

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Prostaglandin G/H synthase 1 (Homo sapiens (Human)) | BDBM13065 (5-(4-chlorophenyl)-1-(4-methoxyphenyl)-3-(trifluor...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Vanderbilt Institute of Chemical Biology Curated by ChEMBL | Assay Description Inhibition of COX1 | J Med Chem 55: 2287-300 (2012) Article DOI: 10.1021/jm201528b BindingDB Entry DOI: 10.7270/Q2WW7JQB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin G/H synthase 2 (Homo sapiens (Human)) | BDBM11639 (4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyraz...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Vanderbilt Institute of Chemical Biology Curated by ChEMBL | Assay Description Inhibition of COX2 | J Med Chem 55: 2287-300 (2012) Article DOI: 10.1021/jm201528b BindingDB Entry DOI: 10.7270/Q2WW7JQB | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Prostaglandin G/H synthase 1 (Homo sapiens (Human)) | BDBM17638 (2-{1-[(4-chlorophenyl)carbonyl]-5-methoxy-2-methyl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
Vanderbilt Institute of Chemical Biology Curated by ChEMBL | Assay Description Inhibition of COX1 | J Med Chem 55: 2287-300 (2012) Article DOI: 10.1021/jm201528b BindingDB Entry DOI: 10.7270/Q2WW7JQB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin G/H synthase 2 (Homo sapiens (Human)) | BDBM11639 (4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyraz...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | n/a | n/a | 54 | n/a | n/a | n/a | n/a | n/a | n/a |
Vanderbilt Institute of Chemical Biology Curated by ChEMBL | Assay Description Inhibition of COX-2 in human HNSCC 1483 cells using [14C] arachidonic acid as substrate preincubated for 30 mins before substrate addition measured a... | J Med Chem 55: 2287-300 (2012) Article DOI: 10.1021/jm201528b BindingDB Entry DOI: 10.7270/Q2WW7JQB | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Prostaglandin G/H synthase 1 (Homo sapiens (Human)) | BDBM50110164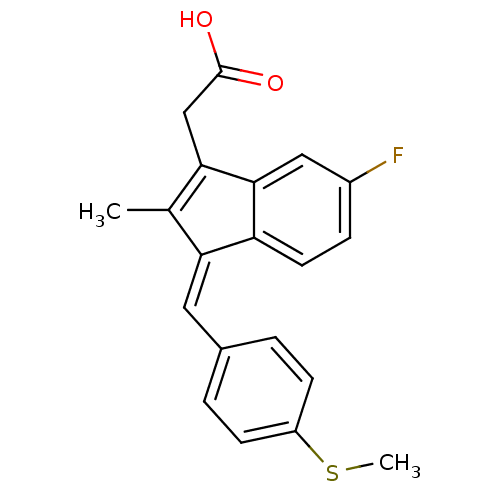 ((Z)-2-(3-(4-(methylthio)benzylidene)-6-fluoro-2-me...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 115 | n/a | n/a | n/a | n/a | n/a | n/a |
Vanderbilt Institute of Chemical Biology Curated by ChEMBL | Assay Description Inhibition of COX1 | J Med Chem 55: 2287-300 (2012) Article DOI: 10.1021/jm201528b BindingDB Entry DOI: 10.7270/Q2WW7JQB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin G/H synthase 1 (Homo sapiens (Human)) | BDBM50272862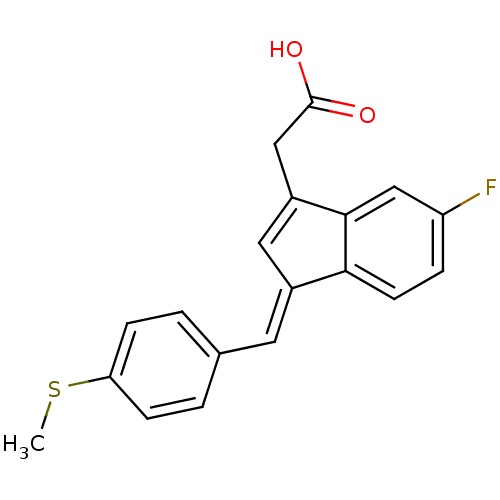 ((E)-2'-des-methyl sulindac sulfide | 2-desmethylsu...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 116 | n/a | n/a | n/a | n/a | n/a | n/a |
Vanderbilt Institute of Chemical Biology Curated by ChEMBL | Assay Description Inhibition of COX-1 in human OVCAR3 cells using [14C] arachidonic acid as substrate preincubated for 30 mins before substrate addition measured after... | J Med Chem 55: 2287-300 (2012) Article DOI: 10.1021/jm201528b BindingDB Entry DOI: 10.7270/Q2WW7JQB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin G/H synthase 2 (Homo sapiens (Human)) | BDBM50110164 ((Z)-2-(3-(4-(methylthio)benzylidene)-6-fluoro-2-me...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a | n/a |
Vanderbilt Institute of Chemical Biology Curated by ChEMBL | Assay Description Inhibition of COX2 | J Med Chem 55: 2287-300 (2012) Article DOI: 10.1021/jm201528b BindingDB Entry DOI: 10.7270/Q2WW7JQB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin G/H synthase 1 (Homo sapiens (Human)) | BDBM13065 (5-(4-chlorophenyl)-1-(4-methoxyphenyl)-3-(trifluor...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 160 | n/a | n/a | n/a | n/a | n/a | n/a |
Vanderbilt Institute of Chemical Biology Curated by ChEMBL | Assay Description Inhibition of COX-1 in human OVCAR3 cells using [14C] arachidonic acid as substrate preincubated for 30 mins before substrate addition measured after... | J Med Chem 55: 2287-300 (2012) Article DOI: 10.1021/jm201528b BindingDB Entry DOI: 10.7270/Q2WW7JQB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin G/H synthase 1 (Homo sapiens (Human)) | BDBM50273065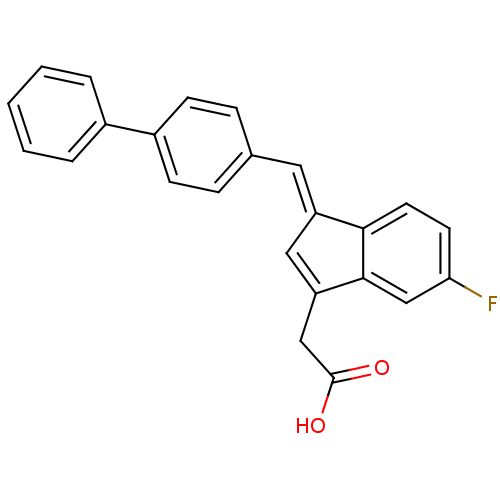 ((E)-2-(1-(Biphenyl-4-ylmethylene)-5-fluoro-1H-inde...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
Vanderbilt Institute of Chemical Biology Curated by ChEMBL | Assay Description Inhibition of COX-1 in human OVCAR3 cells using [14C] arachidonic acid as substrate preincubated for 30 mins before substrate addition measured after... | J Med Chem 55: 2287-300 (2012) Article DOI: 10.1021/jm201528b BindingDB Entry DOI: 10.7270/Q2WW7JQB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin G/H synthase 2 (Homo sapiens (Human)) | BDBM22369 (4-(4-methanesulfonylphenyl)-3-phenyl-2,5-dihydrofu...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 340 | n/a | n/a | n/a | n/a | n/a | n/a |
Vanderbilt Institute of Chemical Biology Curated by ChEMBL | Assay Description Inhibition of COX2 | J Med Chem 55: 2287-300 (2012) Article DOI: 10.1021/jm201528b BindingDB Entry DOI: 10.7270/Q2WW7JQB | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Prostaglandin G/H synthase 1 (Homo sapiens (Human)) | BDBM50388954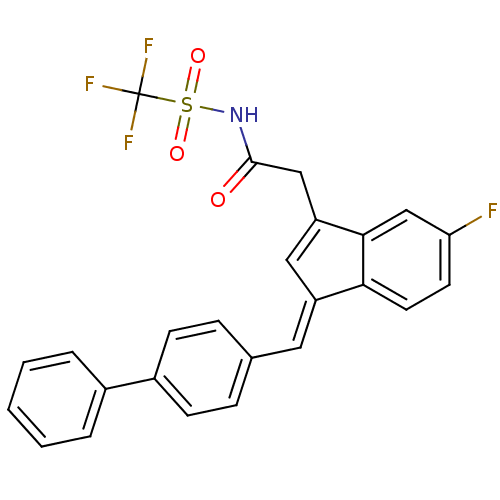 (CHEMBL2063559) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 470 | n/a | n/a | n/a | n/a | n/a | n/a |
Vanderbilt Institute of Chemical Biology Curated by ChEMBL | Assay Description Inhibition of ovine COX-1 using [14C] arachidonic acid as substrate preincubated for 17 mins before substrate addition measured after 3 mins by thin-... | J Med Chem 55: 2287-300 (2012) Article DOI: 10.1021/jm201528b BindingDB Entry DOI: 10.7270/Q2WW7JQB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin G/H synthase 1 (Homo sapiens (Human)) | BDBM50388954 (CHEMBL2063559) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 495 | n/a | n/a | n/a | n/a | n/a | n/a |
Vanderbilt Institute of Chemical Biology Curated by ChEMBL | Assay Description Inhibition of COX-1 in human OVCAR3 cells using [14C] arachidonic acid as substrate preincubated for 30 mins before substrate addition measured after... | J Med Chem 55: 2287-300 (2012) Article DOI: 10.1021/jm201528b BindingDB Entry DOI: 10.7270/Q2WW7JQB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin G/H synthase 1 (Homo sapiens (Human)) | BDBM50273065 ((E)-2-(1-(Biphenyl-4-ylmethylene)-5-fluoro-1H-inde...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 570 | n/a | n/a | n/a | n/a | n/a | n/a |
Vanderbilt Institute of Chemical Biology Curated by ChEMBL | Assay Description Inhibition of ovine COX-1 using [14C] arachidonic acid as substrate preincubated for 17 mins before substrate addition measured after 3 mins by thin-... | J Med Chem 55: 2287-300 (2012) Article DOI: 10.1021/jm201528b BindingDB Entry DOI: 10.7270/Q2WW7JQB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin G/H synthase 2 (Homo sapiens (Human)) | BDBM17638 (2-{1-[(4-chlorophenyl)carbonyl]-5-methoxy-2-methyl...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | n/a | n/a | 750 | n/a | n/a | n/a | n/a | n/a | n/a |
Vanderbilt Institute of Chemical Biology Curated by ChEMBL | Assay Description Inhibition of COX2 | J Med Chem 55: 2287-300 (2012) Article DOI: 10.1021/jm201528b BindingDB Entry DOI: 10.7270/Q2WW7JQB | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Prostaglandin G/H synthase 1 (Homo sapiens (Human)) | BDBM50388956 (CHEMBL2063543) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Vanderbilt Institute of Chemical Biology Curated by ChEMBL | Assay Description Inhibition of ovine COX-1 using [14C] arachidonic acid as substrate preincubated for 17 mins before substrate addition measured after 3 mins by thin-... | J Med Chem 55: 2287-300 (2012) Article DOI: 10.1021/jm201528b BindingDB Entry DOI: 10.7270/Q2WW7JQB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin G/H synthase 1 (Homo sapiens (Human)) | BDBM50272862 ((E)-2'-des-methyl sulindac sulfide | 2-desmethylsu...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Vanderbilt Institute of Chemical Biology Curated by ChEMBL | Assay Description Inhibition of COX1 | J Med Chem 55: 2287-300 (2012) Article DOI: 10.1021/jm201528b BindingDB Entry DOI: 10.7270/Q2WW7JQB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin G/H synthase 1 (Homo sapiens (Human)) | BDBM50272862 ((E)-2'-des-methyl sulindac sulfide | 2-desmethylsu...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Vanderbilt Institute of Chemical Biology Curated by ChEMBL | Assay Description Inhibition of ovine COX-1 using [14C] arachidonic acid as substrate preincubated for 17 mins before substrate addition measured after 3 mins by thin-... | J Med Chem 55: 2287-300 (2012) Article DOI: 10.1021/jm201528b BindingDB Entry DOI: 10.7270/Q2WW7JQB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin G/H synthase 1 (Homo sapiens (Human)) | BDBM50388955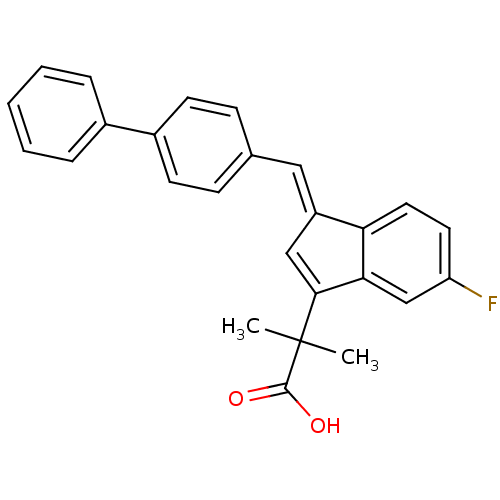 (CHEMBL2063547) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Vanderbilt Institute of Chemical Biology Curated by ChEMBL | Assay Description Inhibition of ovine COX-1 using [14C] arachidonic acid as substrate preincubated for 17 mins before substrate addition measured after 3 mins by thin-... | J Med Chem 55: 2287-300 (2012) Article DOI: 10.1021/jm201528b BindingDB Entry DOI: 10.7270/Q2WW7JQB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin G/H synthase 2 (Mus musculus (Mouse)) | BDBM50273065 ((E)-2-(1-(Biphenyl-4-ylmethylene)-5-fluoro-1H-inde...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Vanderbilt Institute of Chemical Biology Curated by ChEMBL | Assay Description Inhibition of mouse COX-2 using [14C] arachidonic acid as substrate preincubated for 17 mins before substrate addition measured after 3 mins by thin-... | J Med Chem 55: 2287-300 (2012) Article DOI: 10.1021/jm201528b BindingDB Entry DOI: 10.7270/Q2WW7JQB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin G/H synthase 2 (Homo sapiens (Human)) | BDBM50272862 ((E)-2'-des-methyl sulindac sulfide | 2-desmethylsu...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Vanderbilt Institute of Chemical Biology Curated by ChEMBL | Assay Description Inhibition of COX2 | J Med Chem 55: 2287-300 (2012) Article DOI: 10.1021/jm201528b BindingDB Entry DOI: 10.7270/Q2WW7JQB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin G/H synthase 2 (Homo sapiens (Human)) | BDBM13065 (5-(4-chlorophenyl)-1-(4-methoxyphenyl)-3-(trifluor...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 6.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Vanderbilt Institute of Chemical Biology Curated by ChEMBL | Assay Description Inhibition of COX2 | J Med Chem 55: 2287-300 (2012) Article DOI: 10.1021/jm201528b BindingDB Entry DOI: 10.7270/Q2WW7JQB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin G/H synthase 1 (Homo sapiens (Human)) | BDBM11639 (4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyraz...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Vanderbilt Institute of Chemical Biology Curated by ChEMBL | Assay Description Inhibition of COX1 | J Med Chem 55: 2287-300 (2012) Article DOI: 10.1021/jm201528b BindingDB Entry DOI: 10.7270/Q2WW7JQB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin G/H synthase 1 (Homo sapiens (Human)) | BDBM22369 (4-(4-methanesulfonylphenyl)-3-phenyl-2,5-dihydrofu...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 2.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Vanderbilt Institute of Chemical Biology Curated by ChEMBL | Assay Description Inhibition of COX1 | J Med Chem 55: 2287-300 (2012) Article DOI: 10.1021/jm201528b BindingDB Entry DOI: 10.7270/Q2WW7JQB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||