

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Neutrophil elastase (Homo sapiens (Human)) | BDBM50425968 (CHEMBL2314388) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem | Article PubMed | n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human neutrophil elastase using S1384 as substrate preincubated for 5 mins followed by substrate addition | J Med Chem 62: 5501-5511 (2019) Article DOI: 10.1021/acs.jmedchem.9b00379 BindingDB Entry DOI: 10.7270/Q2DR2ZR4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil elastase (Homo sapiens (Human)) | BDBM50501886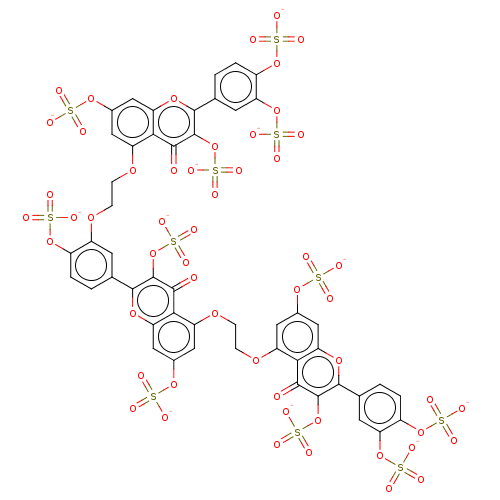 (CHEMBL4475009) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human neutrophil elastase using S1384 as substrate preincubated for 5 mins followed by substrate addition | J Med Chem 62: 5501-5511 (2019) Article DOI: 10.1021/acs.jmedchem.9b00379 BindingDB Entry DOI: 10.7270/Q2DR2ZR4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil elastase (Homo sapiens (Human)) | BDBM50260048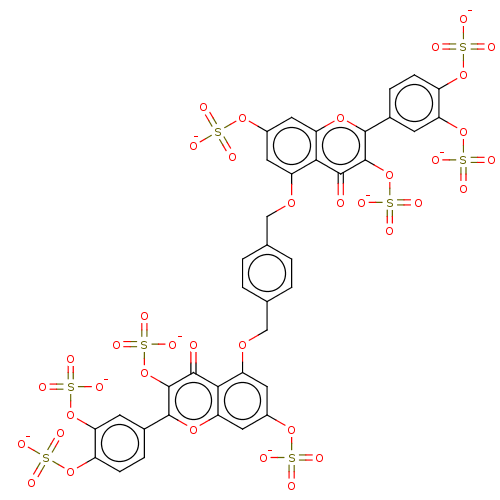 (CHEMBL4084933) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 230 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human neutrophil elastase using S1384 as substrate preincubated for 5 mins followed by substrate addition | J Med Chem 62: 5501-5511 (2019) Article DOI: 10.1021/acs.jmedchem.9b00379 BindingDB Entry DOI: 10.7270/Q2DR2ZR4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil elastase (Homo sapiens (Human)) | BDBM50501891 (CHEMBL4538120) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 250 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human neutrophil elastase using S1384 as substrate preincubated for 5 mins followed by substrate addition | J Med Chem 62: 5501-5511 (2019) Article DOI: 10.1021/acs.jmedchem.9b00379 BindingDB Entry DOI: 10.7270/Q2DR2ZR4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil elastase (Homo sapiens (Human)) | BDBM50260045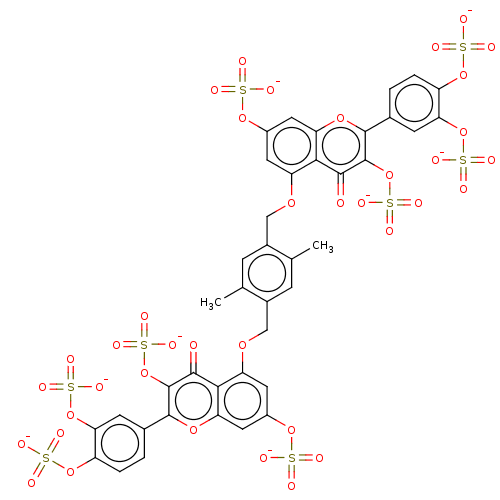 (CHEMBL4103815) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 320 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human neutrophil elastase using S1384 as substrate preincubated for 5 mins followed by substrate addition | J Med Chem 62: 5501-5511 (2019) Article DOI: 10.1021/acs.jmedchem.9b00379 BindingDB Entry DOI: 10.7270/Q2DR2ZR4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil elastase (Homo sapiens (Human)) | BDBM50260052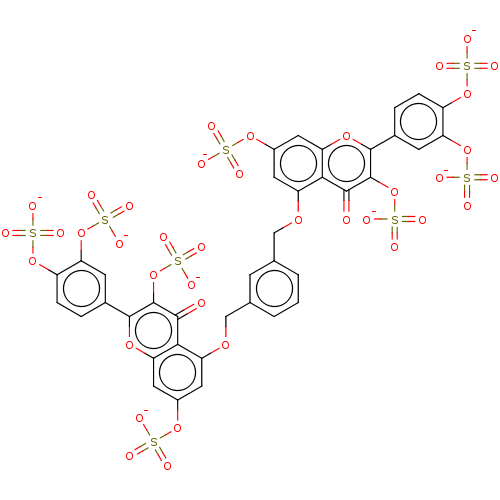 (CHEMBL4079552) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 640 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human neutrophil elastase using S1384 as substrate preincubated for 5 mins followed by substrate addition | J Med Chem 62: 5501-5511 (2019) Article DOI: 10.1021/acs.jmedchem.9b00379 BindingDB Entry DOI: 10.7270/Q2DR2ZR4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil elastase (Homo sapiens (Human)) | BDBM50260049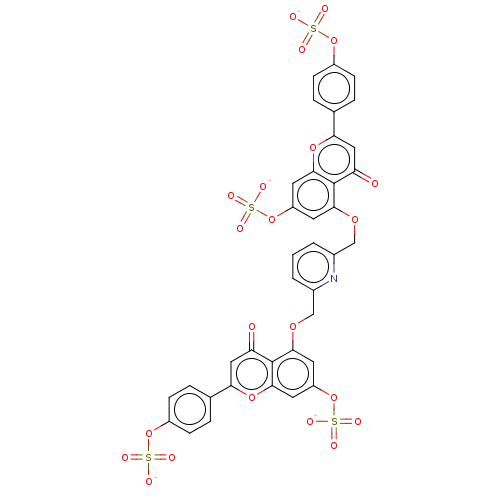 (CHEMBL4104474) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 690 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human neutrophil elastase using S1384 as substrate preincubated for 5 mins followed by substrate addition | J Med Chem 62: 5501-5511 (2019) Article DOI: 10.1021/acs.jmedchem.9b00379 BindingDB Entry DOI: 10.7270/Q2DR2ZR4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil elastase (Homo sapiens (Human)) | BDBM50260053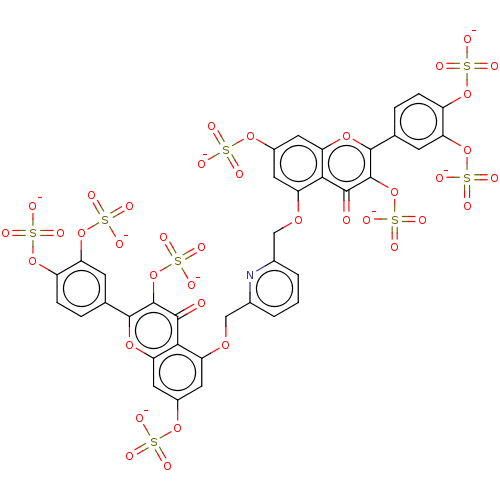 (CHEMBL4076224) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 810 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human neutrophil elastase using S1384 as substrate preincubated for 5 mins followed by substrate addition | J Med Chem 62: 5501-5511 (2019) Article DOI: 10.1021/acs.jmedchem.9b00379 BindingDB Entry DOI: 10.7270/Q2DR2ZR4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cathepsin G (Homo sapiens (Human)) | BDBM50260049 (CHEMBL4104474) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human leukocyte cathepsin G using S7388 as substrate preincubated for 5 mins followed by substrate addition | J Med Chem 62: 5501-5511 (2019) Article DOI: 10.1021/acs.jmedchem.9b00379 BindingDB Entry DOI: 10.7270/Q2DR2ZR4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455303 (CHEMBL4218956) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase KEGG PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Partial allosteric inhibition of human thrombin using S-23666 as substrate measured after 1 min by fibrometric method | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455284 (CHEMBL4210645) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition measured in absence of exosite 2... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455284 (CHEMBL4210645) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition measured in absence of exosite 1... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455284 (CHEMBL4210645) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition measured in presence of 30 uM ex... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil elastase (Homo sapiens (Human)) | BDBM50260058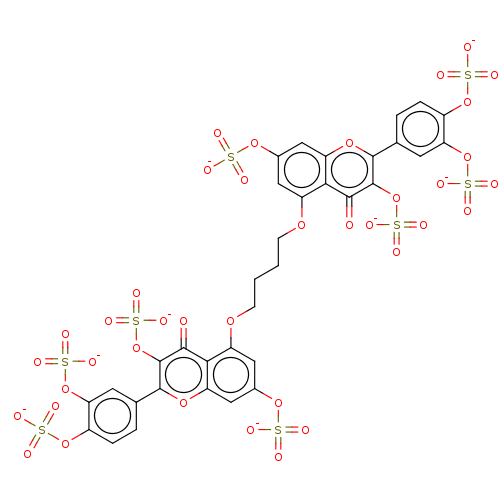 (CHEMBL4095129) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.56E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human neutrophil elastase using S1384 as substrate preincubated for 5 mins followed by substrate addition | J Med Chem 62: 5501-5511 (2019) Article DOI: 10.1021/acs.jmedchem.9b00379 BindingDB Entry DOI: 10.7270/Q2DR2ZR4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455284 (CHEMBL4210645) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition measured in presence of 15 uM ex... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455284 (CHEMBL4210645) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Partial allosteric inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition by spectropho... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455289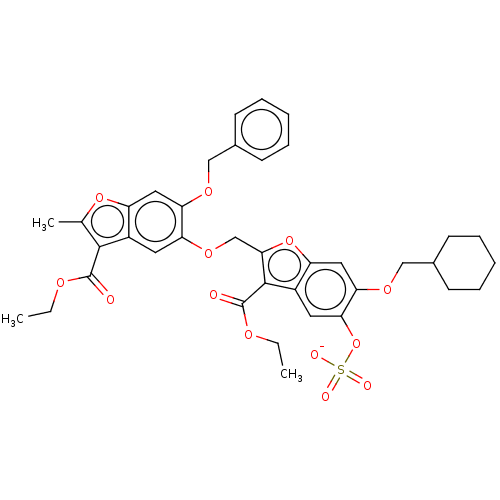 (CHEMBL4212514) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition measured in absence of exosite 1... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455289 (CHEMBL4212514) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition measured in absence of exosite 2... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455292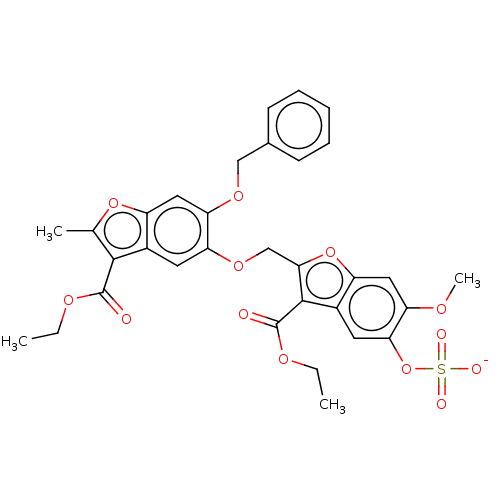 (CHEMBL4215598) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Partial allosteric inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition by spectropho... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil elastase (Homo sapiens (Human)) | BDBM50501887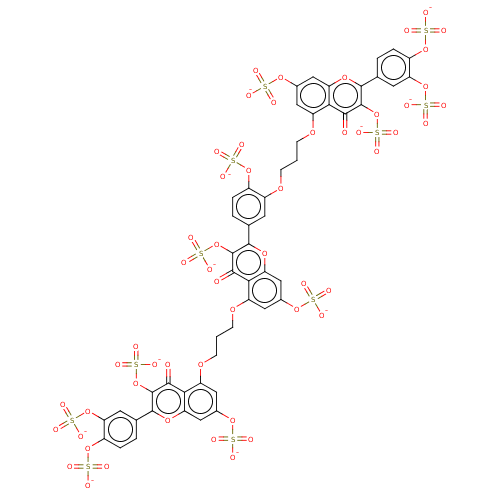 (CHEMBL4454043) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.19E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human neutrophil elastase using S1384 as substrate preincubated for 5 mins followed by substrate addition | J Med Chem 62: 5501-5511 (2019) Article DOI: 10.1021/acs.jmedchem.9b00379 BindingDB Entry DOI: 10.7270/Q2DR2ZR4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455293 (CHEMBL4208688) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Partial allosteric inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition by spectropho... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455291 (CHEMBL4214824) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Partial allosteric inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition by spectropho... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455294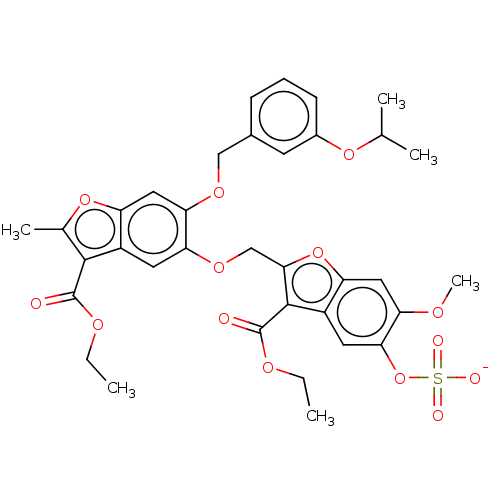 (CHEMBL4203810) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Partial allosteric inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition by spectropho... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455285 (CHEMBL4207062) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Partial allosteric inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition by spectropho... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455289 (CHEMBL4212514) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition measured in presence of 50 uM ex... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455289 (CHEMBL4212514) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition measured in presence of 5 uM exo... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil elastase (Homo sapiens (Human)) | BDBM50260051 (CHEMBL4102885) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.65E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human neutrophil elastase using S1384 as substrate preincubated for 5 mins followed by substrate addition | J Med Chem 62: 5501-5511 (2019) Article DOI: 10.1021/acs.jmedchem.9b00379 BindingDB Entry DOI: 10.7270/Q2DR2ZR4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455289 (CHEMBL4212514) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition measured in presence of 10 uM ex... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455284 (CHEMBL4210645) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition measured in presence of 5 uM exo... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Plasminogen (Homo sapiens (Human)) | BDBM50260048 (CHEMBL4084933) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Medicinal Chemistry, and Institute for Structural Biology, Drug Discovery and Development, Virginia Commonwealth University , Richmond, Virginia 23219, United States. Curated by ChEMBL | Assay Description Allosteric inhibition of human plasmin using chromogenic substrate spectrozyme PL preincubated for 5 mins followed by substrate addition measured ove... | J Med Chem 60: 641-657 (2017) Article DOI: 10.1021/acs.jmedchem.6b01474 BindingDB Entry DOI: 10.7270/Q2CF9SJM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455289 (CHEMBL4212514) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition measured in presence of 20 uM ex... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455289 (CHEMBL4212514) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition measured in presence of 30 uM ex... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455297 (CHEMBL4218791) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Partial allosteric inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition by spectropho... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil elastase (Homo sapiens (Human)) | BDBM50501893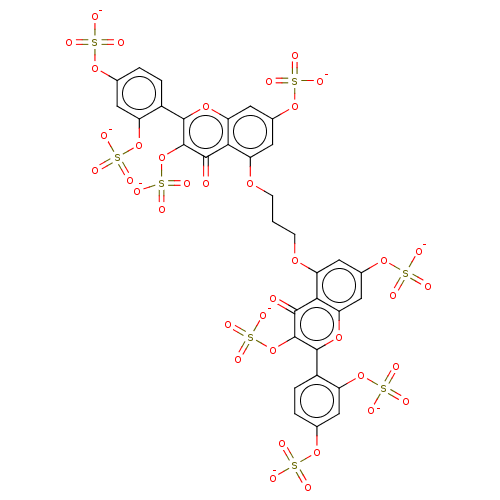 (CHEMBL4435942) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3.23E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human neutrophil elastase using S1384 as substrate preincubated for 5 mins followed by substrate addition | J Med Chem 62: 5501-5511 (2019) Article DOI: 10.1021/acs.jmedchem.9b00379 BindingDB Entry DOI: 10.7270/Q2DR2ZR4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455308 (CHEMBL4203401) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Partial allosteric inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition by spectropho... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455284 (CHEMBL4210645) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition measured in presence of 10 uM ex... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455284 (CHEMBL4210645) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition measured in presence of 20 uM ex... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455289 (CHEMBL4212514) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition measured in presence of 15 uM ex... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455289 (CHEMBL4212514) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Partial allosteric inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition by spectropho... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Plasminogen (Homo sapiens (Human)) | BDBM50260048 (CHEMBL4084933) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Medicinal Chemistry, and Institute for Structural Biology, Drug Discovery and Development, Virginia Commonwealth University , Richmond, Virginia 23219, United States. Curated by ChEMBL | Assay Description Allosteric inhibition of human plasmin using chromogenic substrate spectrozyme PL preincubated for 5 mins followed by substrate addition measured ove... | J Med Chem 60: 641-657 (2017) Article DOI: 10.1021/acs.jmedchem.6b01474 BindingDB Entry DOI: 10.7270/Q2CF9SJM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455295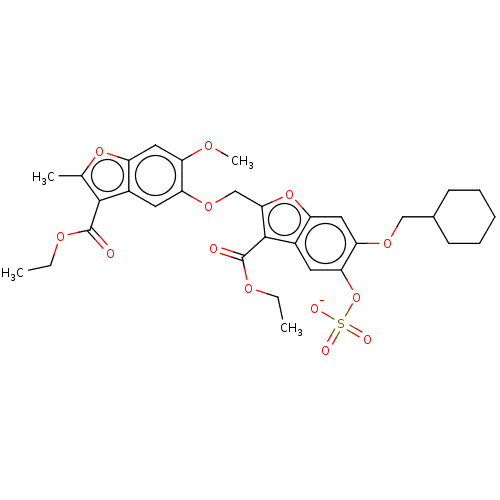 (CHEMBL4214371) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Partial allosteric inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition by spectropho... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455307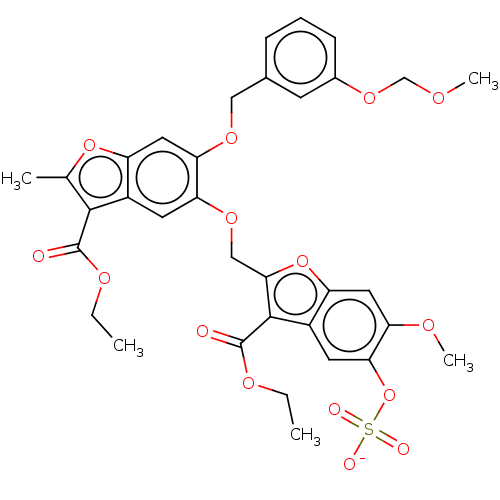 (CHEMBL4215073) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Partial allosteric inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition by spectropho... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Plasminogen (Homo sapiens (Human)) | BDBM50260048 (CHEMBL4084933) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Medicinal Chemistry, and Institute for Structural Biology, Drug Discovery and Development, Virginia Commonwealth University , Richmond, Virginia 23219, United States. Curated by ChEMBL | Assay Description Allosteric inhibition of human plasmin using chromogenic substrate spectrozyme PL preincubated for 5 mins followed by substrate addition measured ove... | J Med Chem 60: 641-657 (2017) Article DOI: 10.1021/acs.jmedchem.6b01474 BindingDB Entry DOI: 10.7270/Q2CF9SJM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50455296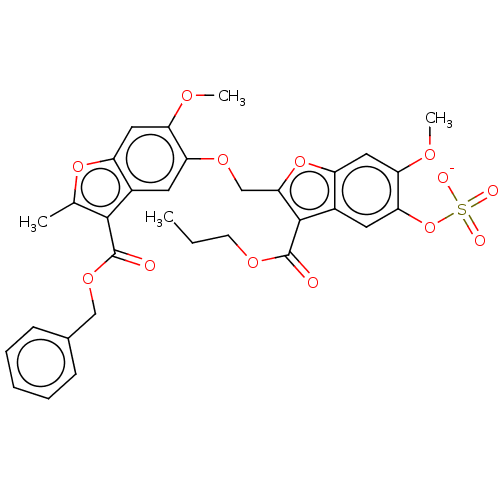 (CHEMBL4202710) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 4.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Partial allosteric inhibition of human thrombin using spectrozyme TH as substrate pretreated for 10 mins followed by substrate addition by spectropho... | Bioorg Med Chem Lett 28: 1101-1105 (2018) Article DOI: 10.1016/j.bmcl.2018.01.069 BindingDB Entry DOI: 10.7270/Q2SF2ZRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Plasminogen (Homo sapiens (Human)) | BDBM50260048 (CHEMBL4084933) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Medicinal Chemistry, and Institute for Structural Biology, Drug Discovery and Development, Virginia Commonwealth University , Richmond, Virginia 23219, United States. Curated by ChEMBL | Assay Description Allosteric inhibition of human plasmin using chromogenic substrate spectrozyme PL preincubated for 5 mins followed by substrate addition measured ove... | J Med Chem 60: 641-657 (2017) Article DOI: 10.1021/acs.jmedchem.6b01474 BindingDB Entry DOI: 10.7270/Q2CF9SJM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Plasminogen (Homo sapiens (Human)) | BDBM50260048 (CHEMBL4084933) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Medicinal Chemistry, and Institute for Structural Biology, Drug Discovery and Development, Virginia Commonwealth University , Richmond, Virginia 23219, United States. Curated by ChEMBL | Assay Description Allosteric inhibition of full length human plasmin by using chromogenic substrate spectrozyme PL preincubated for 5 mins followed by substrate additi... | J Med Chem 60: 641-657 (2017) Article DOI: 10.1021/acs.jmedchem.6b01474 BindingDB Entry DOI: 10.7270/Q2CF9SJM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil elastase (Homo sapiens (Human)) | BDBM50501890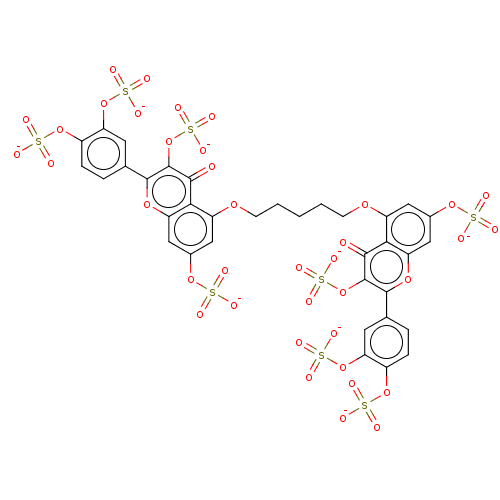 (CHEMBL4578588) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 6.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human neutrophil elastase using S1384 as substrate preincubated for 5 mins followed by substrate addition | J Med Chem 62: 5501-5511 (2019) Article DOI: 10.1021/acs.jmedchem.9b00379 BindingDB Entry DOI: 10.7270/Q2DR2ZR4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Plasminogen (Homo sapiens (Human)) | BDBM50260045 (CHEMBL4103815) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Medicinal Chemistry, and Institute for Structural Biology, Drug Discovery and Development, Virginia Commonwealth University , Richmond, Virginia 23219, United States. Curated by ChEMBL | Assay Description Allosteric inhibition of full length human plasmin by using chromogenic substrate spectrozyme PL preincubated for 5 mins followed by substrate additi... | J Med Chem 60: 641-657 (2017) Article DOI: 10.1021/acs.jmedchem.6b01474 BindingDB Entry DOI: 10.7270/Q2CF9SJM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil elastase (Homo sapiens (Human)) | BDBM50501894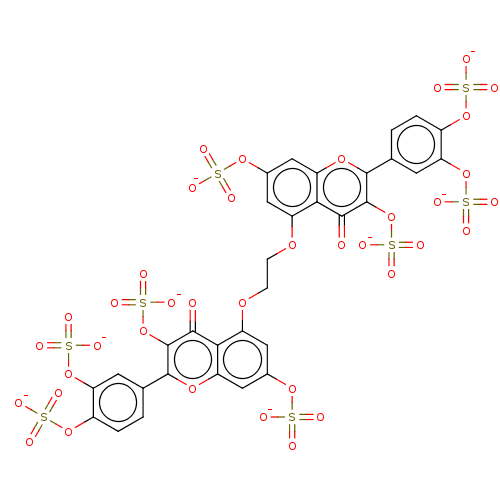 (CHEMBL4569385) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 7.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University Curated by ChEMBL | Assay Description Inhibition of human neutrophil elastase using S1384 as substrate preincubated for 5 mins followed by substrate addition | J Med Chem 62: 5501-5511 (2019) Article DOI: 10.1021/acs.jmedchem.9b00379 BindingDB Entry DOI: 10.7270/Q2DR2ZR4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor XI (Homo sapiens (Human)) | BDBM50612136 (CHEMBL5271926) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | UniChem | n/a | n/a | 8.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 144 total ) | Next | Last >> |