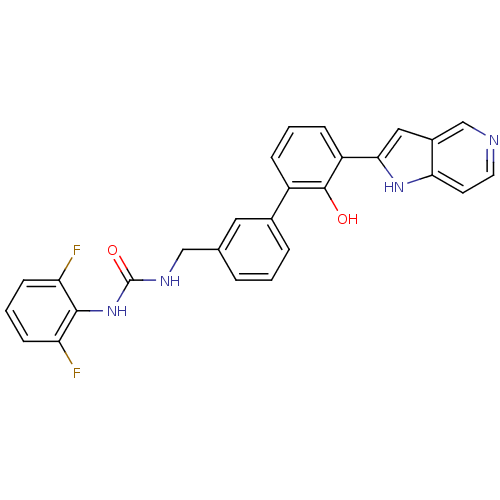 (1-(2,6-difluorophenyl)-3-{[3-(2-hydroxy-3-{1H-pyrr...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 33 | -42.3 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tryptase delta (Homo sapiens (Human)) | BDBM50475860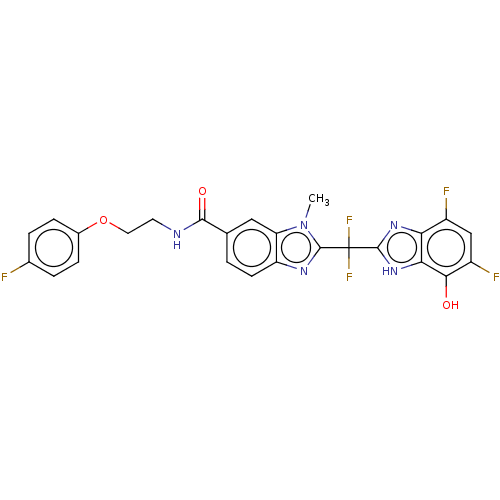 (CHEMBL424699) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 34 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics Curated by ChEMBL | Assay Description Inhibition of mast cell tryptase | Bioorg Med Chem Lett 16: 4053-8 (2006) Article DOI: 10.1016/j.bmcl.2006.05.003 BindingDB Entry DOI: 10.7270/Q26H4M69 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14877 (3-{[3-(2-hydroxy-3-{1H-pyrrolo[3,2-c]pyridin-2-yl}...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 47 | -41.4 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14869 (1-(2-fluorophenyl)-3-{[3-(2-hydroxy-3-{1H-pyrrolo[...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 57 | -40.9 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14876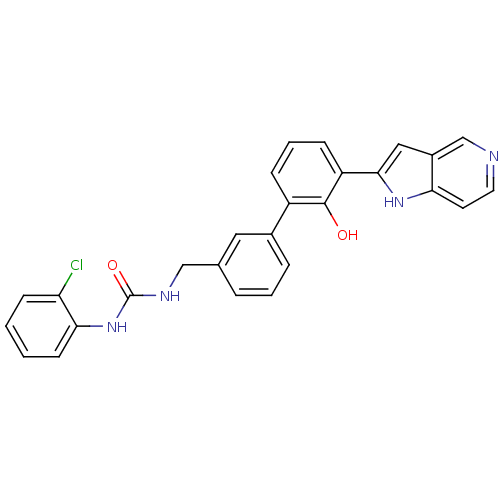 (1-(2-chlorophenyl)-3-{[3-(2-hydroxy-3-{1H-pyrrolo[...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 57 | -40.9 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14883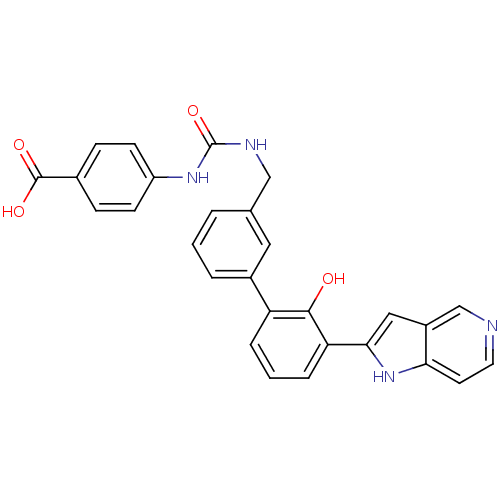 (4-[({[3-(2-hydroxy-3-{1H-pyrrolo[3,2-c]pyridin-2-y...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 58 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14881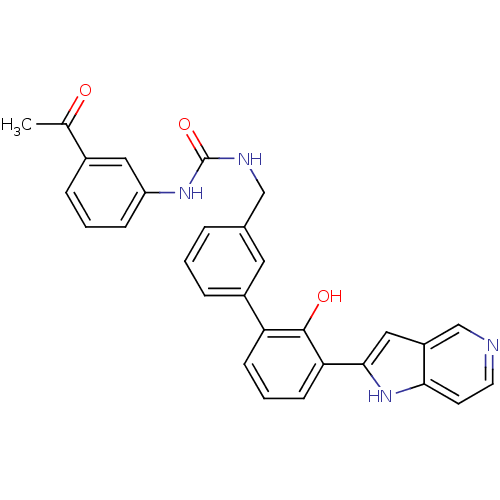 (1-(3-acetylphenyl)-3-{[3-(2-hydroxy-3-{1H-pyrrolo[...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14868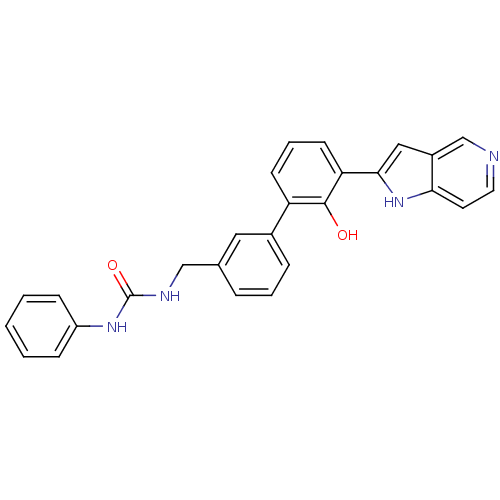 (3-{[3-(2-hydroxy-3-{1H-pyrrolo[3,2-c]pyridin-2-yl}...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 61.5 | -40.7 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14896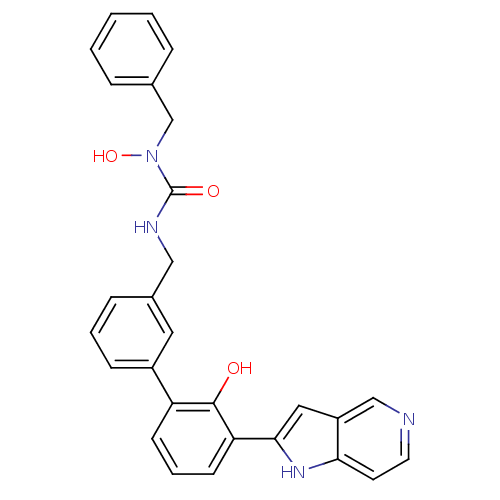 (1-benzyl-1-hydroxy-3-{[3-(2-hydroxy-3-{1H-pyrrolo[...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 72 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14879 (3-{[3-(2-hydroxy-3-{1H-pyrrolo[3,2-c]pyridin-2-yl}...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 88 | -39.9 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14873 (1-(2,4-difluorophenyl)-3-{[3-(2-hydroxy-3-{1H-pyrr...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 100 | -39.6 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14878 (3-{[3-(2-hydroxy-3-{1H-pyrrolo[3,2-c]pyridin-2-yl}...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 100 | -39.6 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14871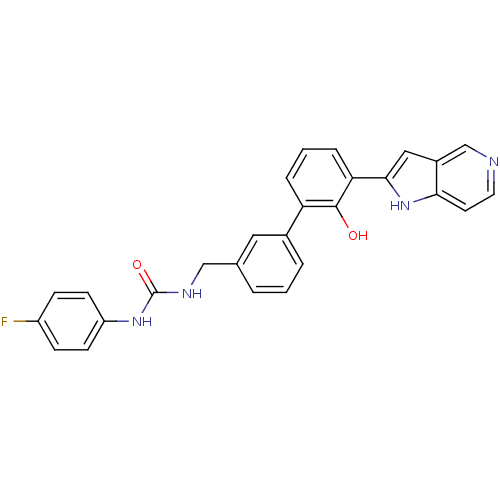 (1-(4-fluorophenyl)-3-{[3-(2-hydroxy-3-{1H-pyrrolo[...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 110 | -39.3 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14885 (1-{[3-(2-hydroxy-3-{1H-pyrrolo[3,2-c]pyridin-2-yl}...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14870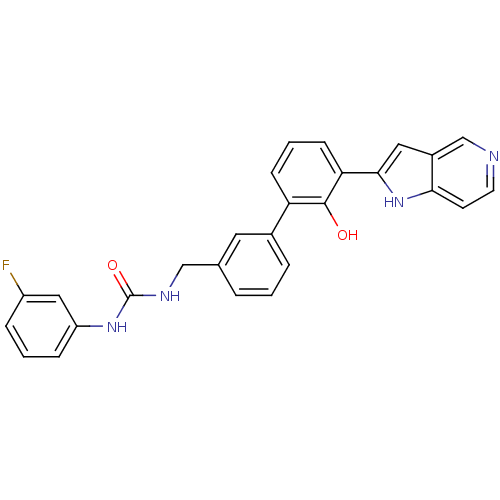 (1-(3-fluorophenyl)-3-{[3-(2-hydroxy-3-{1H-pyrrolo[...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 130 | -38.9 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14887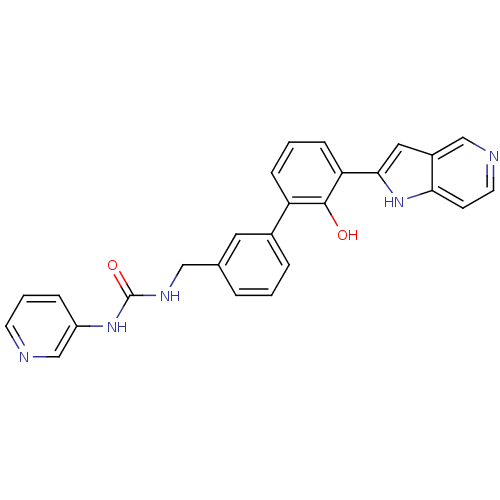 (3-{[3-(2-hydroxy-3-{1H-pyrrolo[3,2-c]pyridin-2-yl}...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 155 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14884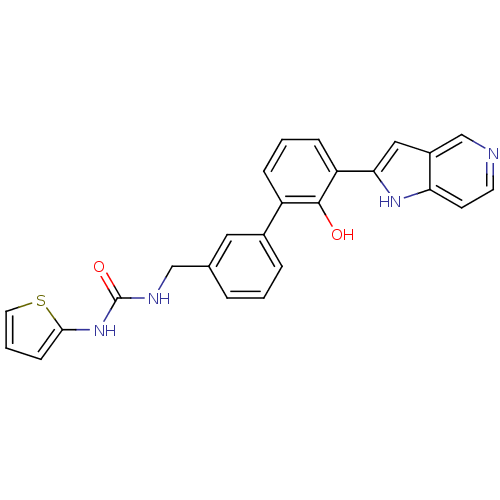 (1-{[3-(2-hydroxy-3-{1H-pyrrolo[3,2-c]pyridin-2-yl}...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14882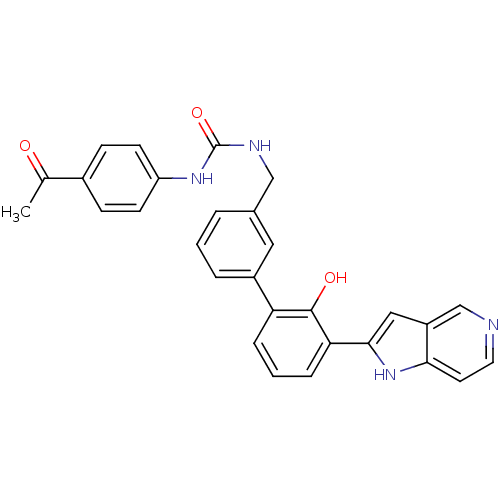 (1-(4-acetylphenyl)-3-{[3-(2-hydroxy-3-{1H-pyrrolo[...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14892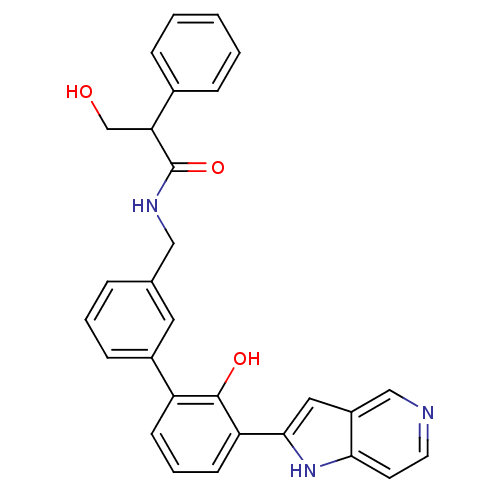 (3-hydroxy-N-{[3-(2-hydroxy-3-{1H-pyrrolo[3,2-c]pyr...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 220 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14866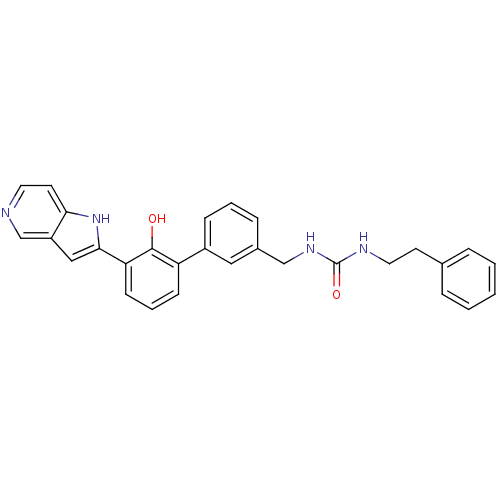 (3-{[3-(2-hydroxy-3-{1H-pyrrolo[3,2-c]pyridin-2-yl}...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 270 | -37.1 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tryptase delta (Homo sapiens (Human)) | BDBM50475861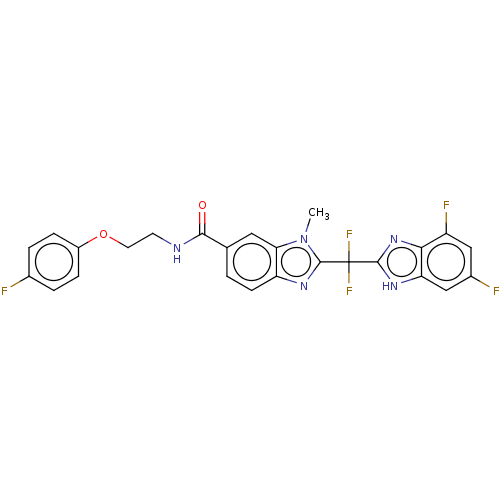 (CHEMBL211733) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics Curated by ChEMBL | Assay Description Inhibition of mast cell tryptase | Bioorg Med Chem Lett 16: 4053-8 (2006) Article DOI: 10.1016/j.bmcl.2006.05.003 BindingDB Entry DOI: 10.7270/Q26H4M69 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14874 (1-(3,4-difluorophenyl)-3-{[3-(2-hydroxy-3-{1H-pyrr...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 330 | -36.6 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14865 (1-{[3-(2-hydroxy-3-{1H-pyrrolo[3,2-c]pyridin-2-yl}...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 335 | -36.6 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14867 (1-{[3-(2-hydroxy-3-{1H-pyrrolo[3,2-c]pyridin-2-yl}...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 350 | -36.5 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14893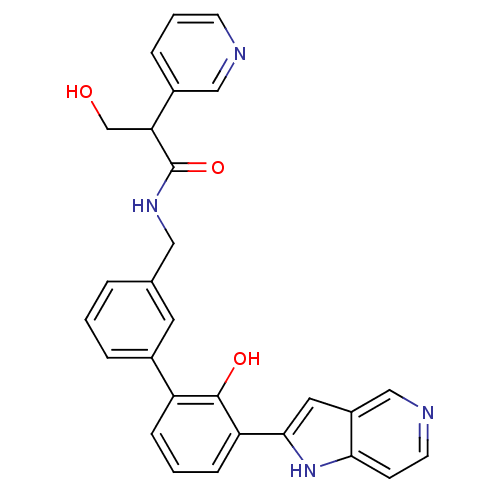 (3-hydroxy-N-{[3-(2-hydroxy-3-{1H-pyrrolo[3,2-c]pyr...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 610 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14864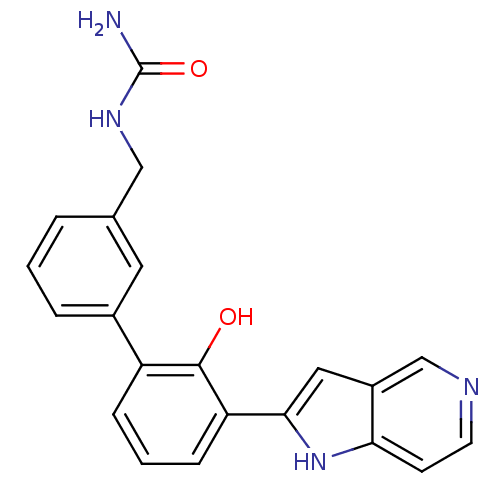 (5-azaindole analog 2 | {[3-(2-hydroxy-3-{1H-pyrrol...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | DrugBank MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | 800 | -34.4 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14875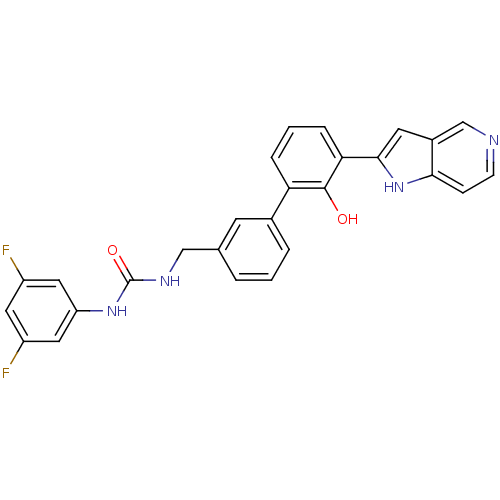 (1-(3,5-difluorophenyl)-3-{[3-(2-hydroxy-3-{1H-pyrr...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 830 | -34.4 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14894 (2-hydroxy-N-{[3-(2-hydroxy-3-{1H-pyrrolo[3,2-c]pyr...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 850 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tryptase delta (Homo sapiens (Human)) | BDBM50475865 (CHEMBL212765) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics Curated by ChEMBL | Assay Description Inhibition of mast cell tryptase | Bioorg Med Chem Lett 16: 4053-8 (2006) Article DOI: 10.1016/j.bmcl.2006.05.003 BindingDB Entry DOI: 10.7270/Q26H4M69 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14890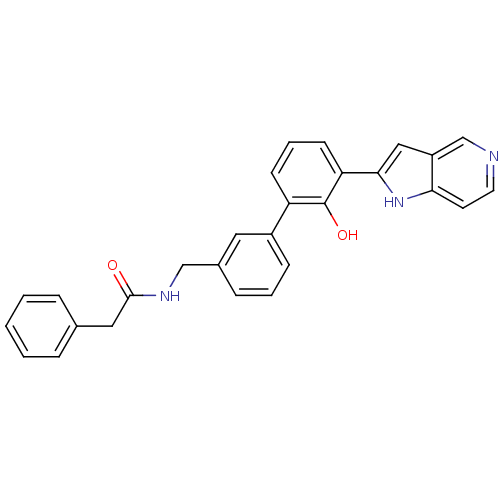 (5-azaindole analog 28 | N-{[3-(2-hydroxy-3-{1H-pyr...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14886 (3-{[3-(2-hydroxy-3-{1H-pyrrolo[3,2-c]pyridin-2-yl}...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tryptase delta (Homo sapiens (Human)) | BDBM50475864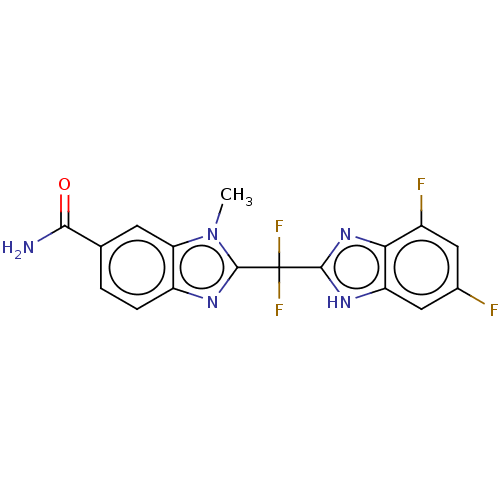 (CHEMBL378085) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics Curated by ChEMBL | Assay Description Inhibition of mast cell tryptase | Bioorg Med Chem Lett 16: 4053-8 (2006) Article DOI: 10.1016/j.bmcl.2006.05.003 BindingDB Entry DOI: 10.7270/Q26H4M69 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM14863 (({3-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-2...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 2.70E+3 | -31.5 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14895 (5-azaindole analog 33 | N-{[3-(2-hydroxy-3-{1H-pyr...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 2.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine protease 1 (Bos taurus (bovine)) | BDBM14863 (({3-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-2...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 3.60E+3 | -30.8 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tryptase delta (Homo sapiens (Human)) | BDBM50475862 (CHEMBL379076) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | KEGG PC cid PC sid UniChem | Article PubMed | 7.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics Curated by ChEMBL | Assay Description Inhibition of mast cell tryptase | Bioorg Med Chem Lett 16: 4053-8 (2006) Article DOI: 10.1016/j.bmcl.2006.05.003 BindingDB Entry DOI: 10.7270/Q26H4M69 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14891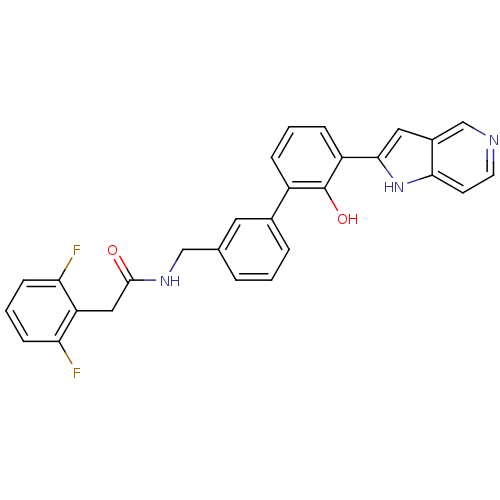 (2-(2,6-difluorophenyl)-N-{[3-(2-hydroxy-3-{1H-pyrr...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 9.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14888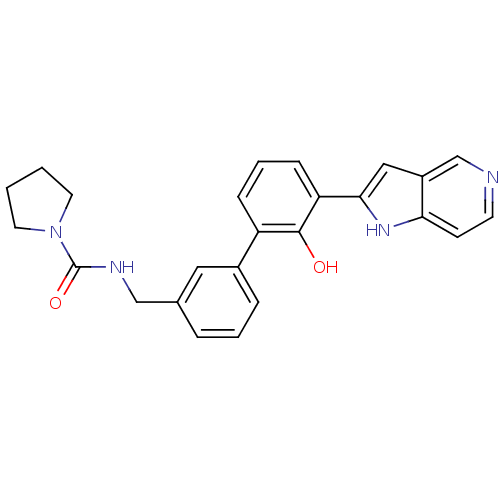 (5-azaindole analog 26 | N-{[3-(2-hydroxy-3-{1H-pyr...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 1.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine protease 1 (Bos taurus (bovine)) | BDBM14864 (5-azaindole analog 2 | {[3-(2-hydroxy-3-{1H-pyrrol...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | DrugBank MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | 4.65E+4 | -24.5 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM14864 (5-azaindole analog 2 | {[3-(2-hydroxy-3-{1H-pyrrol...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | DrugBank MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | 7.15E+4 | -23.4 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM14863 (({3-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-2...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 9.00E+4 | -22.9 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM14864 (5-azaindole analog 2 | {[3-(2-hydroxy-3-{1H-pyrrol...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | DrugBank MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | 1.25E+5 | -22.1 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII (Homo sapiens (Human)) | BDBM14889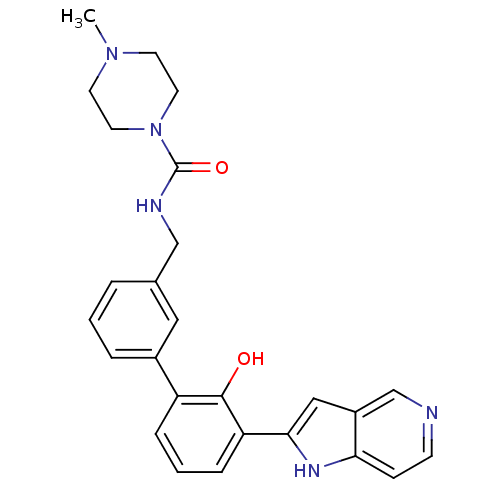 (5-azaindole analog 27 | N-{[3-(2-hydroxy-3-{1H-pyr...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 1.30E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | Bioorg Med Chem Lett 16: 3197-200 (2006) Article DOI: 10.1016/j.bmcl.2006.03.049 BindingDB Entry DOI: 10.7270/Q20P0X9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Receptor-type tyrosine-protein kinase FLT3 (Homo sapiens (Human)) | BDBM50463712 (CHEMBL4247248) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 5.60 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Citation and Details Article DOI: 10.1016/j.bmc.2022.116937 BindingDB Entry DOI: 10.7270/Q27S7SRR | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Receptor-type tyrosine-protein kinase FLT3 (Homo sapiens (Human)) | BDBM21079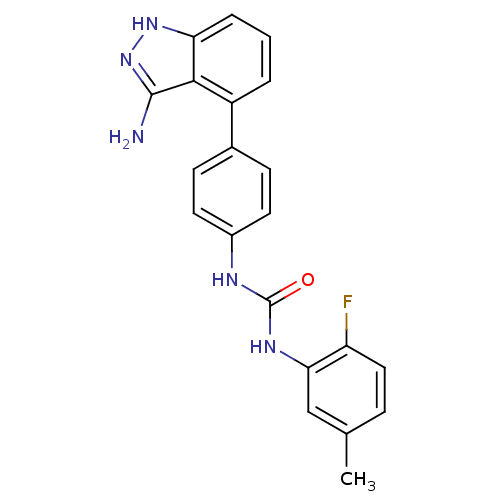 (1-[4-(3-amino-1H-indazol-4-yl)phenyl]-3-(2-fluoro-...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Citation and Details Article DOI: 10.1016/j.bmc.2022.116937 BindingDB Entry DOI: 10.7270/Q27S7SRR | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Presenilin-1 (Homo sapiens (Human)) | BDBM50483944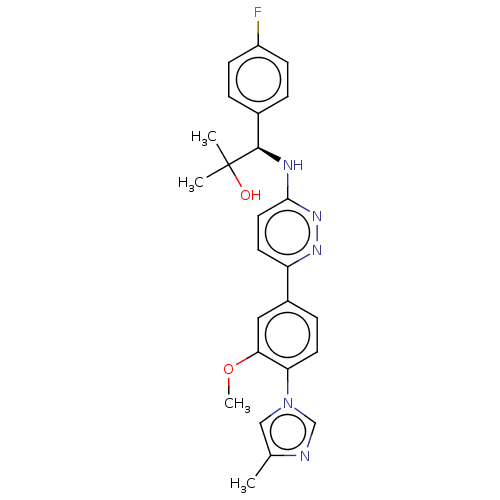 (CHEMBL1797105) | PDB MMDB UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Pharmaceuticals Curated by ChEMBL | Assay Description Inhibition of gamma-secretase in human SHSY5Y cells expressing Swedish variant of APP K595N/M596L assessed as reduction of amyloid beta 42 formation ... | Bioorg Med Chem Lett 21: 4016-9 (2011) Article DOI: 10.1016/j.bmcl.2011.04.143 BindingDB Entry DOI: 10.7270/Q2VH5RPH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Presenilin-1 (Homo sapiens (Human)) | BDBM50483946 (CHEMBL1797111) | PDB MMDB UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Pharmaceuticals Curated by ChEMBL | Assay Description Inhibition of gamma-secretase in human SHSY5Y cells expressing Swedish variant of APP K595N/M596L assessed as reduction of amyloid beta 42 formation ... | Bioorg Med Chem Lett 21: 4016-9 (2011) Article DOI: 10.1016/j.bmcl.2011.04.143 BindingDB Entry DOI: 10.7270/Q2VH5RPH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 129 total ) | Next | Last >> |