 Found 81 hits with Last Name = 'beyer' and Initial = 'ks'
Found 81 hits with Last Name = 'beyer' and Initial = 'ks' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50235302

(CHEMBL4099771)Show SMILES CN(CCCNC(=O)CNC(=O)Nc1ccc2sc(Cl)c(-c3cccnc3C)c2c1)[C@@H]1CCCN(C1)c1ncnc2[nH]ccc12 |r,wU:30.32,(40.26,-16.4,;40.26,-17.94,;41.59,-18.71,;42.93,-17.94,;44.26,-18.71,;45.59,-17.94,;46.93,-18.71,;46.93,-20.25,;48.26,-17.94,;49.59,-18.71,;50.93,-17.94,;50.93,-16.4,;52.26,-18.71,;53.6,-17.95,;53.59,-16.41,;54.92,-15.64,;56.26,-16.41,;57.73,-15.93,;58.64,-17.18,;60.18,-17.18,;57.73,-18.43,;58.21,-19.89,;57.17,-21.03,;57.65,-22.5,;59.16,-22.82,;60.19,-21.66,;59.71,-20.2,;60.74,-19.06,;56.26,-17.95,;54.93,-18.72,;38.93,-18.71,;37.59,-17.93,;36.26,-18.71,;36.26,-20.25,;37.59,-21.01,;38.93,-20.24,;37.59,-22.55,;38.93,-23.31,;38.93,-24.86,;37.6,-25.63,;36.26,-24.86,;34.8,-25.34,;33.89,-24.1,;34.79,-22.85,;36.26,-23.32,)| Show InChI InChI=1S/C32H36ClN9O2S/c1-20-23(7-3-11-34-20)28-25-16-21(8-9-26(25)45-29(28)33)40-32(44)37-17-27(43)35-12-5-14-41(2)22-6-4-15-42(18-22)31-24-10-13-36-30(24)38-19-39-31/h3,7-11,13,16,19,22H,4-6,12,14-15,17-18H2,1-2H3,(H,35,43)(H,36,38,39)(H2,37,40,44)/t22-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Competitive inhibition of DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosomes as substrate preincubated for 30 mins followed by... |
ACS Med Chem Lett 8: 338-343 (2017)
Article DOI: 10.1021/acsmedchemlett.6b00519
BindingDB Entry DOI: 10.7270/Q2WW7KZW |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50075098

(CHEMBL3414626 | US10143704, Compound A2 | US944606...)Show SMILES CC(C)N(C[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12)[C@@H]1C[C@H](CCc2nc3cc(ccc3[nH]2)C(C)(C)C)C1 |r,wU:24.27,22.24,10.11,8.8,wD:5.4,7.12,(-.55,4.43,;-1.02,5.57,;-2.24,5.73,;-.08,6.8,;1.45,6.59,;2.04,5.17,;1.23,3.86,;2.24,2.7,;3.65,3.27,;4.7,2.61,;3.54,4.8,;4.48,5.6,;1.76,1.24,;2.66,.02,;1.76,-1.24,;.3,-.77,;-1.03,-1.56,;-1.03,-2.79,;-2.38,-.77,;-2.38,.77,;-1.03,1.56,;.3,.77,;-.67,8.22,;-.05,9.59,;-1.48,10.19,;-2.06,11.61,;-1.11,12.83,;-1.7,14.26,;-.89,15.53,;-1.87,16.71,;-1.63,18.24,;-2.86,19.2,;-4.29,18.63,;-4.52,17.09,;-3.29,16.14,;-3.19,14.61,;-2.65,20.73,;-1.51,21.19,;-3.62,21.48,;-2.48,21.95,;-2.07,8.77,)| Show InChI InChI=1S/C30H42N8O3/c1-16(2)37(13-22-25(39)26(40)29(41-22)38-15-34-24-27(31)32-14-33-28(24)38)19-10-17(11-19)6-9-23-35-20-8-7-18(30(3,4)5)12-21(20)36-23/h7-8,12,14-17,19,22,25-26,29,39-40H,6,9-11,13H2,1-5H3,(H,35,36)(H2,31,32,33)/t17-,19+,22-,25-,26-,29-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.0120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Competitive inhibition of DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosomes as substrate preincubated for 30 mins followed by... |
ACS Med Chem Lett 8: 338-343 (2017)
Article DOI: 10.1021/acsmedchemlett.6b00519
BindingDB Entry DOI: 10.7270/Q2WW7KZW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50536826

(CHEMBL4590355)Show SMILES CNc1ccnc(Nc2ccc3cc(C)n(-c4ccccc4Oc4cnc5n(C)cnc5c4)c3c2)n1 |(29.26,-10.25,;27.93,-11.02,;27.94,-12.56,;29.28,-13.33,;29.28,-14.87,;27.95,-15.64,;26.62,-14.88,;25.29,-15.64,;23.96,-14.88,;23.95,-13.33,;22.62,-12.56,;21.29,-13.34,;19.82,-12.85,;18.9,-14.1,;17.36,-14.08,;19.8,-15.36,;19.31,-16.81,;20.33,-17.95,;19.85,-19.4,;18.34,-19.72,;17.32,-18.56,;17.81,-17.11,;16.79,-15.96,;15.29,-16.26,;14.81,-17.71,;13.31,-18.01,;12.29,-16.86,;10.74,-16.84,;9.83,-18.07,;10.28,-15.37,;11.54,-14.47,;12.78,-15.39,;14.28,-15.1,;21.28,-14.89,;22.62,-15.65,;26.61,-13.34,)| Show InChI InChI=1S/C27H24N8O/c1-17-12-18-8-9-19(32-27-29-11-10-25(28-2)33-27)13-23(18)35(17)22-6-4-5-7-24(22)36-20-14-21-26(30-15-20)34(3)16-31-21/h4-16H,1-3H3,(H2,28,29,32,33) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosomes as substrate preincubated for 30 mins followed by substrate a... |
ACS Med Chem Lett 7: 735-40 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00167
BindingDB Entry DOI: 10.7270/Q2V69P3G |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50536819
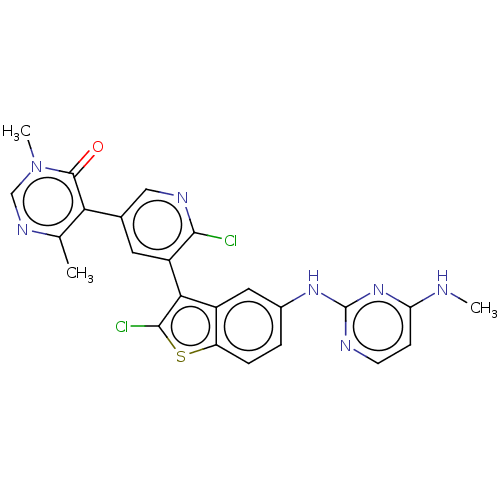
(CHEMBL4534250)Show SMILES CNc1ccnc(Nc2ccc3sc(Cl)c(-c4cc(cnc4Cl)-c4c(C)ncn(C)c4=O)c3c2)n1 |(34.66,-43.04,;33.33,-43.82,;33.34,-45.35,;34.68,-46.12,;34.68,-47.66,;33.35,-48.43,;32.02,-47.66,;30.69,-48.43,;29.36,-47.67,;29.35,-46.12,;28.02,-45.36,;26.69,-46.13,;25.23,-45.65,;24.32,-46.89,;22.78,-46.88,;25.22,-48.14,;24.74,-49.6,;23.23,-49.91,;22.75,-51.37,;23.77,-52.52,;25.29,-52.2,;25.76,-50.74,;27.27,-50.42,;21.24,-51.68,;20.76,-53.14,;21.78,-54.29,;19.25,-53.46,;18.22,-52.31,;18.71,-50.84,;17.68,-49.69,;20.22,-50.53,;20.7,-49.07,;26.69,-47.67,;28.02,-48.44,;32.01,-46.13,)| Show InChI InChI=1S/C24H19Cl2N7OS/c1-12-19(23(34)33(3)11-30-12)13-8-16(21(25)29-10-13)20-15-9-14(4-5-17(15)35-22(20)26)31-24-28-7-6-18(27-2)32-24/h4-11H,1-3H3,(H2,27,28,31,32) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| 0.360 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosomes as substrate preincubated for 30 mins followed by substrate a... |
ACS Med Chem Lett 7: 735-40 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00167
BindingDB Entry DOI: 10.7270/Q2V69P3G |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50075098

(CHEMBL3414626 | US10143704, Compound A2 | US944606...)Show SMILES CC(C)N(C[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12)[C@@H]1C[C@H](CCc2nc3cc(ccc3[nH]2)C(C)(C)C)C1 |r,wU:24.27,22.24,10.11,8.8,wD:5.4,7.12,(-.55,4.43,;-1.02,5.57,;-2.24,5.73,;-.08,6.8,;1.45,6.59,;2.04,5.17,;1.23,3.86,;2.24,2.7,;3.65,3.27,;4.7,2.61,;3.54,4.8,;4.48,5.6,;1.76,1.24,;2.66,.02,;1.76,-1.24,;.3,-.77,;-1.03,-1.56,;-1.03,-2.79,;-2.38,-.77,;-2.38,.77,;-1.03,1.56,;.3,.77,;-.67,8.22,;-.05,9.59,;-1.48,10.19,;-2.06,11.61,;-1.11,12.83,;-1.7,14.26,;-.89,15.53,;-1.87,16.71,;-1.63,18.24,;-2.86,19.2,;-4.29,18.63,;-4.52,17.09,;-3.29,16.14,;-3.19,14.61,;-2.65,20.73,;-1.51,21.19,;-3.62,21.48,;-2.48,21.95,;-2.07,8.77,)| Show InChI InChI=1S/C30H42N8O3/c1-16(2)37(13-22-25(39)26(40)29(41-22)38-15-34-24-27(31)32-14-33-28(24)38)19-10-17(11-19)6-9-23-35-20-8-7-18(30(3,4)5)12-21(20)36-23/h7-8,12,14-17,19,22,25-26,29,39-40H,6,9-11,13H2,1-5H3,(H,35,36)(H2,31,32,33)/t17-,19+,22-,25-,26-,29-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | <0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosomes as substrate preincubated for 30 mins followed by substrate a... |
ACS Med Chem Lett 8: 338-343 (2017)
Article DOI: 10.1021/acsmedchemlett.6b00519
BindingDB Entry DOI: 10.7270/Q2WW7KZW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50235302

(CHEMBL4099771)Show SMILES CN(CCCNC(=O)CNC(=O)Nc1ccc2sc(Cl)c(-c3cccnc3C)c2c1)[C@@H]1CCCN(C1)c1ncnc2[nH]ccc12 |r,wU:30.32,(40.26,-16.4,;40.26,-17.94,;41.59,-18.71,;42.93,-17.94,;44.26,-18.71,;45.59,-17.94,;46.93,-18.71,;46.93,-20.25,;48.26,-17.94,;49.59,-18.71,;50.93,-17.94,;50.93,-16.4,;52.26,-18.71,;53.6,-17.95,;53.59,-16.41,;54.92,-15.64,;56.26,-16.41,;57.73,-15.93,;58.64,-17.18,;60.18,-17.18,;57.73,-18.43,;58.21,-19.89,;57.17,-21.03,;57.65,-22.5,;59.16,-22.82,;60.19,-21.66,;59.71,-20.2,;60.74,-19.06,;56.26,-17.95,;54.93,-18.72,;38.93,-18.71,;37.59,-17.93,;36.26,-18.71,;36.26,-20.25,;37.59,-21.01,;38.93,-20.24,;37.59,-22.55,;38.93,-23.31,;38.93,-24.86,;37.6,-25.63,;36.26,-24.86,;34.8,-25.34,;33.89,-24.1,;34.79,-22.85,;36.26,-23.32,)| Show InChI InChI=1S/C32H36ClN9O2S/c1-20-23(7-3-11-34-20)28-25-16-21(8-9-26(25)45-29(28)33)40-32(44)37-17-27(43)35-12-5-14-41(2)22-6-4-15-42(18-22)31-24-10-13-36-30(24)38-19-39-31/h3,7-11,13,16,19,22H,4-6,12,14-15,17-18H2,1-2H3,(H,35,43)(H,36,38,39)(H2,37,40,44)/t22-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | <0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosomes as substrate preincubated for 30 mins followed by substrate a... |
ACS Med Chem Lett 8: 338-343 (2017)
Article DOI: 10.1021/acsmedchemlett.6b00519
BindingDB Entry DOI: 10.7270/Q2WW7KZW |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50529550
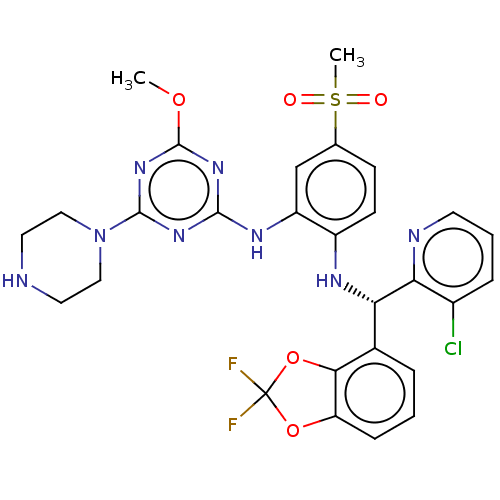
(CHEMBL4446126)Show SMILES COc1nc(Nc2cc(ccc2N[C@@H](c2cccc3OC(F)(F)Oc23)c2ncccc2Cl)S(C)(=O)=O)nc(n1)N1CCNCC1 |r| Show InChI InChI=1S/C28H27ClF2N8O5S/c1-42-27-37-25(36-26(38-27)39-13-11-32-12-14-39)35-20-15-16(45(2,40)41)8-9-19(20)34-22(23-18(29)6-4-10-33-23)17-5-3-7-21-24(17)44-28(30,31)43-21/h3-10,15,22,32,34H,11-14H2,1-2H3,(H,35,36,37,38)/t22-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 0.110 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of 0.05 nM DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosome as substrate and [3H]SAM as co-factor preincubated for... |
ACS Med Chem Lett 10: 1655-1660 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00452
BindingDB Entry DOI: 10.7270/Q2RV0S5T |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50235301
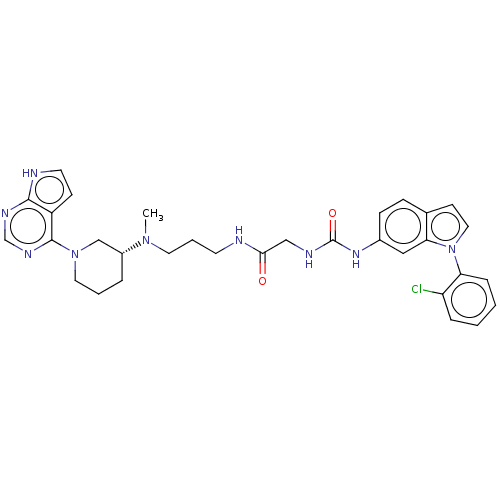
(CHEMBL4081752)Show SMILES CN(CCCNC(=O)CNC(=O)Nc1ccc2ccn(-c3ccccc3Cl)c2c1)[C@@H]1CCCN(C1)c1ncnc2[nH]ccc12 |r| Show InChI InChI=1S/C32H36ClN9O2/c1-40(24-6-4-16-41(20-24)31-25-11-14-35-30(25)37-21-38-31)15-5-13-34-29(43)19-36-32(44)39-23-10-9-22-12-17-42(28(22)18-23)27-8-3-2-7-26(27)33/h2-3,7-12,14,17-18,21,24H,4-6,13,15-16,19-20H2,1H3,(H,34,43)(H,35,37,38)(H2,36,39,44)/t24-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.150 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosomes as substrate preincubated for 30 mins followed by substrate a... |
ACS Med Chem Lett 8: 338-343 (2017)
Article DOI: 10.1021/acsmedchemlett.6b00519
BindingDB Entry DOI: 10.7270/Q2WW7KZW |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50529551
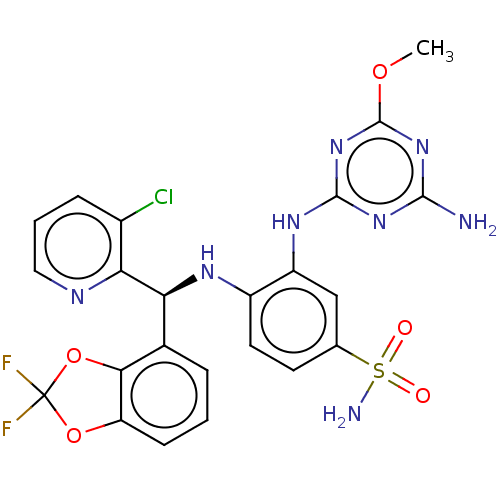
(CHEMBL4435508)Show SMILES COc1nc(N)nc(Nc2cc(ccc2N[C@@H](c2cccc3OC(F)(F)Oc23)c2ncccc2Cl)S(N)(=O)=O)n1 |r| Show InChI InChI=1S/C23H19ClF2N8O5S/c1-37-22-33-20(27)32-21(34-22)31-15-10-11(40(28,35)36)7-8-14(15)30-17(18-13(24)5-3-9-29-18)12-4-2-6-16-19(12)39-23(25,26)38-16/h2-10,17,30H,1H3,(H2,28,35,36)(H3,27,31,32,33,34)/t17-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 0.170 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of 0.05 nM DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosome as substrate and [3H]SAM as co-factor preincubated for... |
ACS Med Chem Lett 10: 1655-1660 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00452
BindingDB Entry DOI: 10.7270/Q2RV0S5T |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50529554
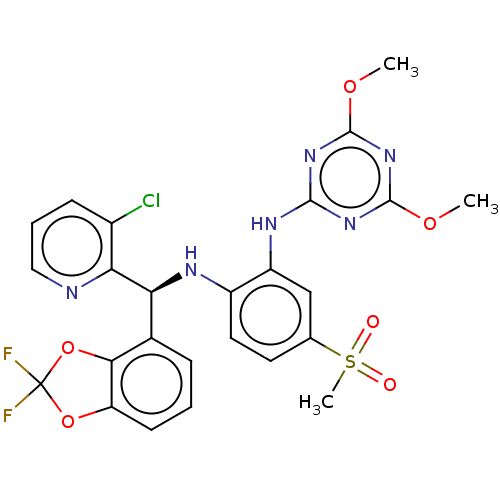
(CHEMBL4567485)Show SMILES COc1nc(Nc2cc(ccc2N[C@@H](c2cccc3OC(F)(F)Oc23)c2ncccc2Cl)S(C)(=O)=O)nc(OC)n1 |r| Show InChI InChI=1S/C25H21ClF2N6O6S/c1-37-23-32-22(33-24(34-23)38-2)31-17-12-13(41(3,35)36)9-10-16(17)30-19(20-15(26)7-5-11-29-20)14-6-4-8-18-21(14)40-25(27,28)39-18/h4-12,19,30H,1-3H3,(H,31,32,33,34)/t19-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.190 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of 0.05 nM DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosome as substrate and [3H]SAM as co-factor preincubated for... |
ACS Med Chem Lett 10: 1655-1660 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00452
BindingDB Entry DOI: 10.7270/Q2RV0S5T |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50536826

(CHEMBL4590355)Show SMILES CNc1ccnc(Nc2ccc3cc(C)n(-c4ccccc4Oc4cnc5n(C)cnc5c4)c3c2)n1 |(29.26,-10.25,;27.93,-11.02,;27.94,-12.56,;29.28,-13.33,;29.28,-14.87,;27.95,-15.64,;26.62,-14.88,;25.29,-15.64,;23.96,-14.88,;23.95,-13.33,;22.62,-12.56,;21.29,-13.34,;19.82,-12.85,;18.9,-14.1,;17.36,-14.08,;19.8,-15.36,;19.31,-16.81,;20.33,-17.95,;19.85,-19.4,;18.34,-19.72,;17.32,-18.56,;17.81,-17.11,;16.79,-15.96,;15.29,-16.26,;14.81,-17.71,;13.31,-18.01,;12.29,-16.86,;10.74,-16.84,;9.83,-18.07,;10.28,-15.37,;11.54,-14.47,;12.78,-15.39,;14.28,-15.1,;21.28,-14.89,;22.62,-15.65,;26.61,-13.34,)| Show InChI InChI=1S/C27H24N8O/c1-17-12-18-8-9-19(32-27-29-11-10-25(28-2)33-27)13-23(18)35(17)22-6-4-5-7-24(22)36-20-14-21-26(30-15-20)34(3)16-31-21/h4-16H,1-3H3,(H2,28,29,32,33) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosomes as substrate preincubated for 30 mins followed by substrate a... |
ACS Med Chem Lett 7: 735-40 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00167
BindingDB Entry DOI: 10.7270/Q2V69P3G |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50529544
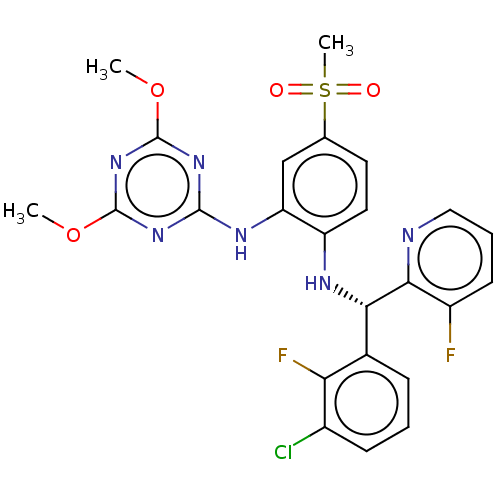
(CHEMBL4557484)Show SMILES COc1nc(Nc2cc(ccc2N[C@@H](c2cccc(Cl)c2F)c2ncccc2F)S(C)(=O)=O)nc(OC)n1 |r| Show InChI InChI=1S/C24H21ClF2N6O4S/c1-36-23-31-22(32-24(33-23)37-2)30-18-12-13(38(3,34)35)9-10-17(18)29-20(21-16(26)8-5-11-28-21)14-6-4-7-15(25)19(14)27/h4-12,20,29H,1-3H3,(H,30,31,32,33)/t20-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.590 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of 0.05 nM DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosome as substrate and [3H]SAM as co-factor preincubated for... |
ACS Med Chem Lett 10: 1655-1660 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00452
BindingDB Entry DOI: 10.7270/Q2RV0S5T |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50536819

(CHEMBL4534250)Show SMILES CNc1ccnc(Nc2ccc3sc(Cl)c(-c4cc(cnc4Cl)-c4c(C)ncn(C)c4=O)c3c2)n1 |(34.66,-43.04,;33.33,-43.82,;33.34,-45.35,;34.68,-46.12,;34.68,-47.66,;33.35,-48.43,;32.02,-47.66,;30.69,-48.43,;29.36,-47.67,;29.35,-46.12,;28.02,-45.36,;26.69,-46.13,;25.23,-45.65,;24.32,-46.89,;22.78,-46.88,;25.22,-48.14,;24.74,-49.6,;23.23,-49.91,;22.75,-51.37,;23.77,-52.52,;25.29,-52.2,;25.76,-50.74,;27.27,-50.42,;21.24,-51.68,;20.76,-53.14,;21.78,-54.29,;19.25,-53.46,;18.22,-52.31,;18.71,-50.84,;17.68,-49.69,;20.22,-50.53,;20.7,-49.07,;26.69,-47.67,;28.02,-48.44,;32.01,-46.13,)| Show InChI InChI=1S/C24H19Cl2N7OS/c1-12-19(23(34)33(3)11-30-12)13-8-16(21(25)29-10-13)20-15-9-14(4-5-17(15)35-22(20)26)31-24-28-7-6-18(27-2)32-24/h4-11H,1-3H3,(H2,27,28,31,32) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosomes as substrate preincubated for 30 mins followed by substrate a... |
ACS Med Chem Lett 7: 735-40 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00167
BindingDB Entry DOI: 10.7270/Q2V69P3G |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50529549

(CHEMBL4448208)Show SMILES CS(=O)(=O)c1ccc(N[C@H](c2ccccn2)c2cccc(Cl)c2F)c(Nc2nccc(n2)C(N)=O)c1 |r| Show InChI InChI=1S/C24H20ClFN6O3S/c1-36(34,35)14-8-9-17(20(13-14)32-24-29-12-10-19(31-24)23(27)33)30-22(18-7-2-3-11-28-18)15-5-4-6-16(25)21(15)26/h2-13,22,30H,1H3,(H2,27,33)(H,29,31,32)/t22-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of 0.05 nM DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosome as substrate and [3H]SAM as co-factor preincubated for... |
ACS Med Chem Lett 10: 1655-1660 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00452
BindingDB Entry DOI: 10.7270/Q2RV0S5T |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50235302

(CHEMBL4099771)Show SMILES CN(CCCNC(=O)CNC(=O)Nc1ccc2sc(Cl)c(-c3cccnc3C)c2c1)[C@@H]1CCCN(C1)c1ncnc2[nH]ccc12 |r,wU:30.32,(40.26,-16.4,;40.26,-17.94,;41.59,-18.71,;42.93,-17.94,;44.26,-18.71,;45.59,-17.94,;46.93,-18.71,;46.93,-20.25,;48.26,-17.94,;49.59,-18.71,;50.93,-17.94,;50.93,-16.4,;52.26,-18.71,;53.6,-17.95,;53.59,-16.41,;54.92,-15.64,;56.26,-16.41,;57.73,-15.93,;58.64,-17.18,;60.18,-17.18,;57.73,-18.43,;58.21,-19.89,;57.17,-21.03,;57.65,-22.5,;59.16,-22.82,;60.19,-21.66,;59.71,-20.2,;60.74,-19.06,;56.26,-17.95,;54.93,-18.72,;38.93,-18.71,;37.59,-17.93,;36.26,-18.71,;36.26,-20.25,;37.59,-21.01,;38.93,-20.24,;37.59,-22.55,;38.93,-23.31,;38.93,-24.86,;37.6,-25.63,;36.26,-24.86,;34.8,-25.34,;33.89,-24.1,;34.79,-22.85,;36.26,-23.32,)| Show InChI InChI=1S/C32H36ClN9O2S/c1-20-23(7-3-11-34-20)28-25-16-21(8-9-26(25)45-29(28)33)40-32(44)37-17-27(43)35-12-5-14-41(2)22-6-4-15-42(18-22)31-24-10-13-36-30(24)38-19-39-31/h3,7-11,13,16,19,22H,4-6,12,14-15,17-18H2,1-2H3,(H,35,43)(H,36,38,39)(H2,37,40,44)/t22-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L in human HeLa cells assessed as reduction in H3K79me2 level after 72 hrs by ELISA |
ACS Med Chem Lett 8: 338-343 (2017)
Article DOI: 10.1021/acsmedchemlett.6b00519
BindingDB Entry DOI: 10.7270/Q2WW7KZW |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50529549

(CHEMBL4448208)Show SMILES CS(=O)(=O)c1ccc(N[C@H](c2ccccn2)c2cccc(Cl)c2F)c(Nc2nccc(n2)C(N)=O)c1 |r| Show InChI InChI=1S/C24H20ClFN6O3S/c1-36(34,35)14-8-9-17(20(13-14)32-24-29-12-10-19(31-24)23(27)33)30-22(18-7-2-3-11-28-18)15-5-4-6-16(25)21(15)26/h2-13,22,30H,1H3,(H2,27,33)(H,29,31,32)/t22-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of 0.5 nM DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosome as substrate and [3H]SAM as co-factor preincubated for ... |
ACS Med Chem Lett 10: 1655-1660 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00452
BindingDB Entry DOI: 10.7270/Q2RV0S5T |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50235300
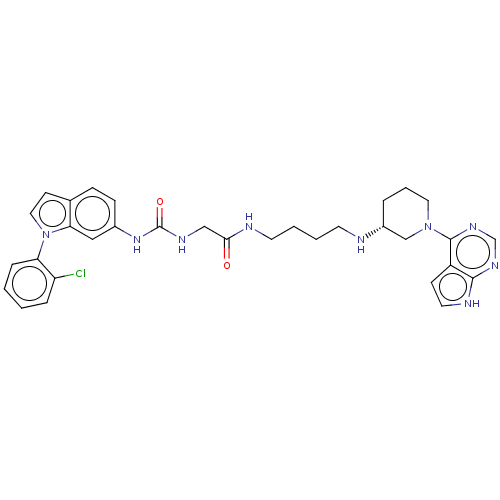
(CHEMBL4087730)Show SMILES Clc1ccccc1-n1ccc2ccc(NC(=O)NCC(=O)NCCCCN[C@@H]3CCCN(C3)c3ncnc4[nH]ccc34)cc12 |r| Show InChI InChI=1S/C32H36ClN9O2/c33-26-7-1-2-8-27(26)42-17-12-22-9-10-23(18-28(22)42)40-32(44)37-19-29(43)35-14-4-3-13-34-24-6-5-16-41(20-24)31-25-11-15-36-30(25)38-21-39-31/h1-2,7-12,15,17-18,21,24,34H,3-6,13-14,16,19-20H2,(H,35,43)(H,36,38,39)(H2,37,40,44)/t24-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosomes as substrate preincubated for 30 mins followed by substrate a... |
ACS Med Chem Lett 8: 338-343 (2017)
Article DOI: 10.1021/acsmedchemlett.6b00519
BindingDB Entry DOI: 10.7270/Q2WW7KZW |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50235299
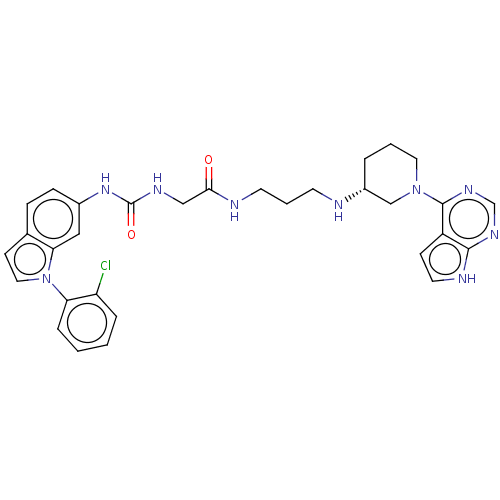
(CHEMBL4066397)Show SMILES Clc1ccccc1-n1ccc2ccc(NC(=O)NCC(=O)NCCCN[C@@H]3CCCN(C3)c3ncnc4[nH]ccc34)cc12 |r| Show InChI InChI=1S/C31H34ClN9O2/c32-25-6-1-2-7-26(25)41-16-11-21-8-9-22(17-27(21)41)39-31(43)36-18-28(42)34-13-4-12-33-23-5-3-15-40(19-23)30-24-10-14-35-29(24)37-20-38-30/h1-2,6-11,14,16-17,20,23,33H,3-5,12-13,15,18-19H2,(H,34,42)(H,35,37,38)(H2,36,39,43)/t23-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosomes as substrate preincubated for 30 mins followed by substrate a... |
ACS Med Chem Lett 8: 338-343 (2017)
Article DOI: 10.1021/acsmedchemlett.6b00519
BindingDB Entry DOI: 10.7270/Q2WW7KZW |
More data for this
Ligand-Target Pair | |
GTPase KRas
(Homo sapiens (Human)) | BDBM608937
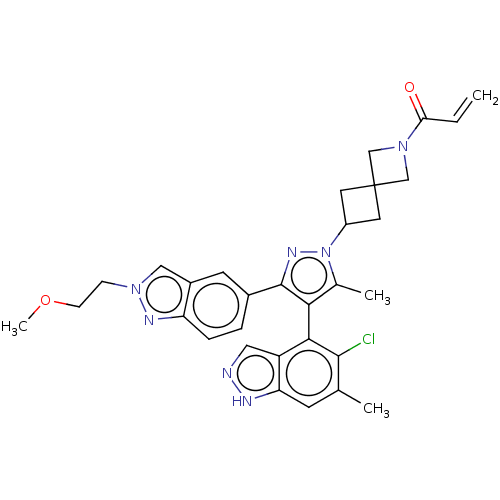
(1-(6-{(4M)-4-(5-Chloro-6- methyl-1H-indazol-4-yl)-...)Show SMILES COCCn1cc2cc(ccc2n1)-c1nn(C2CC3(C2)CN(C3)C(=O)C=C)c(C)c1-c1c(Cl)c(C)cc2[nH]ncc12 |(9.82,-.8,;9.42,-2.28,;7.94,-2.68,;6.85,-1.59,;5.36,-1.99,;4.21,-.96,;2.88,-1.73,;1.42,-1.26,;.27,-2.29,;.59,-3.79,;2.06,-4.27,;3.2,-3.24,;4.73,-3.4,;-1.19,-1.81,;-1.67,-.35,;-3.21,-.35,;-3.98,.99,;-5.47,1.39,;-5.07,2.87,;-3.58,2.48,;-6.55,3.27,;-6.16,4.76,;-4.67,4.36,;-7.25,5.85,;-6.85,7.34,;-8.73,5.45,;-9.82,6.54,;-3.68,-1.81,;-5.15,-2.29,;-2.44,-2.72,;-2.44,-4.26,;-3.77,-5.03,;-5.11,-4.26,;-3.77,-6.57,;-5.11,-7.34,;-2.44,-7.34,;-1.1,-6.57,;.36,-7.04,;1.27,-5.8,;.36,-4.55,;-1.1,-5.03,)| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50235299

(CHEMBL4066397)Show SMILES Clc1ccccc1-n1ccc2ccc(NC(=O)NCC(=O)NCCCN[C@@H]3CCCN(C3)c3ncnc4[nH]ccc34)cc12 |r| Show InChI InChI=1S/C31H34ClN9O2/c32-25-6-1-2-7-26(25)41-16-11-21-8-9-22(17-27(21)41)39-31(43)36-18-28(42)34-13-4-12-33-23-5-3-15-40(19-23)30-24-10-14-35-29(24)37-20-38-30/h1-2,6-11,14,16-17,20,23,33H,3-5,12-13,15,18-19H2,(H,34,42)(H,35,37,38)(H2,36,39,43)/t23-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosomes as substrate preincubated for 30 mins followed by substrate a... |
ACS Med Chem Lett 8: 338-343 (2017)
Article DOI: 10.1021/acsmedchemlett.6b00519
BindingDB Entry DOI: 10.7270/Q2WW7KZW |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50235300

(CHEMBL4087730)Show SMILES Clc1ccccc1-n1ccc2ccc(NC(=O)NCC(=O)NCCCCN[C@@H]3CCCN(C3)c3ncnc4[nH]ccc34)cc12 |r| Show InChI InChI=1S/C32H36ClN9O2/c33-26-7-1-2-8-27(26)42-17-12-22-9-10-23(18-28(22)42)40-32(44)37-19-29(43)35-14-4-3-13-34-24-6-5-16-41(20-24)31-25-11-15-36-30(25)38-21-39-31/h1-2,7-12,15,17-18,21,24,34H,3-6,13-14,16,19-20H2,(H,35,43)(H,36,38,39)(H2,37,40,44)/t24-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosomes as substrate preincubated for 30 mins followed by substrate a... |
ACS Med Chem Lett 8: 338-343 (2017)
Article DOI: 10.1021/acsmedchemlett.6b00519
BindingDB Entry DOI: 10.7270/Q2WW7KZW |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50075098

(CHEMBL3414626 | US10143704, Compound A2 | US944606...)Show SMILES CC(C)N(C[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12)[C@@H]1C[C@H](CCc2nc3cc(ccc3[nH]2)C(C)(C)C)C1 |r,wU:24.27,22.24,10.11,8.8,wD:5.4,7.12,(-.55,4.43,;-1.02,5.57,;-2.24,5.73,;-.08,6.8,;1.45,6.59,;2.04,5.17,;1.23,3.86,;2.24,2.7,;3.65,3.27,;4.7,2.61,;3.54,4.8,;4.48,5.6,;1.76,1.24,;2.66,.02,;1.76,-1.24,;.3,-.77,;-1.03,-1.56,;-1.03,-2.79,;-2.38,-.77,;-2.38,.77,;-1.03,1.56,;.3,.77,;-.67,8.22,;-.05,9.59,;-1.48,10.19,;-2.06,11.61,;-1.11,12.83,;-1.7,14.26,;-.89,15.53,;-1.87,16.71,;-1.63,18.24,;-2.86,19.2,;-4.29,18.63,;-4.52,17.09,;-3.29,16.14,;-3.19,14.61,;-2.65,20.73,;-1.51,21.19,;-3.62,21.48,;-2.48,21.95,;-2.07,8.77,)| Show InChI InChI=1S/C30H42N8O3/c1-16(2)37(13-22-25(39)26(40)29(41-22)38-15-34-24-27(31)32-14-33-28(24)38)19-10-17(11-19)6-9-23-35-20-8-7-18(30(3,4)5)12-21(20)36-23/h7-8,12,14-17,19,22,25-26,29,39-40H,6,9-11,13H2,1-5H3,(H,35,36)(H2,31,32,33)/t17-,19+,22-,25-,26-,29-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L in human HeLa cells assessed as reduction in H3K79me2 level after 72 hrs by ELISA |
ACS Med Chem Lett 8: 338-343 (2017)
Article DOI: 10.1021/acsmedchemlett.6b00519
BindingDB Entry DOI: 10.7270/Q2WW7KZW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
GTPase KRas
(Homo sapiens (Human)) | BDBM50609524
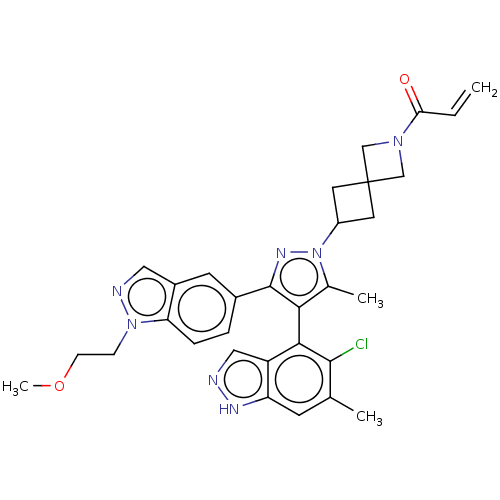
(CHEMBL5281254) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50536831
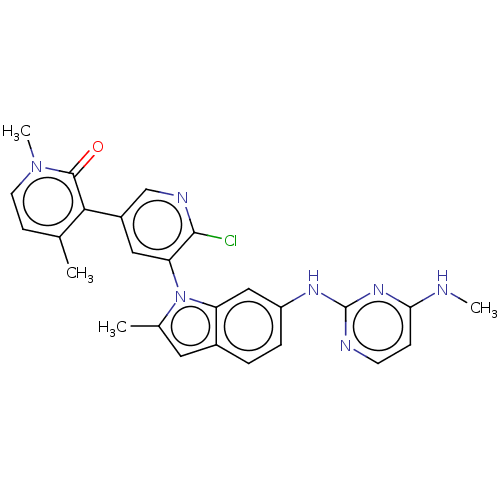
(CHEMBL4549878)Show SMILES CNc1ccnc(Nc2ccc3cc(C)n(-c4cc(cnc4Cl)-c4c(C)ccn(C)c4=O)c3c2)n1 |(56.02,-27.64,;54.69,-28.42,;54.7,-29.96,;56.04,-30.73,;56.05,-32.27,;54.71,-33.04,;53.39,-32.27,;52.05,-33.04,;50.72,-32.27,;50.71,-30.72,;49.38,-29.96,;48.05,-30.74,;46.59,-30.26,;45.68,-31.5,;44.14,-31.5,;46.58,-32.75,;46.11,-34.21,;44.6,-34.53,;44.12,-35.99,;45.15,-37.14,;46.66,-36.81,;47.14,-35.35,;48.64,-35.03,;42.63,-36.31,;42.15,-37.77,;43.18,-38.92,;40.64,-38.08,;39.61,-36.93,;40.1,-35.47,;39.07,-34.32,;41.6,-35.16,;42.08,-33.69,;48.05,-32.27,;49.38,-33.05,;53.37,-30.73,)| Show InChI InChI=1S/C26H24ClN7O/c1-15-8-10-33(4)25(35)23(15)18-12-21(24(27)30-14-18)34-16(2)11-17-5-6-19(13-20(17)34)31-26-29-9-7-22(28-3)32-26/h5-14H,1-4H3,(H2,28,29,31,32) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosomes as substrate preincubated for 30 mins followed by substrate a... |
ACS Med Chem Lett 7: 735-40 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00167
BindingDB Entry DOI: 10.7270/Q2V69P3G |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
GTPase KRas
(Homo sapiens (Human)) | BDBM50609523
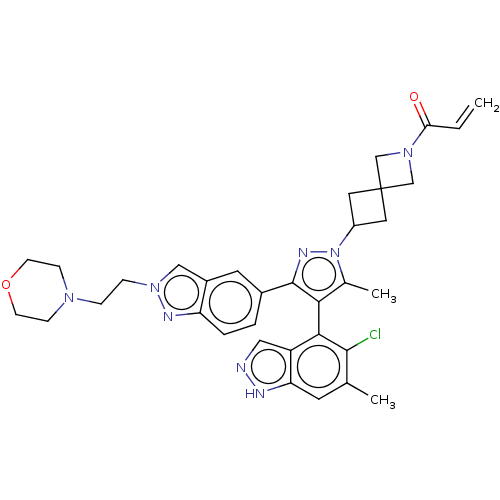
(CHEMBL5271997) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50535036
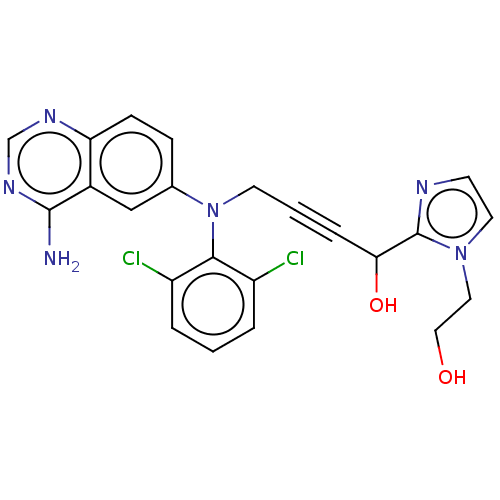
(CHEMBL4452414)Show SMILES Nc1ncnc2ccc(cc12)N(CC#CC(O)c1nccn1CCO)c1c(Cl)cccc1Cl Show InChI InChI=1S/C23H20Cl2N6O2/c24-17-3-1-4-18(25)21(17)31(15-6-7-19-16(13-15)22(26)29-14-28-19)9-2-5-20(33)23-27-8-10-30(23)11-12-32/h1,3-4,6-8,10,13-14,20,32-33H,9,11-12H2,(H2,26,28,29) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human DOTL1 (2 to 416 residues)-mediated methylation of nucleosome preincubated for 30 mins followed by addition of S-[methyl-3H-] aden... |
ACS Med Chem Lett 7: 730-4 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00168
BindingDB Entry DOI: 10.7270/Q2N87F8W |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50536826

(CHEMBL4590355)Show SMILES CNc1ccnc(Nc2ccc3cc(C)n(-c4ccccc4Oc4cnc5n(C)cnc5c4)c3c2)n1 |(29.26,-10.25,;27.93,-11.02,;27.94,-12.56,;29.28,-13.33,;29.28,-14.87,;27.95,-15.64,;26.62,-14.88,;25.29,-15.64,;23.96,-14.88,;23.95,-13.33,;22.62,-12.56,;21.29,-13.34,;19.82,-12.85,;18.9,-14.1,;17.36,-14.08,;19.8,-15.36,;19.31,-16.81,;20.33,-17.95,;19.85,-19.4,;18.34,-19.72,;17.32,-18.56,;17.81,-17.11,;16.79,-15.96,;15.29,-16.26,;14.81,-17.71,;13.31,-18.01,;12.29,-16.86,;10.74,-16.84,;9.83,-18.07,;10.28,-15.37,;11.54,-14.47,;12.78,-15.39,;14.28,-15.1,;21.28,-14.89,;22.62,-15.65,;26.61,-13.34,)| Show InChI InChI=1S/C27H24N8O/c1-17-12-18-8-9-19(32-27-29-11-10-25(28-2)33-27)13-23(18)35(17)22-6-4-5-7-24(22)36-20-14-21-26(30-15-20)34(3)16-31-21/h4-16H,1-3H3,(H2,28,29,32,33) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L in human HeLa cells assessed as reduction in H3K79me2 level after 72 hrs by ELISA |
ACS Med Chem Lett 7: 735-40 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00167
BindingDB Entry DOI: 10.7270/Q2V69P3G |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50529552

(CHEMBL4589340)Show SMILES CS(=O)(=O)c1ccc(N[C@@H](c2cccc(Cl)c2F)c2ncccc2F)c(Nc2ncccn2)c1 |r| Show InChI InChI=1S/C23H18ClF2N5O2S/c1-34(32,33)14-8-9-18(19(13-14)31-23-28-11-4-12-29-23)30-21(22-17(25)7-3-10-27-22)15-5-2-6-16(24)20(15)26/h2-13,21,30H,1H3,(H,28,29,31)/t21-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of 0.5 nM DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosome as substrate and [3H]SAM as co-factor preincubated for ... |
ACS Med Chem Lett 10: 1655-1660 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00452
BindingDB Entry DOI: 10.7270/Q2RV0S5T |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50235302

(CHEMBL4099771)Show SMILES CN(CCCNC(=O)CNC(=O)Nc1ccc2sc(Cl)c(-c3cccnc3C)c2c1)[C@@H]1CCCN(C1)c1ncnc2[nH]ccc12 |r,wU:30.32,(40.26,-16.4,;40.26,-17.94,;41.59,-18.71,;42.93,-17.94,;44.26,-18.71,;45.59,-17.94,;46.93,-18.71,;46.93,-20.25,;48.26,-17.94,;49.59,-18.71,;50.93,-17.94,;50.93,-16.4,;52.26,-18.71,;53.6,-17.95,;53.59,-16.41,;54.92,-15.64,;56.26,-16.41,;57.73,-15.93,;58.64,-17.18,;60.18,-17.18,;57.73,-18.43,;58.21,-19.89,;57.17,-21.03,;57.65,-22.5,;59.16,-22.82,;60.19,-21.66,;59.71,-20.2,;60.74,-19.06,;56.26,-17.95,;54.93,-18.72,;38.93,-18.71,;37.59,-17.93,;36.26,-18.71,;36.26,-20.25,;37.59,-21.01,;38.93,-20.24,;37.59,-22.55,;38.93,-23.31,;38.93,-24.86,;37.6,-25.63,;36.26,-24.86,;34.8,-25.34,;33.89,-24.1,;34.79,-22.85,;36.26,-23.32,)| Show InChI InChI=1S/C32H36ClN9O2S/c1-20-23(7-3-11-34-20)28-25-16-21(8-9-26(25)45-29(28)33)40-32(44)37-17-27(43)35-12-5-14-41(2)22-6-4-15-42(18-22)31-24-10-13-36-30(24)38-19-39-31/h3,7-11,13,16,19,22H,4-6,12,14-15,17-18H2,1-2H3,(H,35,43)(H,36,38,39)(H2,37,40,44)/t22-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L in human MOLM13 cells assessed as suppression of HoxA9 gene after 72 hrs by luciferase reporter gene assay |
ACS Med Chem Lett 8: 338-343 (2017)
Article DOI: 10.1021/acsmedchemlett.6b00519
BindingDB Entry DOI: 10.7270/Q2WW7KZW |
More data for this
Ligand-Target Pair | |
GTPase KRas
(Homo sapiens (Human)) | BDBM608850
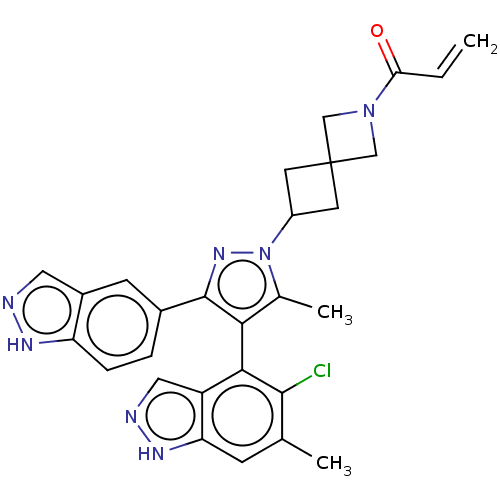
(US11702409, Example 12a | US11702409, Example 12b)Show SMILES Cc1c(c(nn1C1CC2(C1)CN(C2)C(=O)C=C)-c1ccc2[nH]ncc2c1)-c1c(Cl)c(C)cc2[nH]ncc12 |(-2.64,-2.29,;-1.17,-1.81,;.07,-2.72,;1.32,-1.81,;.84,-.35,;-.7,-.35,;-1.47,.99,;-2.95,1.39,;-2.56,2.87,;-1.07,2.48,;-4.04,3.27,;-3.65,4.76,;-2.16,4.36,;-4.73,5.85,;-4.34,7.34,;-6.22,5.45,;-7.31,6.54,;2.78,-2.29,;3.69,-1.04,;5.22,-1.2,;5.85,-2.61,;7.31,-3.08,;7.31,-4.62,;5.85,-5.1,;4.94,-3.85,;3.41,-3.69,;.07,-4.26,;-1.26,-5.03,;-2.59,-4.26,;-1.26,-6.57,;-2.59,-7.34,;.07,-7.34,;1.41,-6.57,;2.87,-7.04,;3.78,-5.8,;2.87,-4.55,;1.41,-5.03,)| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
GTPase KRas
(Homo sapiens (Human)) | BDBM50579985

(JDQ-443 | JDQ443 | Jdq 443 | Jdq-443 | Nvp-jdq-443...)Show SMILES Cc1c(c(nn1C1CC2(C1)CN(C2)C(=O)C=C)-c1ccc2n(C)ncc2c1)-c1c(Cl)c(C)cc2[nH]ncc12 |(8.62,-12.01,;10.09,-11.52,;11.34,-12.43,;12.58,-11.52,;12.1,-10.05,;10.56,-10.06,;9.66,-8.81,;9.89,-7.29,;8.37,-7.05,;8.13,-8.58,;8.77,-5.56,;7.27,-5.16,;6.88,-6.65,;6.5,-3.83,;7.27,-2.49,;4.96,-3.83,;4.19,-2.5,;14.04,-11.99,;15.18,-10.95,;16.64,-11.42,;16.97,-12.93,;18.31,-13.69,;19.71,-13.06,;18,-15.2,;16.47,-15.37,;15.84,-13.97,;14.37,-13.49,;11.35,-13.97,;10.02,-14.74,;8.69,-13.97,;10.02,-16.28,;8.69,-17.05,;11.35,-17.05,;12.69,-16.27,;14.15,-16.75,;15.06,-15.5,;14.15,-14.25,;12.69,-14.73,)| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| | n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
GTPase KRas
(Homo sapiens (Human)) | BDBM608886
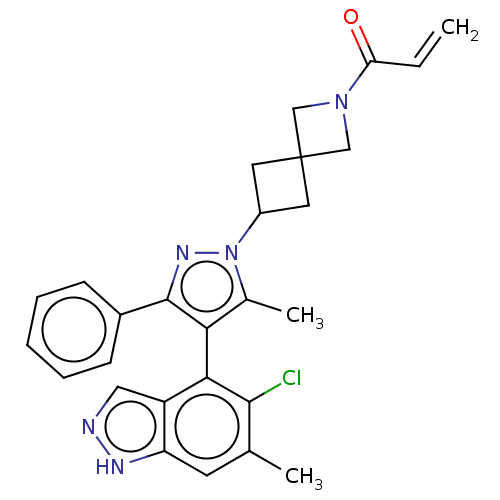
(1-{6-[(4M)-4-(5-Chloro-6- methyl-1H-indazol-4-yl)-...)Show SMILES Cc1c(c(nn1C1CC2(C1)CN(C2)C(=O)C=C)-c1ccccc1)-c1c(Cl)c(C)cc2[nH]ncc12 |(-2.38,-2.83,;-.92,-2.36,;.33,-3.26,;1.57,-2.36,;1.1,-.89,;-.44,-.89,;-1.21,.44,;-2.7,.84,;-2.3,2.33,;-.81,1.93,;-3.79,2.73,;-3.39,4.22,;-1.9,3.82,;-4.48,5.3,;-5.97,4.91,;-4.08,6.79,;-5.17,7.88,;3.04,-2.83,;3.36,-4.34,;4.82,-4.81,;5.97,-3.78,;5.65,-2.28,;4.18,-1.8,;.33,-4.8,;-1.01,-5.57,;-2.34,-4.8,;-1.01,-7.11,;-2.34,-7.88,;.33,-7.88,;1.66,-7.11,;3.13,-7.59,;4.03,-6.34,;3.13,-5.09,;1.66,-5.57,)| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50536821

(CHEMBL4586619)Show SMILES Clc1ccccc1-c1nnnn1CC(=O)NNC(=O)Nc1ccc2ccn(-c3ccccc3Cl)c2c1 Show InChI InChI=1S/C24H18Cl2N8O2/c25-18-6-2-1-5-17(18)23-29-31-32-34(23)14-22(35)28-30-24(36)27-16-10-9-15-11-12-33(21(15)13-16)20-8-4-3-7-19(20)26/h1-13H,14H2,(H,28,35)(H2,27,30,36) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosomes as substrate preincubated for 30 mins followed by substrate a... |
ACS Med Chem Lett 7: 735-40 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00167
BindingDB Entry DOI: 10.7270/Q2V69P3G |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50536819

(CHEMBL4534250)Show SMILES CNc1ccnc(Nc2ccc3sc(Cl)c(-c4cc(cnc4Cl)-c4c(C)ncn(C)c4=O)c3c2)n1 |(34.66,-43.04,;33.33,-43.82,;33.34,-45.35,;34.68,-46.12,;34.68,-47.66,;33.35,-48.43,;32.02,-47.66,;30.69,-48.43,;29.36,-47.67,;29.35,-46.12,;28.02,-45.36,;26.69,-46.13,;25.23,-45.65,;24.32,-46.89,;22.78,-46.88,;25.22,-48.14,;24.74,-49.6,;23.23,-49.91,;22.75,-51.37,;23.77,-52.52,;25.29,-52.2,;25.76,-50.74,;27.27,-50.42,;21.24,-51.68,;20.76,-53.14,;21.78,-54.29,;19.25,-53.46,;18.22,-52.31,;18.71,-50.84,;17.68,-49.69,;20.22,-50.53,;20.7,-49.07,;26.69,-47.67,;28.02,-48.44,;32.01,-46.13,)| Show InChI InChI=1S/C24H19Cl2N7OS/c1-12-19(23(34)33(3)11-30-12)13-8-16(21(25)29-10-13)20-15-9-14(4-5-17(15)35-22(20)26)31-24-28-7-6-18(27-2)32-24/h4-11H,1-3H3,(H2,27,28,31,32) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L in human HeLa cells assessed as reduction in H3K79me2 level after 72 hrs by ELISA |
ACS Med Chem Lett 7: 735-40 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00167
BindingDB Entry DOI: 10.7270/Q2V69P3G |
More data for this
Ligand-Target Pair | |
GTPase KRas
(Homo sapiens (Human)) | BDBM608857
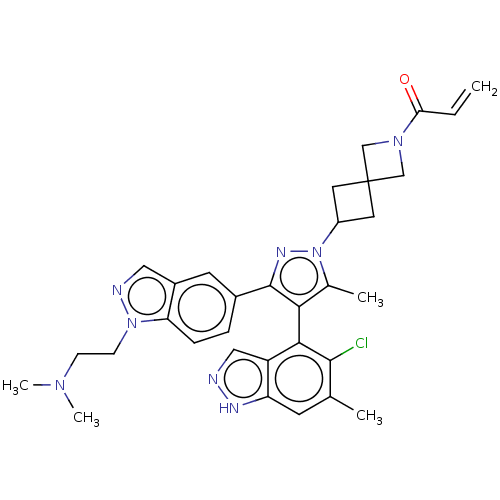
(US11702409, Example 15a | US11702409, Example 15b)Show SMILES CN(C)CCn1ncc2cc(ccc12)-c1nn(C2CC3(C2)CN(C3)C(=O)C=C)c(C)c1-c1c(Cl)c(C)cc2[nH]ncc12 |(6.12,-8.04,;6.89,-6.7,;8.43,-6.7,;6.12,-5.37,;6.89,-4.03,;6.12,-2.7,;6.75,-1.29,;5.6,-.26,;4.27,-1.03,;2.81,-.56,;1.66,-1.59,;1.98,-3.09,;3.45,-3.57,;4.59,-2.54,;.2,-1.11,;-.28,.35,;-1.82,.35,;-2.59,1.69,;-4.08,2.09,;-3.68,3.57,;-2.19,3.17,;-5.17,3.97,;-4.77,5.46,;-3.28,5.06,;-5.86,6.55,;-5.46,8.04,;-7.34,6.15,;-8.43,7.24,;-2.29,-1.11,;-3.76,-1.59,;-1.05,-2.02,;-1.05,-3.56,;-2.38,-4.33,;-3.72,-3.56,;-2.38,-5.87,;-3.72,-6.64,;-1.05,-6.64,;.28,-5.87,;1.75,-6.34,;2.65,-5.1,;1.75,-3.85,;.28,-4.33,)| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50535037

(CHEMBL4436922)Show SMILES Cc1ccc(nc1)C#CCN(c1ccc2ncnc(N)c2c1)c1c(Cl)cccc1Cl Show InChI InChI=1S/C23H17Cl2N5/c1-15-7-8-16(27-13-15)4-3-11-30(22-19(24)5-2-6-20(22)25)17-9-10-21-18(12-17)23(26)29-14-28-21/h2,5-10,12-14H,11H2,1H3,(H2,26,28,29) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 47 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human DOTL1 (2 to 416 residues)-mediated methylation of nucleosome preincubated for 30 mins followed by addition of S-[methyl-3H-] aden... |
ACS Med Chem Lett 7: 730-4 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00168
BindingDB Entry DOI: 10.7270/Q2N87F8W |
More data for this
Ligand-Target Pair | |
GTPase KRas
(Homo sapiens (Human)) | BDBM608856

(US11702409, Example 14)Show SMILES Cc1c(c(nn1C1CC2(C1)CN(C2)C(=O)C=C)-c1ccc2cn[nH]c2c1)-c1c(Cl)c(C)cc2[nH]ncc12 |(-2.51,-2.29,;-1.04,-1.81,;.2,-2.72,;1.45,-1.81,;.97,-.35,;-.57,-.35,;-1.34,.99,;-2.82,1.39,;-2.42,2.87,;-.94,2.48,;-3.91,3.27,;-3.51,4.76,;-2.03,4.36,;-4.6,5.85,;-4.2,7.34,;-6.09,5.45,;-7.18,6.54,;2.92,-2.29,;4.06,-1.26,;5.52,-1.73,;5.84,-3.24,;7.18,-4.01,;6.86,-5.51,;5.33,-5.68,;4.7,-4.27,;3.24,-3.79,;.2,-4.26,;-1.13,-5.03,;-2.46,-4.26,;-1.13,-6.57,;-2.46,-7.34,;.2,-7.34,;1.54,-6.57,;3,-7.04,;3.91,-5.8,;3,-4.55,;1.54,-5.03,)| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50075098

(CHEMBL3414626 | US10143704, Compound A2 | US944606...)Show SMILES CC(C)N(C[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12)[C@@H]1C[C@H](CCc2nc3cc(ccc3[nH]2)C(C)(C)C)C1 |r,wU:24.27,22.24,10.11,8.8,wD:5.4,7.12,(-.55,4.43,;-1.02,5.57,;-2.24,5.73,;-.08,6.8,;1.45,6.59,;2.04,5.17,;1.23,3.86,;2.24,2.7,;3.65,3.27,;4.7,2.61,;3.54,4.8,;4.48,5.6,;1.76,1.24,;2.66,.02,;1.76,-1.24,;.3,-.77,;-1.03,-1.56,;-1.03,-2.79,;-2.38,-.77,;-2.38,.77,;-1.03,1.56,;.3,.77,;-.67,8.22,;-.05,9.59,;-1.48,10.19,;-2.06,11.61,;-1.11,12.83,;-1.7,14.26,;-.89,15.53,;-1.87,16.71,;-1.63,18.24,;-2.86,19.2,;-4.29,18.63,;-4.52,17.09,;-3.29,16.14,;-3.19,14.61,;-2.65,20.73,;-1.51,21.19,;-3.62,21.48,;-2.48,21.95,;-2.07,8.77,)| Show InChI InChI=1S/C30H42N8O3/c1-16(2)37(13-22-25(39)26(40)29(41-22)38-15-34-24-27(31)32-14-33-28(24)38)19-10-17(11-19)6-9-23-35-20-8-7-18(30(3,4)5)12-21(20)36-23/h7-8,12,14-17,19,22,25-26,29,39-40H,6,9-11,13H2,1-5H3,(H,35,36)(H2,31,32,33)/t17-,19+,22-,25-,26-,29-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 52 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L in human MOLM13 cells assessed as suppression of HoxA9 gene after 72 hrs by luciferase reporter gene assay |
ACS Med Chem Lett 8: 338-343 (2017)
Article DOI: 10.1021/acsmedchemlett.6b00519
BindingDB Entry DOI: 10.7270/Q2WW7KZW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50535034
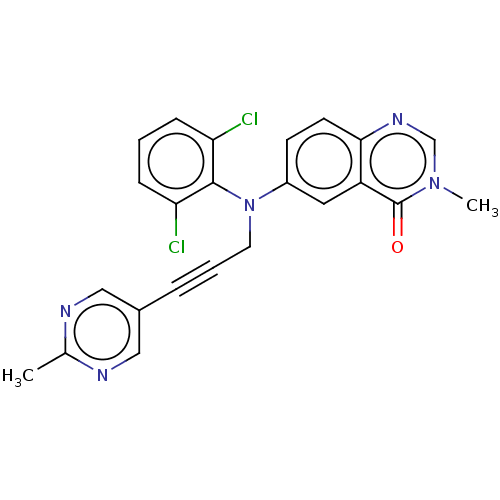
(CHEMBL4548823)Show SMILES Cc1ncc(cn1)C#CCN(c1ccc2ncn(C)c(=O)c2c1)c1c(Cl)cccc1Cl Show InChI InChI=1S/C23H17Cl2N5O/c1-15-26-12-16(13-27-15)5-4-10-30(22-19(24)6-3-7-20(22)25)17-8-9-21-18(11-17)23(31)29(2)14-28-21/h3,6-9,11-14H,10H2,1-2H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 89 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human DOTL1 (2 to 416 residues)-mediated methylation of nucleosome preincubated for 30 mins followed by addition of S-[methyl-3H-] aden... |
ACS Med Chem Lett 7: 730-4 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00168
BindingDB Entry DOI: 10.7270/Q2N87F8W |
More data for this
Ligand-Target Pair | |
GTPase KRas
(Homo sapiens (Human)) | BDBM608888
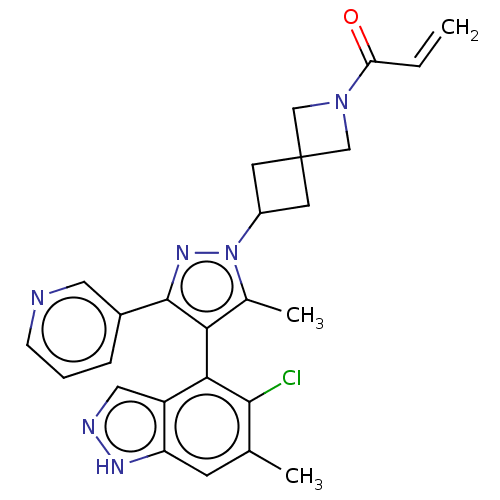
(US11702409, Example 27a | US11702409, Example 27b)Show SMILES Cc1c(c(nn1C1CC2(C1)CN(C2)C(=O)C=C)-c1cccnc1)-c1c(Cl)c(C)cc2[nH]ncc12 |(-2.38,-2.83,;-.92,-2.36,;.33,-3.26,;1.57,-2.36,;1.1,-.89,;-.44,-.89,;-1.21,.44,;-2.7,.84,;-2.3,2.33,;-.81,1.93,;-3.79,2.73,;-3.39,4.22,;-1.9,3.82,;-4.48,5.3,;-5.97,4.91,;-4.08,6.79,;-5.17,7.88,;3.04,-2.83,;3.36,-4.34,;4.82,-4.81,;5.97,-3.78,;5.65,-2.28,;4.18,-1.8,;.33,-4.8,;-1.01,-5.57,;-2.34,-4.8,;-1.01,-7.11,;-2.34,-7.88,;.33,-7.88,;1.66,-7.11,;3.13,-7.59,;4.03,-6.34,;3.13,-5.09,;1.66,-5.57,)| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50529548

(CHEMBL4587469)Show SMILES CS(=O)(=O)c1ccc(N[C@H](c2ccccn2)c2cccc(Cl)c2F)c(Nc2ncccn2)c1 |r| Show InChI InChI=1S/C23H19ClFN5O2S/c1-33(31,32)15-9-10-18(20(14-15)30-23-27-12-5-13-28-23)29-22(19-8-2-3-11-26-19)16-6-4-7-17(24)21(16)25/h2-14,22,29H,1H3,(H,27,28,30)/t22-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 97 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of 0.5 nM DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosome as substrate and [3H]SAM as co-factor preincubated for ... |
ACS Med Chem Lett 10: 1655-1660 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00452
BindingDB Entry DOI: 10.7270/Q2RV0S5T |
More data for this
Ligand-Target Pair | |
GTPase KRas
(Homo sapiens (Human)) | BDBM608896
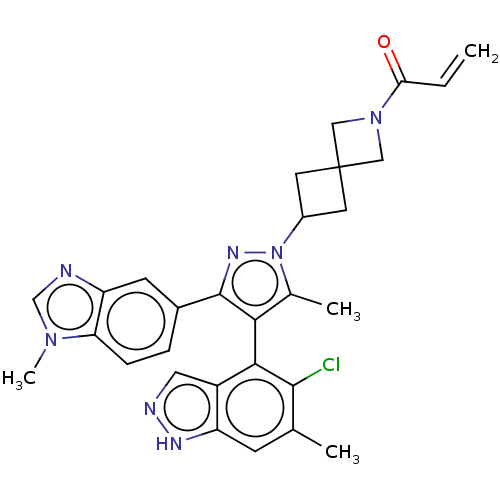
(US11702409, Example 30a | US11702409, Example 30b)Show SMILES Cc1c(c(nn1C1CC2(C1)CN(C2)C(=O)C=C)-c1ccc2n(C)cnc2c1)-c1c(Cl)c(C)cc2[nH]ncc12 |(-2.99,-2.29,;-1.52,-1.81,;-.28,-2.72,;.97,-1.81,;.49,-.35,;-1.05,-.35,;-1.82,.99,;-3.31,1.39,;-2.91,2.87,;-1.42,2.48,;-4.4,3.27,;-4,4.76,;-2.51,4.36,;-5.09,5.85,;-4.69,7.34,;-6.57,5.45,;-7.66,6.54,;2.43,-2.29,;2.75,-3.79,;4.22,-4.27,;5.36,-3.24,;6.89,-3.4,;7.66,-4.73,;7.52,-1.99,;6.37,-.96,;5.04,-1.73,;3.58,-1.26,;-.28,-4.26,;-1.61,-5.03,;-2.95,-4.26,;-1.61,-6.57,;-2.95,-7.34,;-.28,-7.34,;1.05,-6.57,;2.52,-7.04,;3.42,-5.8,;2.52,-4.55,;1.05,-5.03,)| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50536825

(CHEMBL4577516)Show SMILES CNc1ccnc(Nc2ccc3cc(C)n(-c4ccccc4Oc4ccccc4)c3c2)n1 |(17.24,-40.96,;15.91,-41.73,;15.92,-43.27,;17.27,-44.04,;17.27,-45.58,;15.94,-46.35,;14.61,-45.59,;13.28,-46.36,;11.94,-45.59,;11.93,-44.04,;10.6,-43.28,;9.27,-44.05,;7.81,-43.57,;6.9,-44.82,;5.36,-44.82,;7.8,-46.06,;7.33,-47.53,;8.36,-48.67,;7.89,-50.13,;6.37,-50.45,;5.34,-49.31,;5.82,-47.84,;4.79,-46.7,;3.29,-47.01,;2.26,-45.86,;.76,-46.18,;.28,-47.64,;1.31,-48.79,;2.82,-48.47,;9.27,-45.59,;10.6,-46.36,;14.6,-44.05,)| Show InChI InChI=1S/C26H23N5O/c1-18-16-19-12-13-20(29-26-28-15-14-25(27-2)30-26)17-23(19)31(18)22-10-6-7-11-24(22)32-21-8-4-3-5-9-21/h3-17H,1-2H3,(H2,27,28,29,30) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 150 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosomes as substrate preincubated for 30 mins followed by substrate a... |
ACS Med Chem Lett 7: 735-40 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00167
BindingDB Entry DOI: 10.7270/Q2V69P3G |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50529553

(CHEMBL4531049)Show SMILES CS(=O)(=O)c1ccc(N[C@@H](c2cccc(Cl)c2)c2ccccn2)c(Nc2ncccn2)c1 |r| Show InChI InChI=1S/C23H20ClN5O2S/c1-32(30,31)18-9-10-19(21(15-18)29-23-26-12-5-13-27-23)28-22(20-8-2-3-11-25-20)16-6-4-7-17(24)14-16/h2-15,22,28H,1H3,(H,26,27,29)/t22-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 270 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of 0.5 nM DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosome as substrate and [3H]SAM as co-factor preincubated for ... |
ACS Med Chem Lett 10: 1655-1660 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00452
BindingDB Entry DOI: 10.7270/Q2RV0S5T |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50536820

(CHEMBL4573709)Show SMILES CNc1ccnc(Nc2ccc3cc(C)n(-c4cccnc4Cl)c3c2)n1 |(40.94,-15.9,;39.61,-16.67,;39.62,-18.21,;40.96,-18.98,;40.97,-20.52,;39.63,-21.29,;38.31,-20.53,;36.97,-21.3,;35.64,-20.53,;35.63,-18.98,;34.3,-18.22,;32.97,-18.99,;31.51,-18.51,;30.6,-19.76,;29.06,-19.76,;31.5,-21,;31.03,-22.47,;29.52,-22.78,;29.04,-24.24,;30.07,-25.39,;31.58,-25.07,;32.06,-23.61,;33.56,-23.28,;32.97,-20.53,;34.3,-21.3,;38.29,-18.99,)| Show InChI InChI=1S/C19H17ClN6/c1-12-10-13-5-6-14(24-19-23-9-7-17(21-2)25-19)11-16(13)26(12)15-4-3-8-22-18(15)20/h3-11H,1-2H3,(H2,21,23,24,25) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 280 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L (2 to 416 residues) (unknown origin) using biotinylated nucleosomes as substrate preincubated for 30 mins followed by substrate a... |
ACS Med Chem Lett 7: 735-40 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00167
BindingDB Entry DOI: 10.7270/Q2V69P3G |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50536826

(CHEMBL4590355)Show SMILES CNc1ccnc(Nc2ccc3cc(C)n(-c4ccccc4Oc4cnc5n(C)cnc5c4)c3c2)n1 |(29.26,-10.25,;27.93,-11.02,;27.94,-12.56,;29.28,-13.33,;29.28,-14.87,;27.95,-15.64,;26.62,-14.88,;25.29,-15.64,;23.96,-14.88,;23.95,-13.33,;22.62,-12.56,;21.29,-13.34,;19.82,-12.85,;18.9,-14.1,;17.36,-14.08,;19.8,-15.36,;19.31,-16.81,;20.33,-17.95,;19.85,-19.4,;18.34,-19.72,;17.32,-18.56,;17.81,-17.11,;16.79,-15.96,;15.29,-16.26,;14.81,-17.71,;13.31,-18.01,;12.29,-16.86,;10.74,-16.84,;9.83,-18.07,;10.28,-15.37,;11.54,-14.47,;12.78,-15.39,;14.28,-15.1,;21.28,-14.89,;22.62,-15.65,;26.61,-13.34,)| Show InChI InChI=1S/C27H24N8O/c1-17-12-18-8-9-19(32-27-29-11-10-25(28-2)33-27)13-23(18)35(17)22-6-4-5-7-24(22)36-20-14-21-26(30-15-20)34(3)16-31-21/h4-16H,1-3H3,(H2,28,29,32,33) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 340 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L in human MOLM13 cells assessed as suppression of HoxA9 gene after 72 hrs by luciferase reporter gene assay |
ACS Med Chem Lett 7: 735-40 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00167
BindingDB Entry DOI: 10.7270/Q2V69P3G |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50536819

(CHEMBL4534250)Show SMILES CNc1ccnc(Nc2ccc3sc(Cl)c(-c4cc(cnc4Cl)-c4c(C)ncn(C)c4=O)c3c2)n1 |(34.66,-43.04,;33.33,-43.82,;33.34,-45.35,;34.68,-46.12,;34.68,-47.66,;33.35,-48.43,;32.02,-47.66,;30.69,-48.43,;29.36,-47.67,;29.35,-46.12,;28.02,-45.36,;26.69,-46.13,;25.23,-45.65,;24.32,-46.89,;22.78,-46.88,;25.22,-48.14,;24.74,-49.6,;23.23,-49.91,;22.75,-51.37,;23.77,-52.52,;25.29,-52.2,;25.76,-50.74,;27.27,-50.42,;21.24,-51.68,;20.76,-53.14,;21.78,-54.29,;19.25,-53.46,;18.22,-52.31,;18.71,-50.84,;17.68,-49.69,;20.22,-50.53,;20.7,-49.07,;26.69,-47.67,;28.02,-48.44,;32.01,-46.13,)| Show InChI InChI=1S/C24H19Cl2N7OS/c1-12-19(23(34)33(3)11-30-12)13-8-16(21(25)29-10-13)20-15-9-14(4-5-17(15)35-22(20)26)31-24-28-7-6-18(27-2)32-24/h4-11H,1-3H3,(H2,27,28,31,32) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 384 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L in human MOLM13 cells assessed as suppression of HoxA9 gene after 72 hrs by luciferase reporter gene assay |
ACS Med Chem Lett 7: 735-40 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00167
BindingDB Entry DOI: 10.7270/Q2V69P3G |
More data for this
Ligand-Target Pair | |
GTPase KRas
(Homo sapiens (Human)) | BDBM50609519
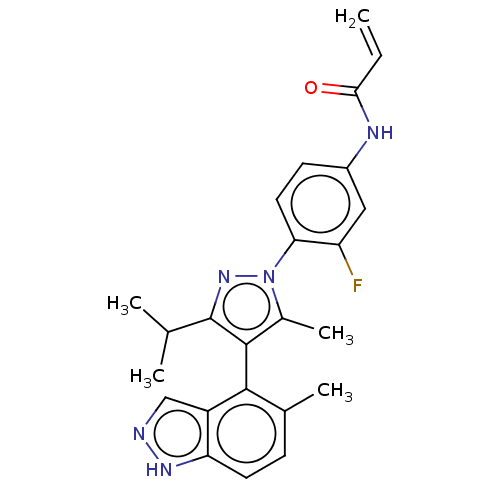
(CHEMBL5266172) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 500 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
GTPase KRas
(Homo sapiens (Human)) | BDBM608868
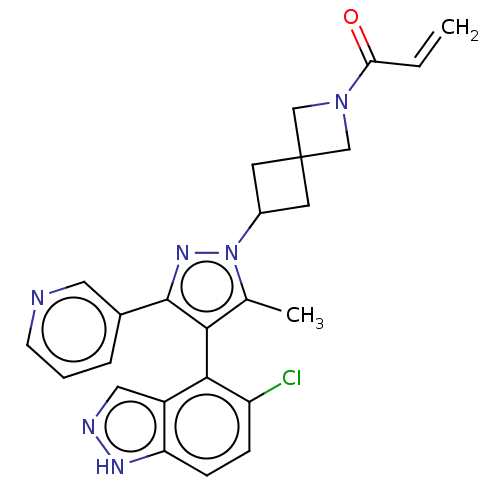
(US11702409, Example 19a | US11702409, Example 19b)Show SMILES Cc1c(c(nn1C1CC2(C1)CN(C2)C(=O)C=C)-c1cccnc1)-c1c(Cl)ccc2[nH]ncc12 |(-2.38,-2.83,;-.92,-2.36,;.33,-3.26,;1.57,-2.36,;1.1,-.89,;-.44,-.89,;-1.21,.44,;-2.7,.84,;-2.3,2.33,;-.81,1.93,;-3.79,2.73,;-3.39,4.22,;-1.9,3.82,;-4.48,5.3,;-5.97,4.91,;-4.08,6.79,;-5.17,7.88,;3.04,-2.83,;3.36,-4.34,;4.82,-4.81,;5.97,-3.78,;5.65,-2.28,;4.18,-1.8,;.33,-4.8,;1.66,-5.57,;3.15,-5.17,;1.66,-7.11,;.33,-7.88,;-1.01,-7.11,;-2.47,-7.59,;-3.38,-6.34,;-2.47,-5.09,;-1.01,-5.57,)| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 500 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
GTPase KRas
(Homo sapiens (Human)) | BDBM50609522

(CHEMBL5291136) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 700 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data