 Found 9130 hits with Last Name = 'heer' and Initial = 'j'
Found 9130 hits with Last Name = 'heer' and Initial = 'j' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Coagulation factor X
(Homo sapiens (Human)) | BDBM17135
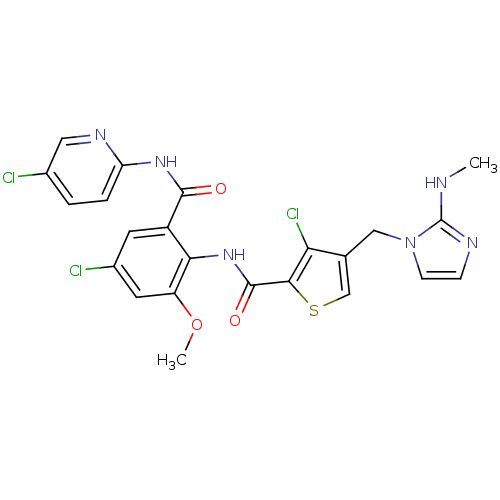
(3-chloro-N-{4-chloro-2-[(5-chloropyridin-2-yl)carb...)Show SMILES CNc1nccn1Cc1csc(C(=O)Nc2c(OC)cc(Cl)cc2C(=O)Nc2ccc(Cl)cn2)c1Cl Show InChI InChI=1S/C23H19Cl3N6O3S/c1-27-23-28-5-6-32(23)10-12-11-36-20(18(12)26)22(34)31-19-15(7-14(25)8-16(19)35-2)21(33)30-17-4-3-13(24)9-29-17/h3-9,11H,10H2,1-2H3,(H,27,28)(H,31,34)(H,29,30,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 0.00500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
J Med Chem 53: 6243-74 (2010)
Article DOI: 10.1021/jm100146h
BindingDB Entry DOI: 10.7270/Q2CR5VBB |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12751

(1-(3-carbamimidoylphenyl)-3-methyl-N-[4-(2-sulfamo...)Show SMILES Cc1cc(C(=O)Nc2ccc(cc2)-c2ccccc2S(N)(=O)=O)n(n1)-c1cccc(c1)C(N)=N Show InChI InChI=1S/C24H22N6O3S/c1-15-13-21(30(29-15)19-6-4-5-17(14-19)23(25)26)24(31)28-18-11-9-16(10-12-18)20-7-2-3-8-22(20)34(27,32)33/h2-14H,1H3,(H3,25,26)(H,28,31)(H2,27,32,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
J Med Chem 53: 6243-74 (2010)
Article DOI: 10.1021/jm100146h
BindingDB Entry DOI: 10.7270/Q2CR5VBB |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50374877
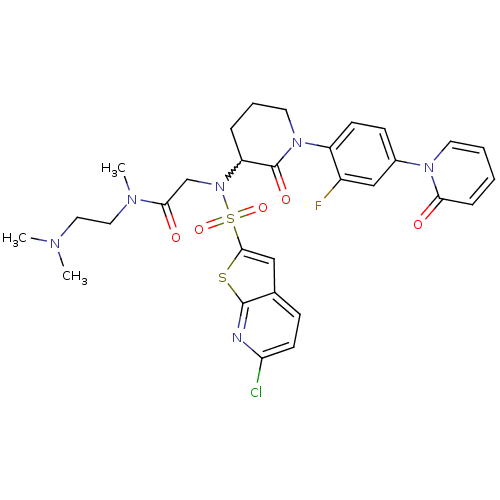
(CHEMBL270221)Show SMILES CN(C)CCN(C)C(=O)CN(C1CCCN(C1=O)c1ccc(cc1F)-n1ccccc1=O)S(=O)(=O)c1cc2ccc(Cl)nc2s1 |w:11.10| Show InChI InChI=1S/C30H32ClFN6O5S2/c1-34(2)15-16-35(3)27(40)19-38(45(42,43)28-17-20-9-12-25(31)33-29(20)44-28)24-7-6-14-37(30(24)41)23-11-10-21(18-22(23)32)36-13-5-4-8-26(36)39/h4-5,8-13,17-18,24H,6-7,14-16,19H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 2428-33 (2008)
Article DOI: 10.1016/j.bmcl.2008.02.054
BindingDB Entry DOI: 10.7270/Q2RX9CZ8 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12751

(1-(3-carbamimidoylphenyl)-3-methyl-N-[4-(2-sulfamo...)Show SMILES Cc1cc(C(=O)Nc2ccc(cc2)-c2ccccc2S(N)(=O)=O)n(n1)-c1cccc(c1)C(N)=N Show InChI InChI=1S/C24H22N6O3S/c1-15-13-21(30(29-15)19-6-4-5-17(14-19)23(25)26)24(31)28-18-11-9-16(10-12-18)20-7-2-3-8-22(20)34(27,32)33/h2-14H,1H3,(H3,25,26)(H,28,31)(H2,27,32,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0130 | -61.5 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter... |
Bioorg Med Chem Lett 14: 5263-7 (2004)
Article DOI: 10.1016/j.bmcl.2004.08.034
BindingDB Entry DOI: 10.7270/Q2TH8JX0 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50374879
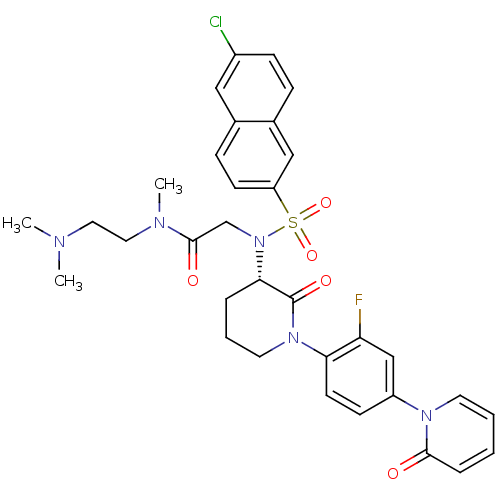
(CHEMBL401958)Show SMILES CN(C)CCN(C)C(=O)CN([C@H]1CCCN(C1=O)c1ccc(cc1F)-n1ccccc1=O)S(=O)(=O)c1ccc2cc(Cl)ccc2c1 Show InChI InChI=1S/C33H35ClFN5O5S/c1-36(2)17-18-37(3)32(42)22-40(46(44,45)27-13-10-23-19-25(34)11-9-24(23)20-27)30-7-6-16-39(33(30)43)29-14-12-26(21-28(29)35)38-15-5-4-8-31(38)41/h4-5,8-15,19-21,30H,6-7,16-18,22H2,1-3H3/t30-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 2428-33 (2008)
Article DOI: 10.1016/j.bmcl.2008.02.054
BindingDB Entry DOI: 10.7270/Q2RX9CZ8 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50374879

(CHEMBL401958)Show SMILES CN(C)CCN(C)C(=O)CN([C@H]1CCCN(C1=O)c1ccc(cc1F)-n1ccccc1=O)S(=O)(=O)c1ccc2cc(Cl)ccc2c1 Show InChI InChI=1S/C33H35ClFN5O5S/c1-36(2)17-18-37(3)32(42)22-40(46(44,45)27-13-10-23-19-25(34)11-9-24(23)20-27)30-7-6-16-39(33(30)43)29-14-12-26(21-28(29)35)38-15-5-4-8-31(38)41/h4-5,8-15,19-21,30H,6-7,16-18,22H2,1-3H3/t30-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 2428-33 (2008)
Article DOI: 10.1016/j.bmcl.2008.02.054
BindingDB Entry DOI: 10.7270/Q2RX9CZ8 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50374878
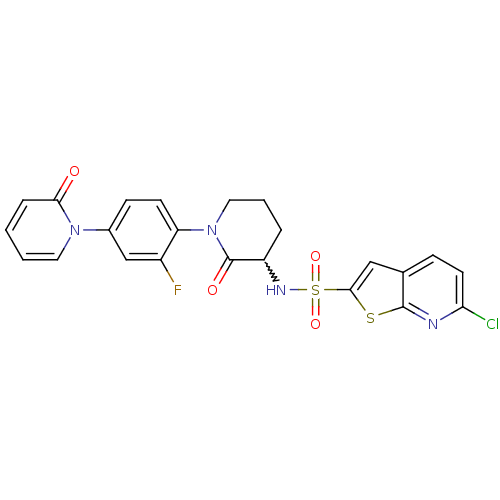
(CHEMBL270862)Show SMILES Fc1cc(ccc1N1CCCC(NS(=O)(=O)c2cc3ccc(Cl)nc3s2)C1=O)-n1ccccc1=O |w:11.12| Show InChI InChI=1S/C23H18ClFN4O4S2/c24-19-9-6-14-12-21(34-22(14)26-19)35(32,33)27-17-4-3-11-29(23(17)31)18-8-7-15(13-16(18)25)28-10-2-1-5-20(28)30/h1-2,5-10,12-13,17,27H,3-4,11H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 2428-33 (2008)
Article DOI: 10.1016/j.bmcl.2008.02.054
BindingDB Entry DOI: 10.7270/Q2RX9CZ8 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12754
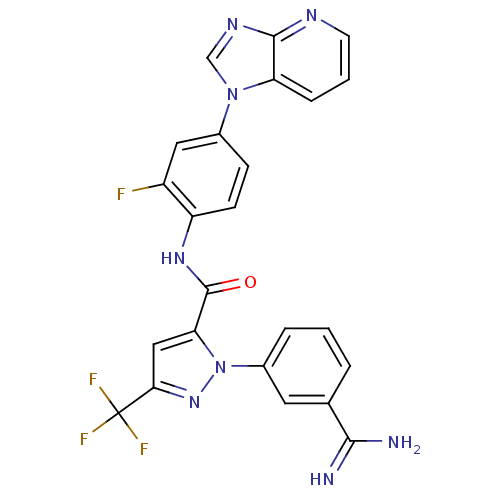
(1-(3-carbamimidoylphenyl)-N-(2-fluoro-4-{1H-imidaz...)Show SMILES NC(=N)c1cccc(c1)-n1nc(cc1C(=O)Nc1ccc(cc1F)-n1cnc2ncccc12)C(F)(F)F Show InChI InChI=1S/C24H16F4N8O/c25-16-10-14(35-12-32-22-18(35)5-2-8-31-22)6-7-17(16)33-23(37)19-11-20(24(26,27)28)34-36(19)15-4-1-3-13(9-15)21(29)30/h1-12H,(H3,29,30)(H,33,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0240 | -60.0 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter... |
Bioorg Med Chem Lett 14: 5263-7 (2004)
Article DOI: 10.1016/j.bmcl.2004.08.034
BindingDB Entry DOI: 10.7270/Q2TH8JX0 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12757
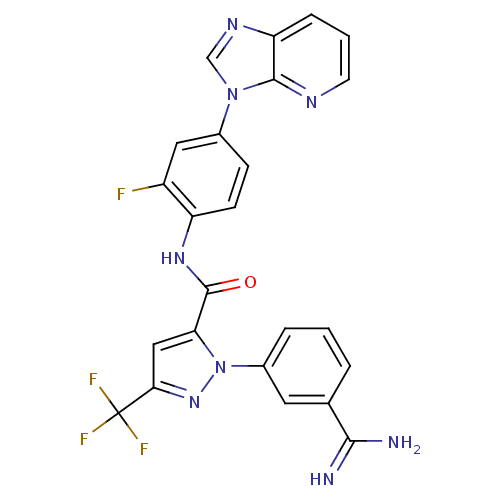
(1-(3-carbamimidoylphenyl)-N-(2-fluoro-4-{3H-imidaz...)Show SMILES NC(=N)c1cccc(c1)-n1nc(cc1C(=O)Nc1ccc(cc1F)-n1cnc2cccnc12)C(F)(F)F Show InChI InChI=1S/C24H16F4N8O/c25-16-10-14(35-12-32-18-5-2-8-31-22(18)35)6-7-17(16)33-23(37)19-11-20(24(26,27)28)34-36(19)15-4-1-3-13(9-15)21(29)30/h1-12H,(H3,29,30)(H,33,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0280 | -59.6 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter... |
Bioorg Med Chem Lett 14: 5263-7 (2004)
Article DOI: 10.1016/j.bmcl.2004.08.034
BindingDB Entry DOI: 10.7270/Q2TH8JX0 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12693
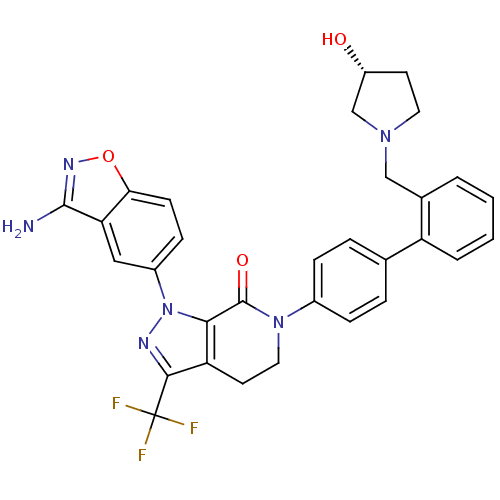
(1-(3-amino-1,2-benzoxazol-5-yl)-6-[4-(2-{[(3R)-3-h...)Show SMILES Nc1noc2ccc(cc12)-n1nc(c2CCN(C(=O)c12)c1ccc(cc1)-c1ccccc1CN1CC[C@@H](O)C1)C(F)(F)F |r| Show InChI InChI=1S/C31H27F3N6O3/c32-31(33,34)28-24-12-14-39(30(42)27(24)40(36-28)21-9-10-26-25(15-21)29(35)37-43-26)20-7-5-18(6-8-20)23-4-2-1-3-19(23)16-38-13-11-22(41)17-38/h1-10,15,22,41H,11-14,16-17H2,(H2,35,37)/t22-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
J Med Chem 53: 6243-74 (2010)
Article DOI: 10.1021/jm100146h
BindingDB Entry DOI: 10.7270/Q2CR5VBB |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12755
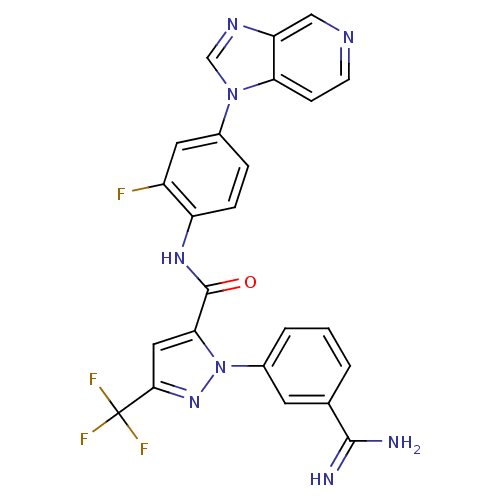
(1-(3-carbamimidoylphenyl)-N-(2-fluoro-4-{1H-imidaz...)Show SMILES NC(=N)c1cccc(c1)-n1nc(cc1C(=O)Nc1ccc(cc1F)-n1cnc2cnccc12)C(F)(F)F Show InChI InChI=1S/C24H16F4N8O/c25-16-9-14(35-12-32-18-11-31-7-6-19(18)35)4-5-17(16)33-23(37)20-10-21(24(26,27)28)34-36(20)15-3-1-2-13(8-15)22(29)30/h1-12H,(H3,29,30)(H,33,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0360 | -59.0 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter... |
Bioorg Med Chem Lett 14: 5263-7 (2004)
Article DOI: 10.1016/j.bmcl.2004.08.034
BindingDB Entry DOI: 10.7270/Q2TH8JX0 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50374876
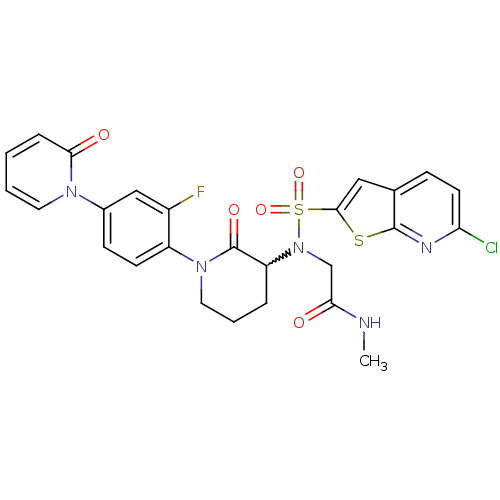
(CHEMBL270034)Show SMILES CNC(=O)CN(C1CCCN(C1=O)c1ccc(cc1F)-n1ccccc1=O)S(=O)(=O)c1cc2ccc(Cl)nc2s1 |w:6.5| Show InChI InChI=1S/C26H23ClFN5O5S2/c1-29-22(34)15-33(40(37,38)24-13-16-7-10-21(27)30-25(16)39-24)20-5-4-12-32(26(20)36)19-9-8-17(14-18(19)28)31-11-3-2-6-23(31)35/h2-3,6-11,13-14,20H,4-5,12,15H2,1H3,(H,29,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 2428-33 (2008)
Article DOI: 10.1016/j.bmcl.2008.02.054
BindingDB Entry DOI: 10.7270/Q2RX9CZ8 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50374871
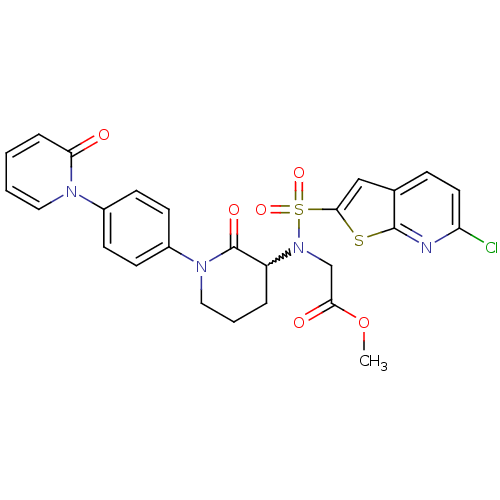
(CHEMBL258274)Show SMILES COC(=O)CN(C1CCCN(C1=O)c1ccc(cc1)-n1ccccc1=O)S(=O)(=O)c1cc2ccc(Cl)nc2s1 |w:6.5| Show InChI InChI=1S/C26H23ClN4O6S2/c1-37-23(33)16-31(39(35,36)24-15-17-7-12-21(27)28-25(17)38-24)20-5-4-14-30(26(20)34)19-10-8-18(9-11-19)29-13-3-2-6-22(29)32/h2-3,6-13,15,20H,4-5,14,16H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0430 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 2428-33 (2008)
Article DOI: 10.1016/j.bmcl.2008.02.054
BindingDB Entry DOI: 10.7270/Q2RX9CZ8 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50249295
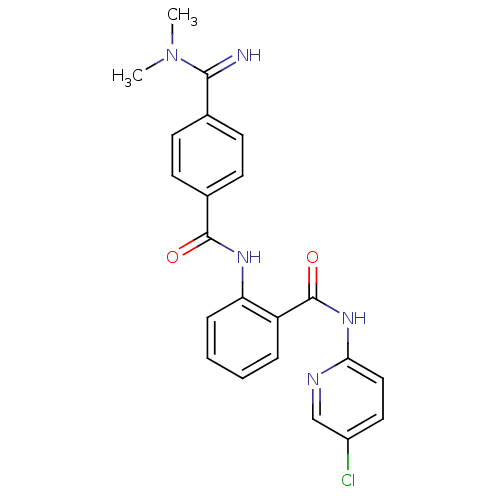
(CHEMBL471725 | N-(5-chloropyridin-2-yl)-2-(4-(N,N-...)Show SMILES CN(C)C(=N)c1ccc(cc1)C(=O)Nc1ccccc1C(=O)Nc1ccc(Cl)cn1 Show InChI InChI=1S/C22H20ClN5O2/c1-28(2)20(24)14-7-9-15(10-8-14)21(29)26-18-6-4-3-5-17(18)22(30)27-19-12-11-16(23)13-25-19/h3-13,24H,1-2H3,(H,26,29)(H,25,27,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0440 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
J Med Chem 53: 6243-74 (2010)
Article DOI: 10.1021/jm100146h
BindingDB Entry DOI: 10.7270/Q2CR5VBB |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50193861
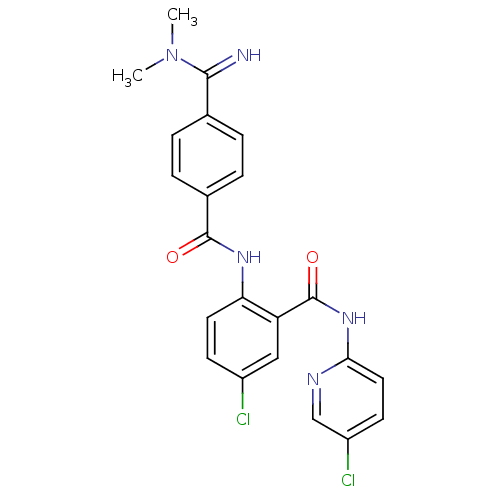
(5-chloro-N-(5-chloro-pyridin-2-yl)-2-[4-(N,N-dimet...)Show SMILES CN(C)C(=N)c1ccc(cc1)C(=O)Nc1ccc(Cl)cc1C(=O)Nc1ccc(Cl)cn1 Show InChI InChI=1S/C22H19Cl2N5O2/c1-29(2)20(25)13-3-5-14(6-4-13)21(30)27-18-9-7-15(23)11-17(18)22(31)28-19-10-8-16(24)12-26-19/h3-12,25H,1-2H3,(H,27,30)(H,26,28,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0440 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
J Med Chem 53: 6243-74 (2010)
Article DOI: 10.1021/jm100146h
BindingDB Entry DOI: 10.7270/Q2CR5VBB |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12756
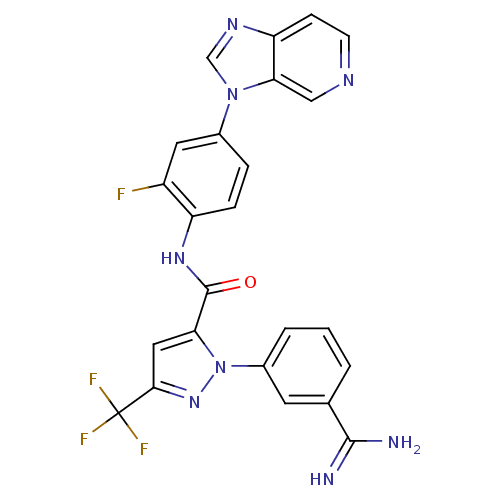
(1-(3-carbamimidoylphenyl)-N-(2-fluoro-4-{3H-imidaz...)Show SMILES NC(=N)c1cccc(c1)-n1nc(cc1C(=O)Nc1ccc(cc1F)-n1cnc2ccncc12)C(F)(F)F Show InChI InChI=1S/C24H16F4N8O/c25-16-9-14(35-12-32-18-6-7-31-11-20(18)35)4-5-17(16)33-23(37)19-10-21(24(26,27)28)34-36(19)15-3-1-2-13(8-15)22(29)30/h1-12H,(H3,29,30)(H,33,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0440 | -58.5 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter... |
Bioorg Med Chem Lett 14: 5263-7 (2004)
Article DOI: 10.1016/j.bmcl.2004.08.034
BindingDB Entry DOI: 10.7270/Q2TH8JX0 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50374875
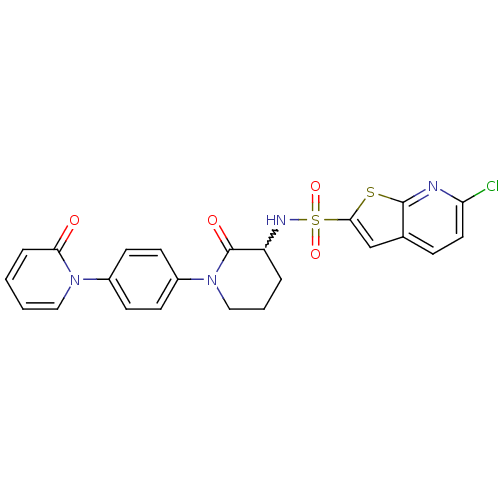
(CHEMBL269955)Show SMILES Clc1ccc2cc(sc2n1)S(=O)(=O)NC1CCCN(C1=O)c1ccc(cc1)-n1ccccc1=O |w:14.15| Show InChI InChI=1S/C23H19ClN4O4S2/c24-19-11-6-15-14-21(33-22(15)25-19)34(31,32)26-18-4-3-13-28(23(18)30)17-9-7-16(8-10-17)27-12-2-1-5-20(27)29/h1-2,5-12,14,18,26H,3-4,13H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 2428-33 (2008)
Article DOI: 10.1016/j.bmcl.2008.02.054
BindingDB Entry DOI: 10.7270/Q2RX9CZ8 |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50419334
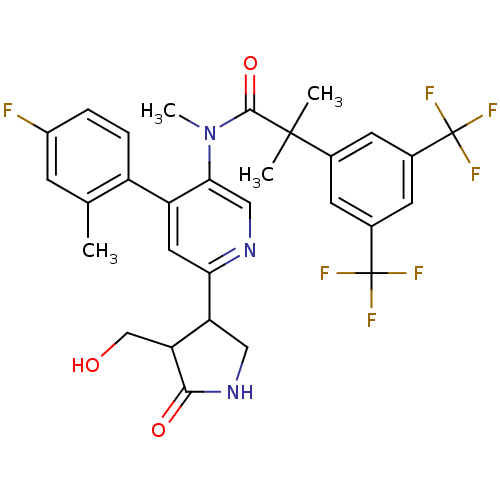
(CHEMBL1911965 | CHEMBL1911967)Show SMILES CN(C(=O)C(C)(C)c1cc(cc(c1)C(F)(F)F)C(F)(F)F)c1cnc(cc1-c1ccc(F)cc1C)C1CNC(=O)C1CO Show InChI InChI=1S/C30H28F7N3O3/c1-15-7-19(31)5-6-20(15)21-11-24(22-12-39-26(42)23(22)14-41)38-13-25(21)40(4)27(43)28(2,3)16-8-17(29(32,33)34)10-18(9-16)30(35,36)37/h5-11,13,22-23,41H,12,14H2,1-4H3,(H,39,42) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0501 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Medicines Research Centre
Curated by ChEMBL
| Assay Description
Antagonist activity at human recombinant NK1 receptor expressed in human U20S cells assessed as effect on substance P-induced intracellular calcium l... |
Bioorg Med Chem Lett 21: 6899-904 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.116
BindingDB Entry DOI: 10.7270/Q24X592P |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50328717

(5-Chloro-N-(5-chloro-pyridin-2-yl)-3-methoxy-2-[4-...)Show SMILES COc1cc(Cl)cc(C(=O)Nc2ccc(Cl)cn2)c1NC(=O)c1ccc(cc1)N1CCCCC1=O Show InChI InChI=1S/C25H22Cl2N4O4/c1-35-20-13-17(27)12-19(25(34)29-21-10-7-16(26)14-28-21)23(20)30-24(33)15-5-8-18(9-6-15)31-11-3-2-4-22(31)32/h5-10,12-14H,2-4,11H2,1H3,(H,30,33)(H,28,29,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0570 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
J Med Chem 53: 6243-74 (2010)
Article DOI: 10.1021/jm100146h
BindingDB Entry DOI: 10.7270/Q2CR5VBB |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50289438
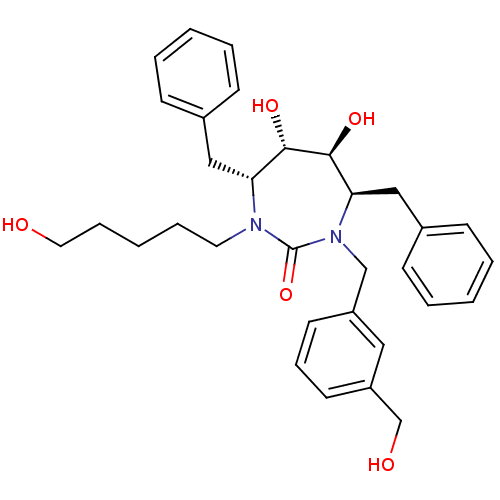
((4R,5S,6S,7R)-4,7-Dibenzyl-5,6-dihydroxy-1-(3-hydr...)Show SMILES OCCCCCN1[C@H](Cc2ccccc2)[C@H](O)[C@@H](O)[C@@H](Cc2ccccc2)N(Cc2cccc(CO)c2)C1=O Show InChI InChI=1S/C32H40N2O5/c35-18-9-3-8-17-33-28(20-24-11-4-1-5-12-24)30(37)31(38)29(21-25-13-6-2-7-14-25)34(32(33)39)22-26-15-10-16-27(19-26)23-36/h1-2,4-7,10-16,19,28-31,35-38H,3,8-9,17-18,20-23H2/t28-,29-,30+,31+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.0580 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 7: 1365-1370 (1997)
Article DOI: 10.1016/S0960-894X(97)00165-0
BindingDB Entry DOI: 10.7270/Q21G0M7B |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12766
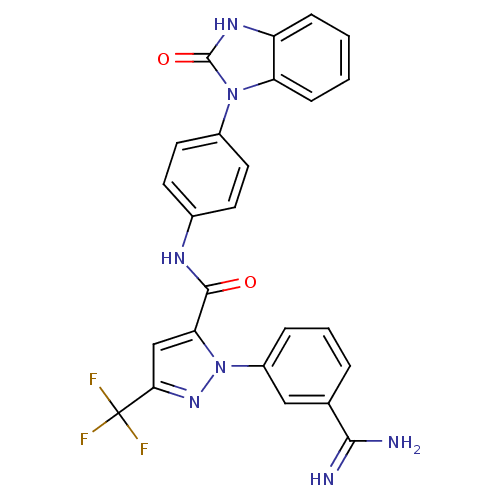
(1-(3-carbamimidoylphenyl)-N-[4-(2-oxo-2,7a-dihydro...)Show SMILES NC(=N)c1cccc(c1)-n1nc(cc1C(=O)Nc1ccc(cc1)-n1c2ccccc2[nH]c1=O)C(F)(F)F Show InChI InChI=1S/C25H18F3N7O2/c26-25(27,28)21-13-20(35(33-21)17-5-3-4-14(12-17)22(29)30)23(36)31-15-8-10-16(11-9-15)34-19-7-2-1-6-18(19)32-24(34)37/h1-13H,(H3,29,30)(H,31,36)(H,32,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0580 | -57.8 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter... |
Bioorg Med Chem Lett 14: 5263-7 (2004)
Article DOI: 10.1016/j.bmcl.2004.08.034
BindingDB Entry DOI: 10.7270/Q2TH8JX0 |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50419337
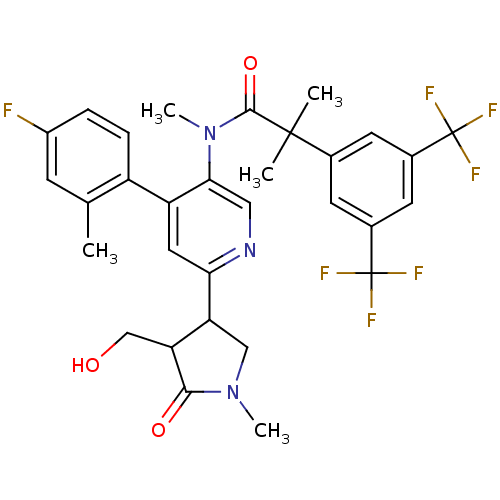
(CHEMBL1911968)Show SMILES CN(C(=O)C(C)(C)c1cc(cc(c1)C(F)(F)F)C(F)(F)F)c1cnc(cc1-c1ccc(F)cc1C)C1CN(C)C(=O)C1CO Show InChI InChI=1S/C31H30F7N3O3/c1-16-8-20(32)6-7-21(16)22-12-25(23-14-40(4)27(43)24(23)15-42)39-13-26(22)41(5)28(44)29(2,3)17-9-18(30(33,34)35)11-19(10-17)31(36,37)38/h6-13,23-24,42H,14-15H2,1-5H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0631 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Medicines Research Centre
Curated by ChEMBL
| Assay Description
Antagonist activity at human recombinant NK1 receptor expressed in human U20S cells assessed as effect on substance P-induced intracellular calcium l... |
Bioorg Med Chem Lett 21: 6899-904 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.116
BindingDB Entry DOI: 10.7270/Q24X592P |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50419332
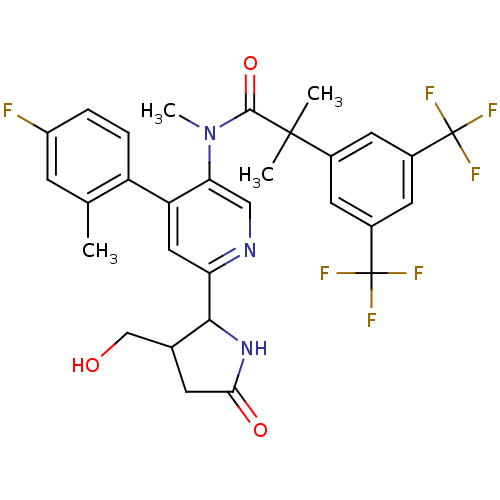
(CHEMBL1911964)Show SMILES CN(C(=O)C(C)(C)c1cc(cc(c1)C(F)(F)F)C(F)(F)F)c1cnc(cc1-c1ccc(F)cc1C)C1NC(=O)CC1CO Show InChI InChI=1S/C30H28F7N3O3/c1-15-7-20(31)5-6-21(15)22-12-23(26-16(14-41)8-25(42)39-26)38-13-24(22)40(4)27(43)28(2,3)17-9-18(29(32,33)34)11-19(10-17)30(35,36)37/h5-7,9-13,16,26,41H,8,14H2,1-4H3,(H,39,42) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.0794 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Medicines Research Centre
Curated by ChEMBL
| Assay Description
Antagonist activity at human recombinant NK1 receptor expressed in human U20S cells assessed as effect on substance P-induced intracellular calcium l... |
Bioorg Med Chem Lett 21: 6899-904 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.116
BindingDB Entry DOI: 10.7270/Q24X592P |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12763
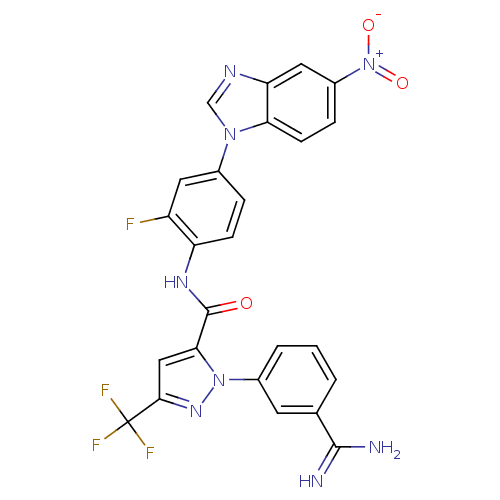
(1-(3-carbamimidoylphenyl)-N-[2-fluoro-4-(5-nitro-1...)Show SMILES NC(=N)c1cccc(c1)-n1nc(cc1C(=O)Nc1ccc(cc1F)-n1cnc2cc(ccc12)[N+]([O-])=O)C(F)(F)F Show InChI InChI=1S/C25H16F4N8O3/c26-17-9-14(35-12-32-19-10-16(37(39)40)5-7-20(19)35)4-6-18(17)33-24(38)21-11-22(25(27,28)29)34-36(21)15-3-1-2-13(8-15)23(30)31/h1-12H,(H3,30,31)(H,33,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0800 | -57.1 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter... |
Bioorg Med Chem Lett 14: 5263-7 (2004)
Article DOI: 10.1016/j.bmcl.2004.08.034
BindingDB Entry DOI: 10.7270/Q2TH8JX0 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM19023

(1-(4-methoxyphenyl)-7-oxo-6-[4-(2-oxopiperidin-1-y...)Show SMILES COc1ccc(cc1)-n1nc(C(N)=O)c2CCN(C(=O)c12)c1ccc(cc1)N1CCCCC1=O Show InChI InChI=1S/C25H25N5O4/c1-34-19-11-9-18(10-12-19)30-23-20(22(27-30)24(26)32)13-15-29(25(23)33)17-7-5-16(6-8-17)28-14-3-2-4-21(28)31/h5-12H,2-4,13-15H2,1H3,(H2,26,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
J Med Chem 53: 6243-74 (2010)
Article DOI: 10.1021/jm100146h
BindingDB Entry DOI: 10.7270/Q2CR5VBB |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50289443
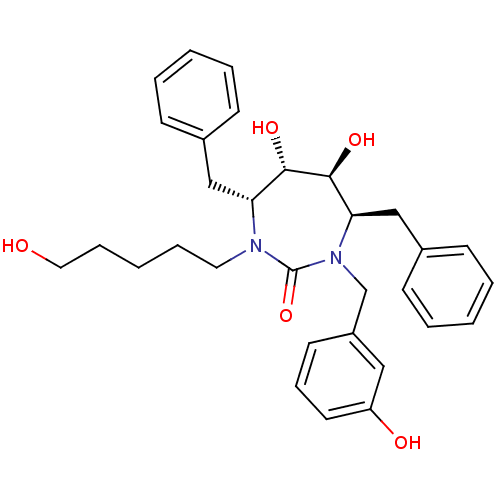
((4R,5S,6S,7R)-4,7-Dibenzyl-5,6-dihydroxy-1-(3-hydr...)Show SMILES OCCCCCN1[C@H](Cc2ccccc2)[C@H](O)[C@@H](O)[C@@H](Cc2ccccc2)N(Cc2cccc(O)c2)C1=O Show InChI InChI=1S/C31H38N2O5/c34-18-9-3-8-17-32-27(20-23-11-4-1-5-12-23)29(36)30(37)28(21-24-13-6-2-7-14-24)33(31(32)38)22-25-15-10-16-26(35)19-25/h1-2,4-7,10-16,19,27-30,34-37H,3,8-9,17-18,20-22H2/t27-,28-,29+,30+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.0810 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 7: 1365-1370 (1997)
Article DOI: 10.1016/S0960-894X(97)00165-0
BindingDB Entry DOI: 10.7270/Q21G0M7B |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12767
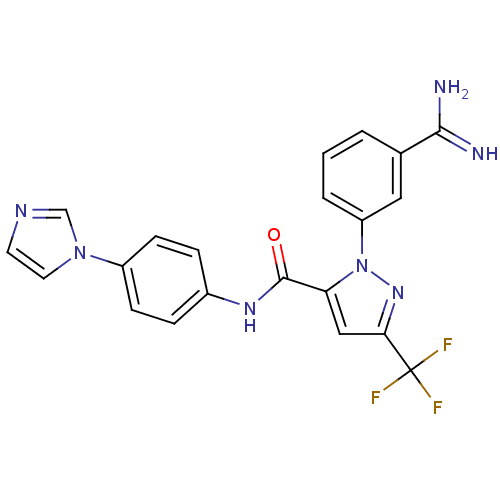
(1-(3-carbamimidoylphenyl)-N-[4-(1H-imidazol-1-yl)p...)Show SMILES NC(=N)c1cccc(c1)-n1nc(cc1C(=O)Nc1ccc(cc1)-n1ccnc1)C(F)(F)F Show InChI InChI=1S/C21H16F3N7O/c22-21(23,24)18-11-17(31(29-18)16-3-1-2-13(10-16)19(25)26)20(32)28-14-4-6-15(7-5-14)30-9-8-27-12-30/h1-12H,(H3,25,26)(H,28,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0910 | -56.7 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter... |
Bioorg Med Chem Lett 14: 5263-7 (2004)
Article DOI: 10.1016/j.bmcl.2004.08.034
BindingDB Entry DOI: 10.7270/Q2TH8JX0 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12752
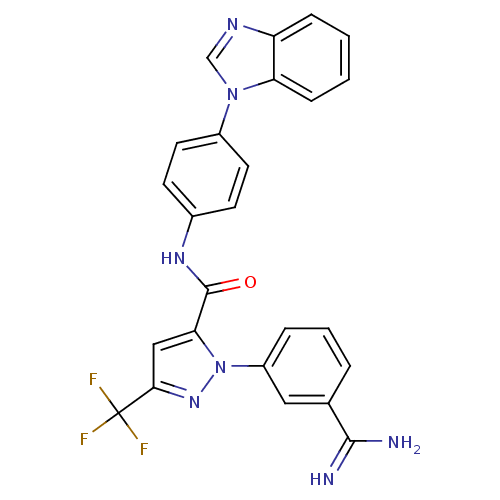
(N-[4-(1H-1,3-benzodiazol-1-yl)phenyl]-1-(3-carbami...)Show SMILES NC(=N)c1cccc(c1)-n1nc(cc1C(=O)Nc1ccc(cc1)-n1cnc2ccccc12)C(F)(F)F Show InChI InChI=1S/C25H18F3N7O/c26-25(27,28)22-13-21(35(33-22)18-5-3-4-15(12-18)23(29)30)24(36)32-16-8-10-17(11-9-16)34-14-31-19-6-1-2-7-20(19)34/h1-14H,(H3,29,30)(H,32,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0940 | -56.7 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter... |
Bioorg Med Chem Lett 14: 5263-7 (2004)
Article DOI: 10.1016/j.bmcl.2004.08.034
BindingDB Entry DOI: 10.7270/Q2TH8JX0 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12753
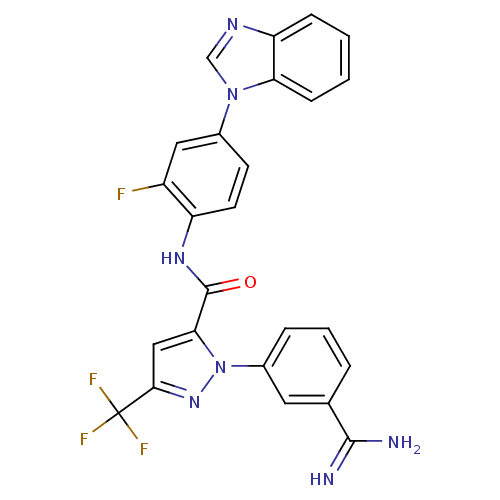
(N-[4-(1H-1,3-benzodiazol-1-yl)-2-fluorophenyl]-1-(...)Show SMILES NC(=N)c1cccc(c1)-n1nc(cc1C(=O)Nc1ccc(cc1F)-n1cnc2ccccc12)C(F)(F)F Show InChI InChI=1S/C25H17F4N7O/c26-17-11-15(35-13-32-19-6-1-2-7-20(19)35)8-9-18(17)33-24(37)21-12-22(25(27,28)29)34-36(21)16-5-3-4-14(10-16)23(30)31/h1-13H,(H3,30,31)(H,33,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.100 | -56.5 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter... |
Bioorg Med Chem Lett 14: 5263-7 (2004)
Article DOI: 10.1016/j.bmcl.2004.08.034
BindingDB Entry DOI: 10.7270/Q2TH8JX0 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50328716

(CHEMBL1270651 | N-(4-chloro-2-(5-chloropyridin-2-y...)Show SMILES CN(C)Cc1ccccc1-c1ccc(cc1)C(=O)Nc1ccc(Cl)cc1C(=O)Nc1ccc(Cl)cn1 Show InChI InChI=1S/C28H24Cl2N4O2/c1-34(2)17-20-5-3-4-6-23(20)18-7-9-19(10-8-18)27(35)32-25-13-11-21(29)15-24(25)28(36)33-26-14-12-22(30)16-31-26/h3-16H,17H2,1-2H3,(H,32,35)(H,31,33,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
J Med Chem 53: 6243-74 (2010)
Article DOI: 10.1021/jm100146h
BindingDB Entry DOI: 10.7270/Q2CR5VBB |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50084664

(({3-(5-Carbamimidoyl-2-hydroxy-phenoxy)-2,6-difluo...)Show SMILES CN(CC(O)=O)c1c(F)c(Oc2cccc(c2)C2=NCCN2C)cc(Oc2cc(ccc2O)C(N)=N)c1F |t:18| Show InChI InChI=1S/C26H25F2N5O5/c1-32-9-8-31-26(32)15-4-3-5-16(10-15)37-19-12-20(23(28)24(22(19)27)33(2)13-21(35)36)38-18-11-14(25(29)30)6-7-17(18)34/h3-7,10-12,34H,8-9,13H2,1-2H3,(H3,29,30)(H,35,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
J Med Chem 53: 6243-74 (2010)
Article DOI: 10.1021/jm100146h
BindingDB Entry DOI: 10.7270/Q2CR5VBB |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50289441
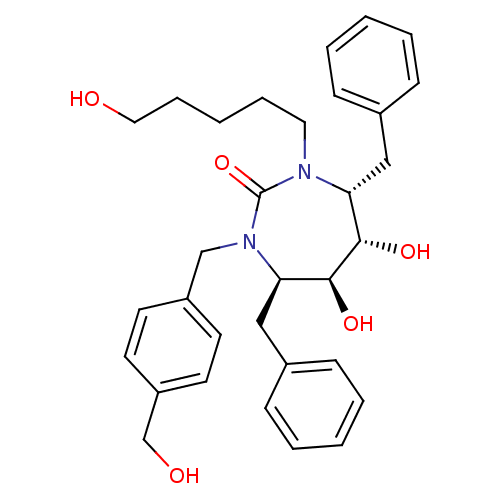
((4R,5S,6S,7R)-4,7-Dibenzyl-5,6-dihydroxy-1-(4-hydr...)Show SMILES OCCCCCN1[C@H](Cc2ccccc2)[C@H](O)[C@@H](O)[C@@H](Cc2ccccc2)N(Cc2ccc(CO)cc2)C1=O Show InChI InChI=1S/C32H40N2O5/c35-19-9-3-8-18-33-28(20-24-10-4-1-5-11-24)30(37)31(38)29(21-25-12-6-2-7-13-25)34(32(33)39)22-26-14-16-27(23-36)17-15-26/h1-2,4-7,10-17,28-31,35-38H,3,8-9,18-23H2/t28-,29-,30+,31+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 7: 1365-1370 (1997)
Article DOI: 10.1016/S0960-894X(97)00165-0
BindingDB Entry DOI: 10.7270/Q21G0M7B |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50419333
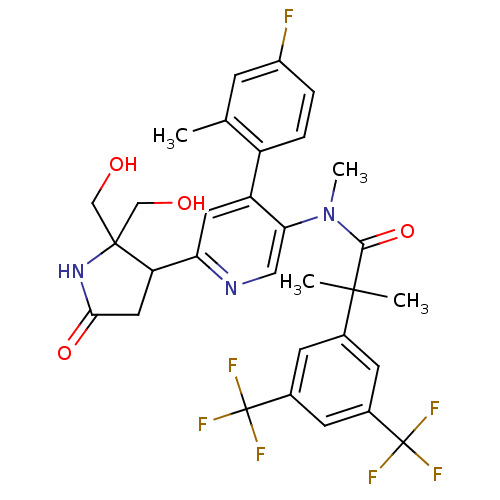
(CHEMBL1911970)Show SMILES CN(C(=O)C(C)(C)c1cc(cc(c1)C(F)(F)F)C(F)(F)F)c1cnc(cc1-c1ccc(F)cc1C)C1CC(=O)NC1(CO)CO Show InChI InChI=1S/C31H30F7N3O4/c1-16-7-20(32)5-6-21(16)22-11-24(23-12-26(44)40-29(23,14-42)15-43)39-13-25(22)41(4)27(45)28(2,3)17-8-18(30(33,34)35)10-19(9-17)31(36,37)38/h5-11,13,23,42-43H,12,14-15H2,1-4H3,(H,40,44) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Medicines Research Centre
Curated by ChEMBL
| Assay Description
Antagonist activity at human recombinant NK1 receptor expressed in human U20S cells assessed as effect on substance P-induced intracellular calcium l... |
Bioorg Med Chem Lett 21: 6899-904 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.116
BindingDB Entry DOI: 10.7270/Q24X592P |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50374874
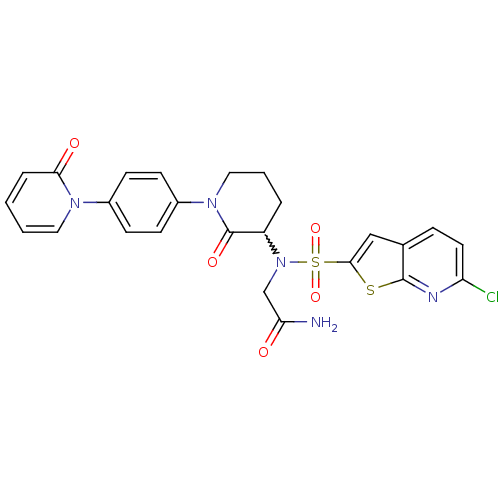
(CHEMBL256820)Show SMILES NC(=O)CN(C1CCCN(C1=O)c1ccc(cc1)-n1ccccc1=O)S(=O)(=O)c1cc2ccc(Cl)nc2s1 |w:5.4| Show InChI InChI=1S/C25H22ClN5O5S2/c26-20-11-6-16-14-23(37-24(16)28-20)38(35,36)31(15-21(27)32)19-4-3-13-30(25(19)34)18-9-7-17(8-10-18)29-12-2-1-5-22(29)33/h1-2,5-12,14,19H,3-4,13,15H2,(H2,27,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 2428-33 (2008)
Article DOI: 10.1016/j.bmcl.2008.02.054
BindingDB Entry DOI: 10.7270/Q2RX9CZ8 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50374880

(CHEMBL271904)Show SMILES Fc1cc(ccc1N1CCCC(NS(=O)(=O)c2ccc3cc(Cl)ccc3c2)C1=O)-n1ccccc1=O |w:11.12| Show InChI InChI=1S/C26H21ClFN3O4S/c27-19-8-6-18-15-21(10-7-17(18)14-19)36(34,35)29-23-4-3-13-31(26(23)33)24-11-9-20(16-22(24)28)30-12-2-1-5-25(30)32/h1-2,5-12,14-16,23,29H,3-4,13H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 2428-33 (2008)
Article DOI: 10.1016/j.bmcl.2008.02.054
BindingDB Entry DOI: 10.7270/Q2RX9CZ8 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50249298
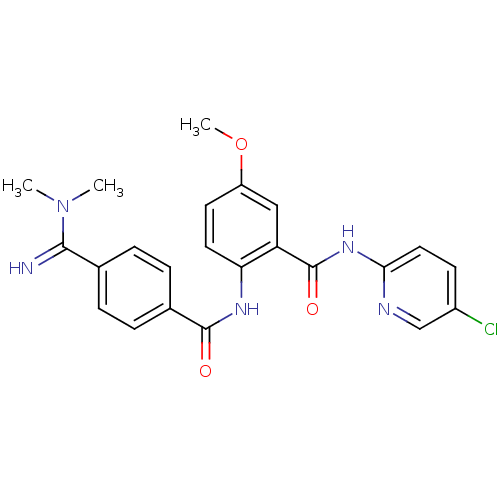
(BEVYXXA | CHEMBL512351 | N-(5-chloropyridin-2-yl)-...)Show SMILES COc1ccc(NC(=O)c2ccc(cc2)C(=N)N(C)C)c(c1)C(=O)Nc1ccc(Cl)cn1 Show InChI InChI=1S/C23H22ClN5O3/c1-29(2)21(25)14-4-6-15(7-5-14)22(30)27-19-10-9-17(32-3)12-18(19)23(31)28-20-11-8-16(24)13-26-20/h4-13,25H,1-3H3,(H,27,30)(H,26,28,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
J Med Chem 53: 6243-74 (2010)
Article DOI: 10.1021/jm100146h
BindingDB Entry DOI: 10.7270/Q2CR5VBB |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12762
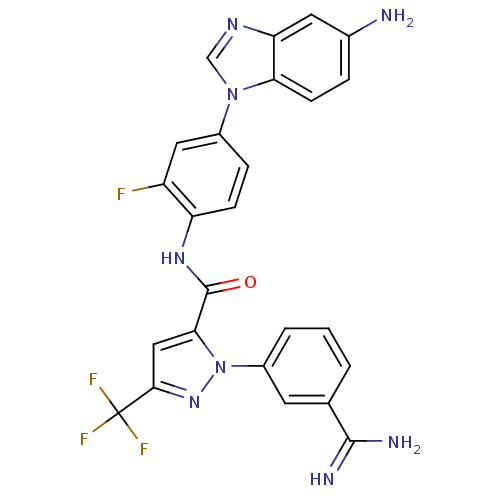
(N-[4-(5-amino-1H-1,3-benzodiazol-1-yl)-2-fluorophe...)Show SMILES NC(=N)c1cccc(c1)-n1nc(cc1C(=O)Nc1ccc(cc1F)-n1cnc2cc(N)ccc12)C(F)(F)F Show InChI InChI=1S/C25H18F4N8O/c26-17-10-15(36-12-33-19-9-14(30)4-7-20(19)36)5-6-18(17)34-24(38)21-11-22(25(27,28)29)35-37(21)16-3-1-2-13(8-16)23(31)32/h1-12H,30H2,(H3,31,32)(H,34,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.120 | -56.1 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter... |
Bioorg Med Chem Lett 14: 5263-7 (2004)
Article DOI: 10.1016/j.bmcl.2004.08.034
BindingDB Entry DOI: 10.7270/Q2TH8JX0 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50374873

(CHEMBL442457)Show SMILES OC(=O)CN(C1CCCN(C1=O)c1ccc(cc1)-n1ccccc1=O)S(=O)(=O)c1cc2ccc(Cl)nc2s1 |w:5.4| Show InChI InChI=1S/C25H21ClN4O6S2/c26-20-11-6-16-14-23(37-24(16)27-20)38(35,36)30(15-22(32)33)19-4-3-13-29(25(19)34)18-9-7-17(8-10-18)28-12-2-1-5-21(28)31/h1-2,5-12,14,19H,3-4,13,15H2,(H,32,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 2428-33 (2008)
Article DOI: 10.1016/j.bmcl.2008.02.054
BindingDB Entry DOI: 10.7270/Q2RX9CZ8 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50087533
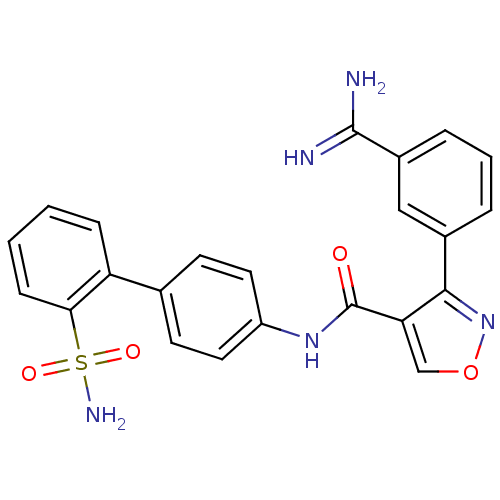
(3-(3-Carbamimidoyl-phenyl)-isoxazole-4-carboxylic ...)Show SMILES NC(=N)c1cccc(c1)-c1nocc1C(=O)Nc1ccc(cc1)-c1ccccc1S(N)(=O)=O Show InChI InChI=1S/C23H19N5O4S/c24-22(25)16-5-3-4-15(12-16)21-19(13-32-28-21)23(29)27-17-10-8-14(9-11-17)18-6-1-2-7-20(18)33(26,30)31/h1-13H,(H3,24,25)(H,27,29)(H2,26,30,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
J Med Chem 53: 6243-74 (2010)
Article DOI: 10.1021/jm100146h
BindingDB Entry DOI: 10.7270/Q2CR5VBB |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12657
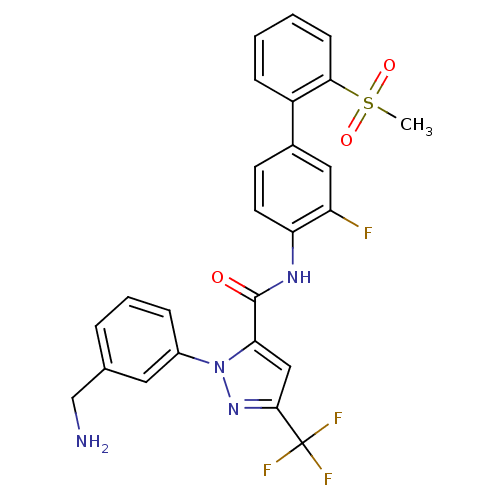
(1-[3-(Aminomethyl)phenyl]-N-[3-fluoro-2-(methylsul...)Show SMILES CS(=O)(=O)c1ccccc1-c1ccc(NC(=O)c2cc(nn2-c2cccc(CN)c2)C(F)(F)F)c(F)c1 Show InChI InChI=1S/C25H20F4N4O3S/c1-37(35,36)22-8-3-2-7-18(22)16-9-10-20(19(26)12-16)31-24(34)21-13-23(25(27,28)29)32-33(21)17-6-4-5-15(11-17)14-30/h2-13H,14,30H2,1H3,(H,31,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
J Med Chem 53: 6243-74 (2010)
Article DOI: 10.1021/jm100146h
BindingDB Entry DOI: 10.7270/Q2CR5VBB |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50419334

(CHEMBL1911965 | CHEMBL1911967)Show SMILES CN(C(=O)C(C)(C)c1cc(cc(c1)C(F)(F)F)C(F)(F)F)c1cnc(cc1-c1ccc(F)cc1C)C1CNC(=O)C1CO Show InChI InChI=1S/C30H28F7N3O3/c1-15-7-19(31)5-6-20(15)21-11-24(22-12-39-26(42)23(22)14-41)38-13-25(21)40(4)27(43)28(2,3)16-8-17(29(32,33)34)10-18(9-16)30(35,36)37/h5-11,13,22-23,41H,12,14H2,1-4H3,(H,39,42) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.158 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Medicines Research Centre
Curated by ChEMBL
| Assay Description
Antagonist activity at human recombinant NK1 receptor expressed in human U20S cells assessed as effect on substance P-induced intracellular calcium l... |
Bioorg Med Chem Lett 21: 6899-904 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.116
BindingDB Entry DOI: 10.7270/Q24X592P |
More data for this
Ligand-Target Pair | |
Coagulation factor XI
(Homo sapiens (Human)) | BDBM50230322
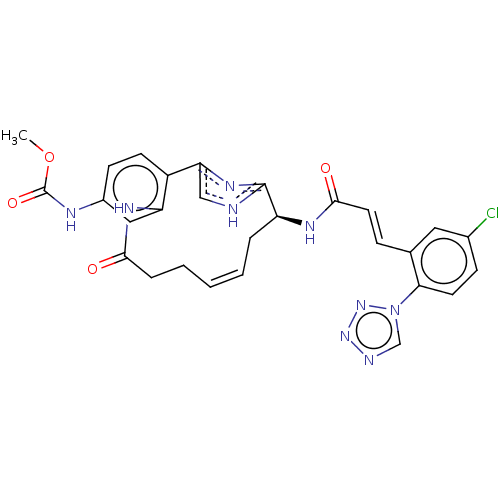
(CHEMBL4071545)Show SMILES COC(=O)Nc1ccc2-c3c[nH]c(n3)[C@H](C\C=C\CCC(=O)Nc2c1)NC(=O)\C=C\c1cc(Cl)ccc1-n1cnnn1 |r,t:17| Show InChI InChI=1S/C28H26ClN9O4/c1-42-28(41)32-19-9-10-20-22(14-19)34-25(39)6-4-2-3-5-21(27-30-15-23(20)35-27)33-26(40)12-7-17-13-18(29)8-11-24(17)38-16-31-36-37-38/h2-3,7-16,21H,4-6H2,1H3,(H,30,35)(H,32,41)(H,33,40)(H,34,39)/b3-2+,12-7+/t21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of activated human coagulation factor 11 using pyroGlu-Pro-Arg-pNA as substrate incubated for 10 to 120 mins by spectrofluorometric method |
Bioorg Med Chem Lett 29: (2019)
Article DOI: 10.1016/j.bmcl.2019.08.008
BindingDB Entry DOI: 10.7270/Q23F4T2C |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12864
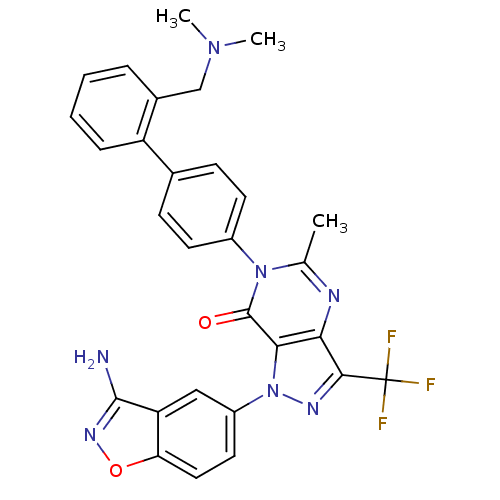
(1-(3-amino-1,2-benzoxazol-5-yl)-6-(4-{2-[(dimethyl...)Show SMILES CN(C)Cc1ccccc1-c1ccc(cc1)-n1c(C)nc2c(nn(-c3ccc4onc(N)c4c3)c2c1=O)C(F)(F)F Show InChI InChI=1S/C29H24F3N7O2/c1-16-34-24-25(39(35-26(24)29(30,31)32)20-12-13-23-22(14-20)27(33)36-41-23)28(40)38(16)19-10-8-17(9-11-19)21-7-5-4-6-18(21)15-37(2)3/h4-14H,15H2,1-3H3,(H2,33,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
J Med Chem 53: 6243-74 (2010)
Article DOI: 10.1021/jm100146h
BindingDB Entry DOI: 10.7270/Q2CR5VBB |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12733

(3-[6-(4-{2-[(dimethylamino)methyl]phenyl}phenyl)-7...)Show SMILES CN(C)Cc1ccccc1-c1ccc(cc1)N1CCc2c(nn(c2C1=O)-c1cccc(c1)C(N)=O)C(F)(F)F Show InChI InChI=1S/C29H26F3N5O2/c1-35(2)17-20-6-3-4-9-23(20)18-10-12-21(13-11-18)36-15-14-24-25(28(36)39)37(34-26(24)29(30,31)32)22-8-5-7-19(16-22)27(33)38/h3-13,16H,14-15,17H2,1-2H3,(H2,33,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
J Med Chem 53: 6243-74 (2010)
Article DOI: 10.1021/jm100146h
BindingDB Entry DOI: 10.7270/Q2CR5VBB |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12676

(1-(3-Aminobenzisoxazol-5-yl)-3-trifluoromethyl-N-[...)Show SMILES CN(C)Cc1nccn1-c1ccc(NC(=O)c2cc(nn2-c2ccc3onc(N)c3c2)C(F)(F)F)c(F)c1 Show InChI InChI=1S/C24H20F4N8O2/c1-34(2)12-21-30-7-8-35(21)13-3-5-17(16(25)10-13)31-23(37)18-11-20(24(26,27)28)32-36(18)14-4-6-19-15(9-14)22(29)33-38-19/h3-11H,12H2,1-2H3,(H2,29,33)(H,31,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
J Med Chem 53: 6243-74 (2010)
Article DOI: 10.1021/jm100146h
BindingDB Entry DOI: 10.7270/Q2CR5VBB |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12769
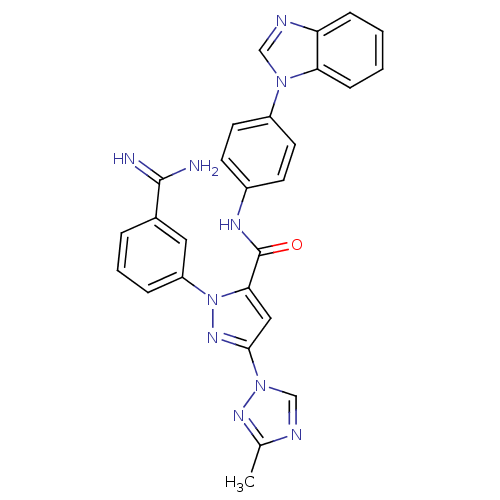
(N-[4-(1H-1,3-benzodiazol-1-yl)phenyl]-1-(3-carbami...)Show SMILES Cc1ncn(n1)-c1cc(C(=O)Nc2ccc(cc2)-n2cnc3ccccc23)n(n1)-c1cccc(c1)C(N)=N Show InChI InChI=1S/C27H22N10O/c1-17-30-16-36(33-17)25-14-24(37(34-25)21-6-4-5-18(13-21)26(28)29)27(38)32-19-9-11-20(12-10-19)35-15-31-22-7-2-3-8-23(22)35/h2-16H,1H3,(H3,28,29)(H,32,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.190 | -54.9 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter... |
Bioorg Med Chem Lett 14: 5263-7 (2004)
Article DOI: 10.1016/j.bmcl.2004.08.034
BindingDB Entry DOI: 10.7270/Q2TH8JX0 |
More data for this
Ligand-Target Pair | |
Coagulation factor XI
(Homo sapiens (Human)) | BDBM50448583
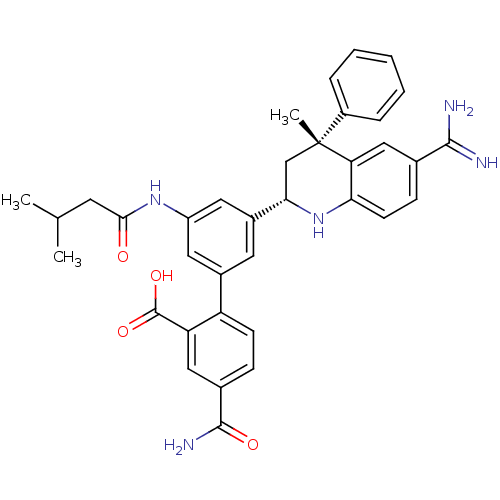
(CHEMBL3127491)Show SMILES CC(C)CC(=O)Nc1cc(cc(c1)-c1ccc(cc1C(O)=O)C(N)=O)[C@@H]1C[C@](C)(c2ccccc2)c2cc(ccc2N1)C(N)=N |r| Show InChI InChI=1S/C36H37N5O4/c1-20(2)13-32(42)40-26-15-23(27-11-9-22(34(39)43)17-28(27)35(44)45)14-24(16-26)31-19-36(3,25-7-5-4-6-8-25)29-18-21(33(37)38)10-12-30(29)41-31/h4-12,14-18,20,31,41H,13,19H2,1-3H3,(H3,37,38)(H2,39,43)(H,40,42)(H,44,45)/t31-,36+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Inhibition of human coagulation factor 11a using p-nitroaniline as substrate assessed as substrate hydrolysis by spectrophotometrically |
J Med Chem 57: 9915-32 (2014)
Article DOI: 10.1021/jm5010607
BindingDB Entry DOI: 10.7270/Q2RV0Q9S |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12772
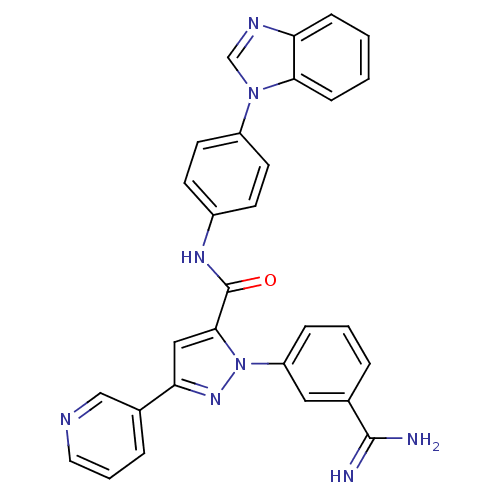
(N-[4-(1H-1,3-benzodiazol-1-yl)phenyl]-1-(3-carbami...)Show SMILES NC(=N)c1cccc(c1)-n1nc(cc1C(=O)Nc1ccc(cc1)-n1cnc2ccccc12)-c1cccnc1 Show InChI InChI=1S/C29H22N8O/c30-28(31)19-5-3-7-23(15-19)37-27(16-25(35-37)20-6-4-14-32-17-20)29(38)34-21-10-12-22(13-11-21)36-18-33-24-8-1-2-9-26(24)36/h1-18H,(H3,30,31)(H,34,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | -54.8 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter... |
Bioorg Med Chem Lett 14: 5263-7 (2004)
Article DOI: 10.1016/j.bmcl.2004.08.034
BindingDB Entry DOI: 10.7270/Q2TH8JX0 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50228676
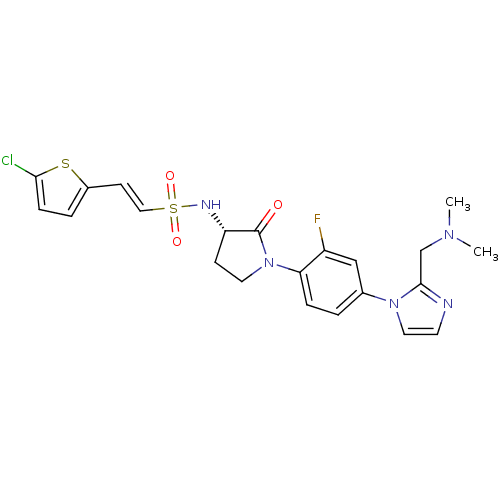
((S)-2-(5-chlorothiophen-2-yl)-N-(1-(4-(2-((dimethy...)Show SMILES CN(C)Cc1nccn1-c1ccc(N2CC[C@H](NS(=O)(=O)\C=C\c3ccc(Cl)s3)C2=O)c(F)c1 Show InChI InChI=1S/C22H23ClFN5O3S2/c1-27(2)14-21-25-9-11-28(21)15-3-5-19(17(24)13-15)29-10-7-18(22(29)30)26-34(31,32)12-8-16-4-6-20(23)33-16/h3-6,8-9,11-13,18,26H,7,10,14H2,1-2H3/b12-8+/t18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
J Med Chem 53: 6243-74 (2010)
Article DOI: 10.1021/jm100146h
BindingDB Entry DOI: 10.7270/Q2CR5VBB |
More data for this
Ligand-Target Pair | |
Coagulation factor XI
(Homo sapiens (Human)) | BDBM50448581
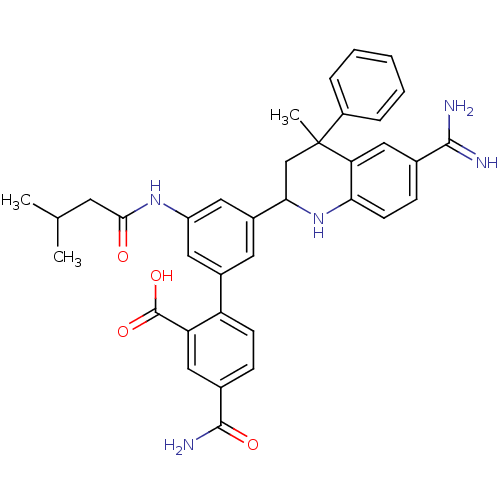
(CHEMBL3127463)Show SMILES CC(C)CC(=O)Nc1cc(cc(c1)-c1ccc(cc1C(O)=O)C(N)=O)C1CC(C)(c2ccccc2)c2cc(ccc2N1)C(N)=N Show InChI InChI=1S/C36H37N5O4/c1-20(2)13-32(42)40-26-15-23(27-11-9-22(34(39)43)17-28(27)35(44)45)14-24(16-26)31-19-36(3,25-7-5-4-6-8-25)29-18-21(33(37)38)10-12-30(29)41-31/h4-12,14-18,20,31,41H,13,19H2,1-3H3,(H3,37,38)(H2,39,43)(H,40,42)(H,44,45) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Binding affinity to human factor 11a assessed as release of p-nitroaniline after 10 to 120 mins by spectrophotometric analysis |
J Med Chem 57: 955-69 (2014)
Article DOI: 10.1021/jm401670x
BindingDB Entry DOI: 10.7270/Q2G44RTQ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data