 Found 183 hits with Last Name = 'congy' and Initial = 'c'
Found 183 hits with Last Name = 'congy' and Initial = 'c' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cannabinoid receptor 2
(Rattus norvegicus (Rat)) | BDBM21280
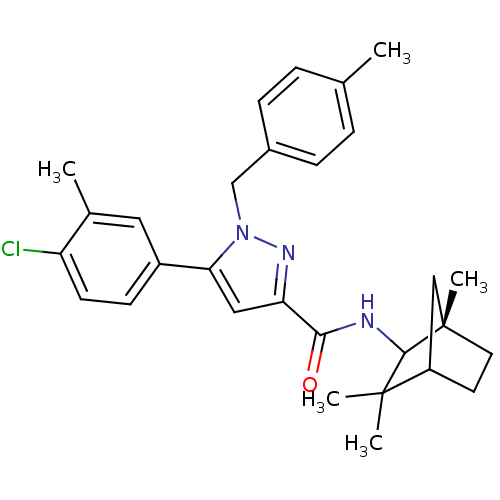
(5-(4-chloro-3-methylphenyl)-1-[(4-methylphenyl)met...)Show SMILES Cc1ccc(Cn2nc(cc2-c2ccc(Cl)c(C)c2)C(=O)NC2[C@@]3(C)CCC(C3)C2(C)C)cc1 |r,TLB:21:22:28:26.25| Show InChI InChI=1S/C29H34ClN3O/c1-18-6-8-20(9-7-18)17-33-25(21-10-11-23(30)19(2)14-21)15-24(32-33)26(34)31-27-28(3,4)22-12-13-29(27,5)16-22/h6-11,14-15,22,27H,12-13,16-17H2,1-5H3,(H,31,34)/t22?,27?,29-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 284: 644-50 (1998)
BindingDB Entry DOI: 10.7270/Q2D50KHT |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Rattus norvegicus (rat)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.560 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 2
(Homo sapiens (Human)) | BDBM21280

(5-(4-chloro-3-methylphenyl)-1-[(4-methylphenyl)met...)Show SMILES Cc1ccc(Cn2nc(cc2-c2ccc(Cl)c(C)c2)C(=O)NC2[C@@]3(C)CCC(C3)C2(C)C)cc1 |r,TLB:21:22:28:26.25| Show InChI InChI=1S/C29H34ClN3O/c1-18-6-8-20(9-7-18)17-33-25(21-10-11-23(30)19(2)14-21)15-24(32-33)26(34)31-27-28(3,4)22-12-13-29(27,5)16-22/h6-11,14-15,22,27H,12-13,16-17H2,1-5H3,(H,31,34)/t22?,27?,29-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 284: 644-50 (1998)
BindingDB Entry DOI: 10.7270/Q2D50KHT |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 3.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Rattus norvegicus (rat)) | BDBM81780
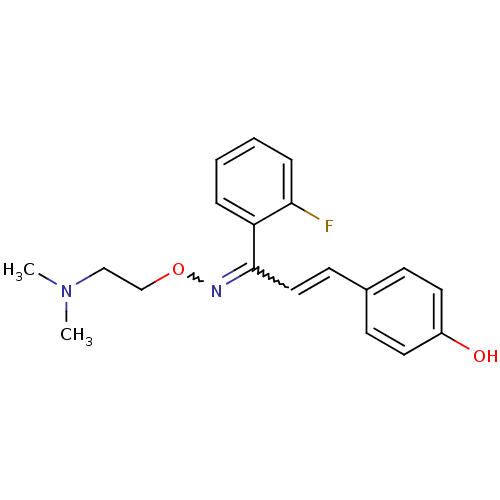
(CAS_6438382 | NSC_6438382 | SR 46349B)Show SMILES CN(C)CCON=C(C=Cc1ccc(O)cc1)c1ccccc1F |w:6.5,8.7| Show InChI InChI=1S/C19H21FN2O2/c1-22(2)13-14-24-21-19(17-5-3-4-6-18(17)20)12-9-15-7-10-16(23)11-8-15/h3-12,23H,13-14H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| PubMed
| 5.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 262: 759-68 (1992)
BindingDB Entry DOI: 10.7270/Q27S7M7Z |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Rattus norvegicus (rat)) | BDBM21395
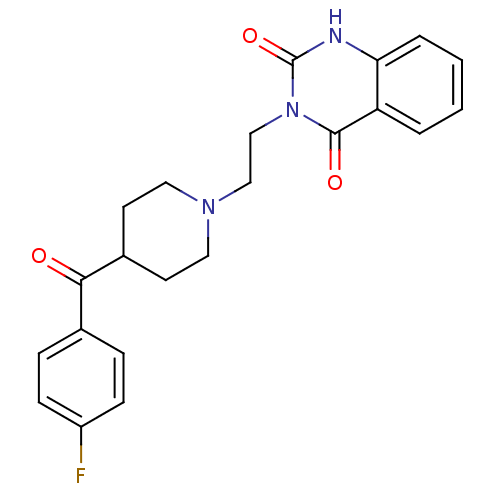
(3-(2-(4-(4-Fluorobenzoyl)piperidinol)ethyl)-2,4(1H...)Show SMILES Fc1ccc(cc1)C(=O)C1CCN(CCn2c(=O)[nH]c3ccccc3c2=O)CC1 Show InChI InChI=1S/C22H22FN3O3/c23-17-7-5-15(6-8-17)20(27)16-9-11-25(12-10-16)13-14-26-21(28)18-3-1-2-4-19(18)24-22(26)29/h1-8,16H,9-14H2,(H,24,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 14.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 262: 759-68 (1992)
BindingDB Entry DOI: 10.7270/Q27S7M7Z |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Rattus norvegicus (rat)) | BDBM50001775
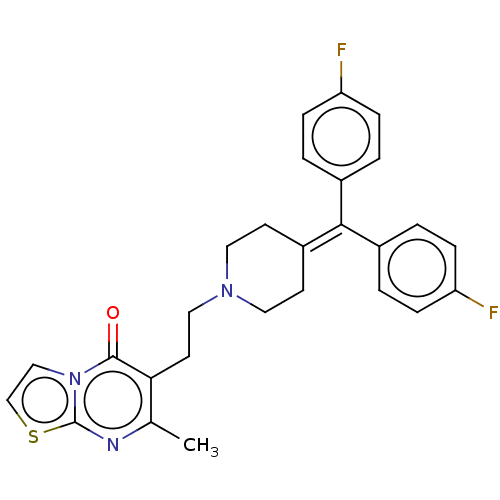
((ritanserin)6-(2-{4-[Bis-(4-fluoro-phenyl)-methyle...)Show SMILES [#6]-c1nc2sccn2c(=O)c1-[#6]-[#6]-[#7]-1-[#6]-[#6]\[#6](-[#6]-[#6]-1)=[#6](\c1ccc(F)cc1)-c1ccc(F)cc1 Show InChI InChI=1S/C27H25F2N3OS/c1-18-24(26(33)32-16-17-34-27(32)30-18)12-15-31-13-10-21(11-14-31)25(19-2-6-22(28)7-3-19)20-4-8-23(29)9-5-20/h2-9,16-17H,10-15H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 58.1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 262: 759-68 (1992)
BindingDB Entry DOI: 10.7270/Q27S7M7Z |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2C
(PIG) | BDBM81780

(CAS_6438382 | NSC_6438382 | SR 46349B)Show SMILES CN(C)CCON=C(C=Cc1ccc(O)cc1)c1ccccc1F |w:6.5,8.7| Show InChI InChI=1S/C19H21FN2O2/c1-22(2)13-14-24-21-19(17-5-3-4-6-18(17)20)12-9-15-7-10-16(23)11-8-15/h3-12,23H,13-14H2,1-2H3 | PDB
Reactome pathway
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| PubMed
| 120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 262: 759-68 (1992)
BindingDB Entry DOI: 10.7270/Q27S7M7Z |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Rattus norvegicus (rat)) | BDBM21280

(5-(4-chloro-3-methylphenyl)-1-[(4-methylphenyl)met...)Show SMILES Cc1ccc(Cn2nc(cc2-c2ccc(Cl)c(C)c2)C(=O)NC2[C@@]3(C)CCC(C3)C2(C)C)cc1 |r,TLB:21:22:28:26.25| Show InChI InChI=1S/C29H34ClN3O/c1-18-6-8-20(9-7-18)17-33-25(21-10-11-23(30)19(2)14-21)15-24(32-33)26(34)31-27-28(3,4)22-12-13-29(27,5)16-22/h6-11,14-15,22,27H,12-13,16-17H2,1-5H3,(H,31,34)/t22?,27?,29-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| PubMed
| 305 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 284: 644-50 (1998)
BindingDB Entry DOI: 10.7270/Q2D50KHT |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Rattus norvegicus (rat)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 349 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM21280

(5-(4-chloro-3-methylphenyl)-1-[(4-methylphenyl)met...)Show SMILES Cc1ccc(Cn2nc(cc2-c2ccc(Cl)c(C)c2)C(=O)NC2[C@@]3(C)CCC(C3)C2(C)C)cc1 |r,TLB:21:22:28:26.25| Show InChI InChI=1S/C29H34ClN3O/c1-18-6-8-20(9-7-18)17-33-25(21-10-11-23(30)19(2)14-21)15-24(32-33)26(34)31-27-28(3,4)22-12-13-29(27,5)16-22/h6-11,14-15,22,27H,12-13,16-17H2,1-5H3,(H,31,34)/t22?,27?,29-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| PubMed
| 437 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 284: 644-50 (1998)
BindingDB Entry DOI: 10.7270/Q2D50KHT |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 2
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 442 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Alpha-2C adrenergic receptor
(RAT) | BDBM81780

(CAS_6438382 | NSC_6438382 | SR 46349B)Show SMILES CN(C)CCON=C(C=Cc1ccc(O)cc1)c1ccccc1F |w:6.5,8.7| Show InChI InChI=1S/C19H21FN2O2/c1-22(2)13-14-24-21-19(17-5-3-4-6-18(17)20)12-9-15-7-10-16(23)11-8-15/h3-12,23H,13-14H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 262: 759-68 (1992)
BindingDB Entry DOI: 10.7270/Q27S7M7Z |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1B
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1D
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2C
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 3A
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 4
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 5A
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Transducer protein HtrVI
(Halobacterium salinarum R1) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | KEGG
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 7
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Mus musculus) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Alpha-1A adrenergic receptor
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Alpha-2A adrenergic receptor
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Alpha-2B adrenergic receptor
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Alpha-2C adrenergic receptor
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Beta-1 adrenergic receptor
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Type-1 angiotensin II receptor
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Type-2 angiotensin II receptor
(RAT) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Gamma-aminobutyric acid receptor subunit alpha-1
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Peripheral-type benzodiazepine receptor-associated protein 1
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Neuromedin-B receptor
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Gastrin-releasing peptide receptor
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Voltage-dependent calcium channel gamma-1 subunit
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Voltage-dependent L-type calcium channel subunit alpha-1C
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Voltage-dependent N-type calcium channel subunit alpha-1B
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Calcitonin gene-related peptide 1
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
Reactome pathway
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Chloride channel protein 1
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M4
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M5
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
D(1A) dopamine receptor
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |
D(3) dopamine receptor
(Homo sapiens (Human)) | BDBM50171290

(5-(4-Bromo-phenyl)-1-(2,4-dichloro-phenyl)-4-ethyl...)Show SMILES CCc1c(nn(c1-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C23H23BrCl2N4O/c1-2-18-21(23(31)28-29-12-4-3-5-13-29)27-30(20-11-10-17(25)14-19(20)26)22(18)15-6-8-16(24)9-7-15/h6-11,14H,2-5,12-13H2,1H3,(H,28,31) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Synthelabo Recherche
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 905-14 (2004)
Article DOI: 10.1124/jpet.104.067884
BindingDB Entry DOI: 10.7270/Q2RR1WTX |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data