 Found 29 hits with Last Name = 'kassekert' and Initial = 'la'
Found 29 hits with Last Name = 'kassekert' and Initial = 'la' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM50498523
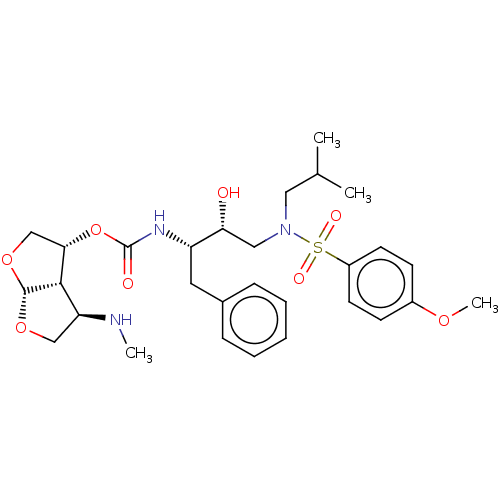
(CHEMBL3605643)Show SMILES [H][C@]12OC[C@H](NC)[C@@]1([H])[C@H](CO2)OC(=O)N[C@@H](Cc1ccccc1)[C@H](O)CN(CC(C)C)S(=O)(=O)c1ccc(OC)cc1 |r| Show InChI InChI=1S/C29H41N3O8S/c1-19(2)15-32(41(35,36)22-12-10-21(37-4)11-13-22)16-25(33)23(14-20-8-6-5-7-9-20)31-29(34)40-26-18-39-28-27(26)24(30-3)17-38-28/h5-13,19,23-28,30,33H,14-18H2,1-4H3,(H,31,34)/t23-,24-,25+,26-,27-,28+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| 0.00150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 protease |
J Med Chem 58: 6994-7006 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00900
BindingDB Entry DOI: 10.7270/Q2862KFS |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM50498524
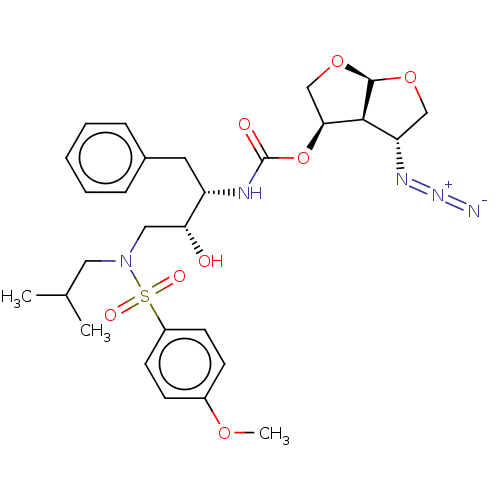
(CHEMBL3605638)Show SMILES [H][C@@]12OC[C@H](OC(=O)N[C@@H](Cc3ccccc3)[C@H](O)CN(CC(C)C)S(=O)(=O)c3ccc(OC)cc3)[C@]1([H])[C@H](CO2)N=[N+]=[N-] |r| Show InChI InChI=1S/C28H37N5O8S/c1-18(2)14-33(42(36,37)21-11-9-20(38-3)10-12-21)15-24(34)22(13-19-7-5-4-6-8-19)30-28(35)41-25-17-40-27-26(25)23(16-39-27)31-32-29/h4-12,18,22-27,34H,13-17H2,1-3H3,(H,30,35)/t22-,23-,24+,25-,26-,27+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00390 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 protease |
J Med Chem 58: 6994-7006 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00900
BindingDB Entry DOI: 10.7270/Q2862KFS |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM50498534

(CHEMBL3605635)Show SMILES [H][C@]12OC[C@H](NC(C)C)[C@@]1([H])[C@H](CO2)OC(=O)N[C@@H](Cc1ccccc1)[C@H](O)CN(CC(C)C)S(=O)(=O)c1ccc(OC)cc1 |r| Show InChI InChI=1S/C31H45N3O8S/c1-20(2)16-34(43(37,38)24-13-11-23(39-5)12-14-24)17-27(35)25(15-22-9-7-6-8-10-22)33-31(36)42-28-19-41-30-29(28)26(18-40-30)32-21(3)4/h6-14,20-21,25-30,32,35H,15-19H2,1-5H3,(H,33,36)/t25-,26-,27+,28-,29-,30+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00630 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 protease |
J Med Chem 58: 6994-7006 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00900
BindingDB Entry DOI: 10.7270/Q2862KFS |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM50498528

(CHEMBL3605644)Show SMILES [H][C@]12OC[C@H](NC)[C@@]1([H])[C@H](CO2)OC(=O)N[C@@H](Cc1ccccc1)[C@H](O)CN(CC(C)C)S(=O)(=O)c1ccc(N)cc1 |r| Show InChI InChI=1S/C28H40N4O7S/c1-18(2)14-32(40(35,36)21-11-9-20(29)10-12-21)15-24(33)22(13-19-7-5-4-6-8-19)31-28(34)39-25-17-38-27-26(25)23(30-3)16-37-27/h4-12,18,22-27,30,33H,13-17,29H2,1-3H3,(H,31,34)/t22-,23-,24+,25-,26-,27+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| 0.00990 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 protease |
J Med Chem 58: 6994-7006 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00900
BindingDB Entry DOI: 10.7270/Q2862KFS |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM50498531
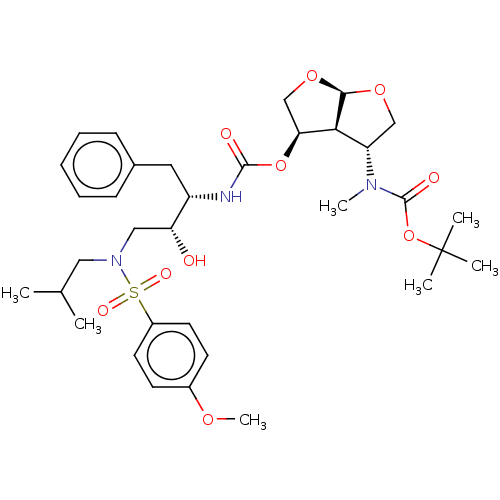
(CHEMBL3605642)Show SMILES [H][C@@]12OC[C@H](OC(=O)N[C@@H](Cc3ccccc3)[C@H](O)CN(CC(C)C)S(=O)(=O)c3ccc(OC)cc3)[C@]1([H])[C@H](CO2)N(C)C(=O)OC(C)(C)C |r| Show InChI InChI=1S/C34H49N3O10S/c1-22(2)18-37(48(41,42)25-15-13-24(43-7)14-16-25)19-28(38)26(17-23-11-9-8-10-12-23)35-32(39)46-29-21-45-31-30(29)27(20-44-31)36(6)33(40)47-34(3,4)5/h8-16,22,26-31,38H,17-21H2,1-7H3,(H,35,39)/t26-,27-,28+,29-,30-,31+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 protease |
J Med Chem 58: 6994-7006 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00900
BindingDB Entry DOI: 10.7270/Q2862KFS |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM50498527

(CHEMBL3605640)Show SMILES [H][C@]12OC[C@H](NC(=O)OC(C)(C)C)[C@@]1([H])[C@H](CO2)OC(=O)N[C@@H](Cc1ccccc1)[C@H](O)CN(CC(C)C)S(=O)(=O)c1ccc(OC)cc1 |r| Show InChI InChI=1S/C33H47N3O10S/c1-21(2)17-36(47(40,41)24-14-12-23(42-6)13-15-24)18-27(37)25(16-22-10-8-7-9-11-22)34-31(38)45-28-20-44-30-29(28)26(19-43-30)35-32(39)46-33(3,4)5/h7-15,21,25-30,37H,16-20H2,1-6H3,(H,34,38)(H,35,39)/t25-,26-,27+,28-,29-,30+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 protease |
J Med Chem 58: 6994-7006 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00900
BindingDB Entry DOI: 10.7270/Q2862KFS |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM50498526
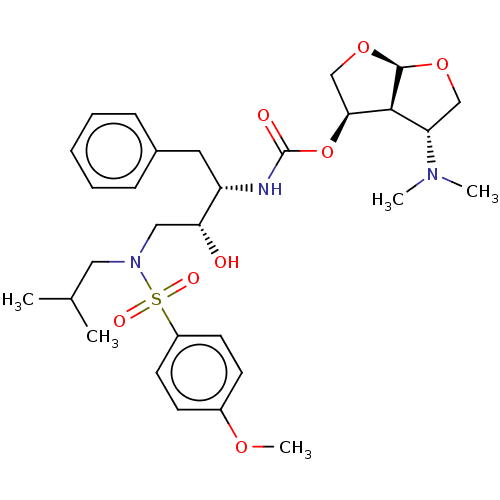
(CHEMBL3605636)Show SMILES [H][C@@]12OC[C@H](OC(=O)N[C@@H](Cc3ccccc3)[C@H](O)CN(CC(C)C)S(=O)(=O)c3ccc(OC)cc3)[C@]1([H])[C@H](CO2)N(C)C |r| Show InChI InChI=1S/C30H43N3O8S/c1-20(2)16-33(42(36,37)23-13-11-22(38-5)12-14-23)17-26(34)24(15-21-9-7-6-8-10-21)31-30(35)41-27-19-40-29-28(27)25(18-39-29)32(3)4/h6-14,20,24-29,34H,15-19H2,1-5H3,(H,31,35)/t24-,25-,26+,27-,28-,29+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 protease |
J Med Chem 58: 6994-7006 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00900
BindingDB Entry DOI: 10.7270/Q2862KFS |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM50498530

(CHEMBL3605637)Show SMILES [H][C@@]12OC[C@H](OC(=O)N[C@@H](Cc3ccccc3)[C@H](O)CN(CC(C)C)S(=O)(=O)c3ccc(OC)cc3)[C@]1([H])[C@H](CO2)N(CC)CC |r| Show InChI InChI=1S/C32H47N3O8S/c1-6-34(7-2)27-20-41-31-30(27)29(21-42-31)43-32(37)33-26(17-23-11-9-8-10-12-23)28(36)19-35(18-22(3)4)44(38,39)25-15-13-24(40-5)14-16-25/h8-16,22,26-31,36H,6-7,17-21H2,1-5H3,(H,33,37)/t26-,27-,28+,29-,30-,31+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 protease |
J Med Chem 58: 6994-7006 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00900
BindingDB Entry DOI: 10.7270/Q2862KFS |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM8125

((3R,3aS,6aR)-hexahydrofuro[2,3-b]furan-3-yl N-[(2S...)Show SMILES [H][C@@]1(CO[C@@]2([H])OCC[C@@]12[H])OC(=O)N[C@@H](Cc1ccccc1)[C@H](O)CN(CC(C)C)S(=O)(=O)c1ccc(N)cc1 |r| Show InChI InChI=1S/C27H37N3O7S/c1-18(2)15-30(38(33,34)21-10-8-20(28)9-11-21)16-24(31)23(14-19-6-4-3-5-7-19)29-27(32)37-25-17-36-26-22(25)12-13-35-26/h3-11,18,22-26,31H,12-17,28H2,1-2H3,(H,29,32)/t22-,23-,24+,25-,26+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.0160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV-1 protease by fluorometric assay |
Bioorg Med Chem Lett 25: 4903-4909 (2015)
Article DOI: 10.1016/j.bmcl.2015.05.052
BindingDB Entry DOI: 10.7270/Q2P84FW4 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM8125

((3R,3aS,6aR)-hexahydrofuro[2,3-b]furan-3-yl N-[(2S...)Show SMILES [H][C@@]1(CO[C@@]2([H])OCC[C@@]12[H])OC(=O)N[C@@H](Cc1ccccc1)[C@H](O)CN(CC(C)C)S(=O)(=O)c1ccc(N)cc1 |r| Show InChI InChI=1S/C27H37N3O7S/c1-18(2)15-30(38(33,34)21-10-8-20(28)9-11-21)16-24(31)23(14-19-6-4-3-5-7-19)29-27(32)37-25-17-36-26-22(25)12-13-35-26/h3-11,18,22-26,31H,12-17,28H2,1-2H3,(H,29,32)/t22-,23-,24+,25-,26+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.0160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 protease |
J Med Chem 58: 6994-7006 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00900
BindingDB Entry DOI: 10.7270/Q2862KFS |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM50498532

(CHEMBL3605641)Show SMILES [H][C@]12OC[C@H](NC(=O)OC)[C@@]1([H])[C@H](CO2)OC(=O)N[C@@H](Cc1ccccc1)[C@H](O)CN(CC(C)C)S(=O)(=O)c1ccc(OC)cc1 |r| Show InChI InChI=1S/C30H41N3O10S/c1-19(2)15-33(44(37,38)22-12-10-21(39-3)11-13-22)16-25(34)23(14-20-8-6-5-7-9-20)31-30(36)43-26-18-42-28-27(26)24(17-41-28)32-29(35)40-4/h5-13,19,23-28,34H,14-18H2,1-4H3,(H,31,36)(H,32,35)/t23-,24-,25+,26-,27-,28+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 protease |
J Med Chem 58: 6994-7006 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00900
BindingDB Entry DOI: 10.7270/Q2862KFS |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM50498533
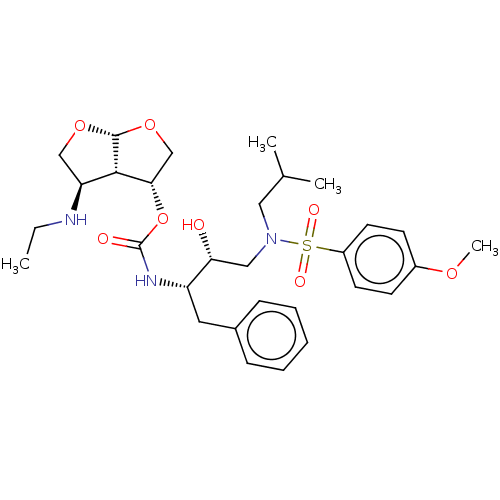
(CHEMBL3605646)Show SMILES [H][C@]12OC[C@H](NCC)[C@@]1([H])[C@H](CO2)OC(=O)N[C@@H](Cc1ccccc1)[C@H](O)CN(CC(C)C)S(=O)(=O)c1ccc(OC)cc1 |r| Show InChI InChI=1S/C30H43N3O8S/c1-5-31-25-18-39-29-28(25)27(19-40-29)41-30(35)32-24(15-21-9-7-6-8-10-21)26(34)17-33(16-20(2)3)42(36,37)23-13-11-22(38-4)12-14-23/h6-14,20,24-29,31,34H,5,15-19H2,1-4H3,(H,32,35)/t24-,25-,26+,27-,28-,29+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 protease |
J Med Chem 58: 6994-7006 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00900
BindingDB Entry DOI: 10.7270/Q2862KFS |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM50498529
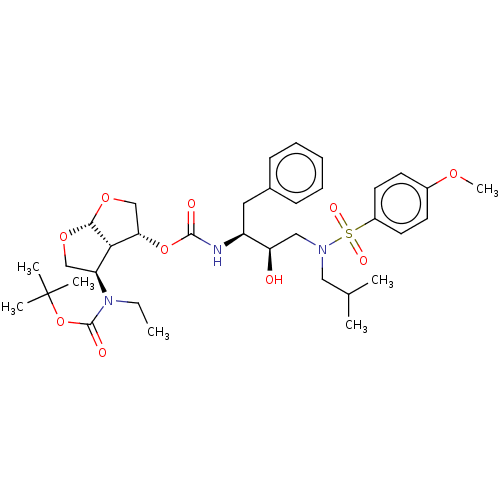
(CHEMBL3605645)Show SMILES [H][C@@]12OC[C@H](OC(=O)N[C@@H](Cc3ccccc3)[C@H](O)CN(CC(C)C)S(=O)(=O)c3ccc(OC)cc3)[C@]1([H])[C@H](CO2)N(CC)C(=O)OC(C)(C)C |r| Show InChI InChI=1S/C35H51N3O10S/c1-8-38(34(41)48-35(4,5)6)28-21-45-32-31(28)30(22-46-32)47-33(40)36-27(18-24-12-10-9-11-13-24)29(39)20-37(19-23(2)3)49(42,43)26-16-14-25(44-7)15-17-26/h9-17,23,27-32,39H,8,18-22H2,1-7H3,(H,36,40)/t27-,28-,29+,30-,31-,32+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0480 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 protease |
J Med Chem 58: 6994-7006 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00900
BindingDB Entry DOI: 10.7270/Q2862KFS |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM50498806
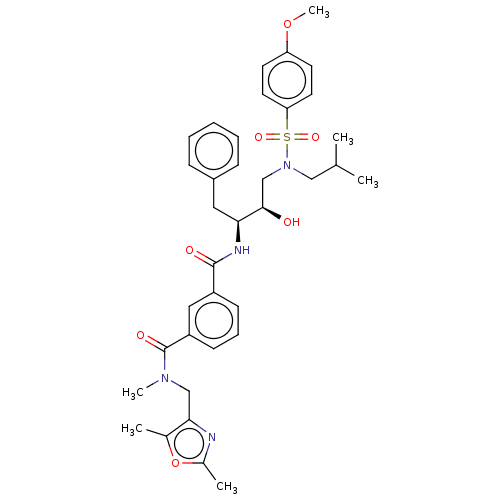
(CHEMBL3627879)Show SMILES COc1ccc(cc1)S(=O)(=O)N(CC(C)C)C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)c1cccc(c1)C(=O)N(C)Cc1nc(C)oc1C |r| Show InChI InChI=1S/C36H44N4O7S/c1-24(2)21-40(48(44,45)31-17-15-30(46-6)16-18-31)23-34(41)32(19-27-11-8-7-9-12-27)38-35(42)28-13-10-14-29(20-28)36(43)39(5)22-33-25(3)47-26(4)37-33/h7-18,20,24,32,34,41H,19,21-23H2,1-6H3,(H,38,42)/t32-,34+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV-1 protease by fluorometric assay |
Bioorg Med Chem Lett 25: 4903-4909 (2015)
Article DOI: 10.1016/j.bmcl.2015.05.052
BindingDB Entry DOI: 10.7270/Q2P84FW4 |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM171372
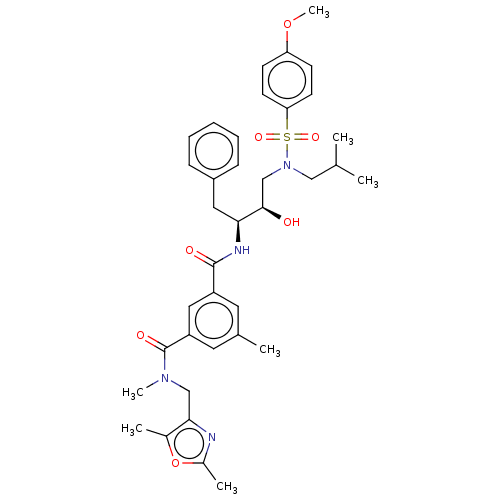
(US9085571, Table 1, Compound 20)Show SMILES COc1ccc(cc1)S(=O)(=O)N(CC(C)C)C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)c1cc(C)cc(c1)C(=O)N(C)Cc1nc(C)oc1C |r| Show InChI InChI=1S/C37H46N4O7S/c1-24(2)21-41(49(45,46)32-15-13-31(47-7)14-16-32)23-35(42)33(19-28-11-9-8-10-12-28)39-36(43)29-17-25(3)18-30(20-29)37(44)40(6)22-34-26(4)48-27(5)38-34/h8-18,20,24,33,35,42H,19,21-23H2,1-7H3,(H,39,43)/t33-,35+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV-1 protease by fluorometric assay |
Bioorg Med Chem Lett 25: 4903-4909 (2015)
Article DOI: 10.1016/j.bmcl.2015.05.052
BindingDB Entry DOI: 10.7270/Q2P84FW4 |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM50498808
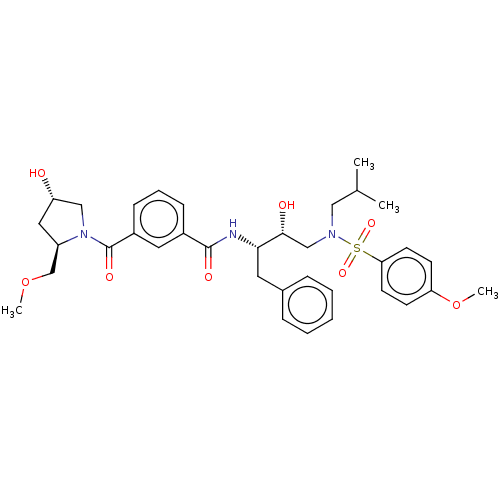
(CHEMBL3627874)Show SMILES COC[C@H]1C[C@H](O)CN1C(=O)c1cccc(c1)C(=O)N[C@@H](Cc1ccccc1)[C@H](O)CN(CC(C)C)S(=O)(=O)c1ccc(OC)cc1 |r| Show InChI InChI=1S/C35H45N3O8S/c1-24(2)20-37(47(43,44)31-15-13-30(46-4)14-16-31)22-33(40)32(17-25-9-6-5-7-10-25)36-34(41)26-11-8-12-27(18-26)35(42)38-21-29(39)19-28(38)23-45-3/h5-16,18,24,28-29,32-33,39-40H,17,19-23H2,1-4H3,(H,36,41)/t28-,29+,32+,33-/m1/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV-1 protease by fluorometric assay |
Bioorg Med Chem Lett 25: 4903-4909 (2015)
Article DOI: 10.1016/j.bmcl.2015.05.052
BindingDB Entry DOI: 10.7270/Q2P84FW4 |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM50498814
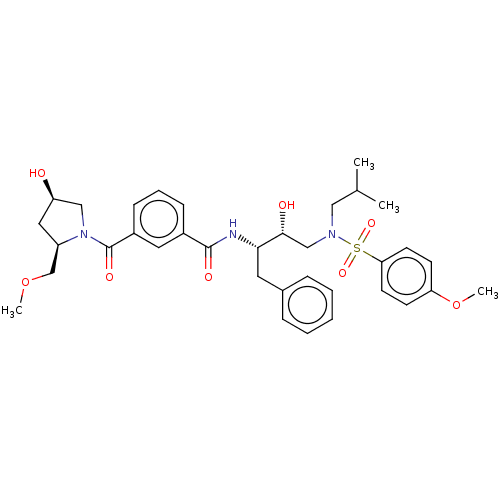
(CHEMBL3627876)Show SMILES COC[C@H]1C[C@@H](O)CN1C(=O)c1cccc(c1)C(=O)N[C@@H](Cc1ccccc1)[C@H](O)CN(CC(C)C)S(=O)(=O)c1ccc(OC)cc1 |r| Show InChI InChI=1S/C35H45N3O8S/c1-24(2)20-37(47(43,44)31-15-13-30(46-4)14-16-31)22-33(40)32(17-25-9-6-5-7-10-25)36-34(41)26-11-8-12-27(18-26)35(42)38-21-29(39)19-28(38)23-45-3/h5-16,18,24,28-29,32-33,39-40H,17,19-23H2,1-4H3,(H,36,41)/t28-,29-,32+,33-/m1/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV-1 protease by fluorometric assay |
Bioorg Med Chem Lett 25: 4903-4909 (2015)
Article DOI: 10.1016/j.bmcl.2015.05.052
BindingDB Entry DOI: 10.7270/Q2P84FW4 |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM171370
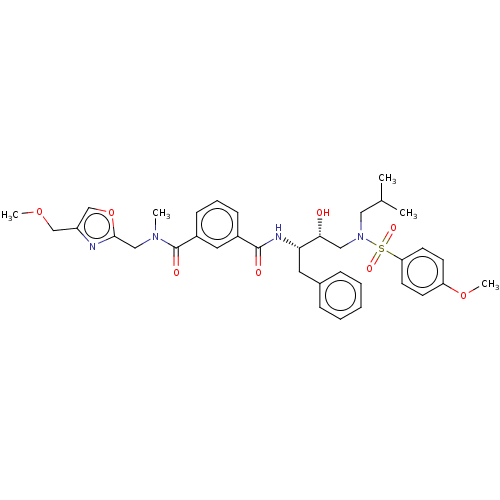
(US9085571, Table 1, Compound 18)Show SMILES COCc1coc(CN(C)C(=O)c2cccc(c2)C(=O)N[C@@H](Cc2ccccc2)[C@H](O)CN(CC(C)C)S(=O)(=O)c2ccc(OC)cc2)n1 |r| Show InChI InChI=1S/C36H44N4O8S/c1-25(2)20-40(49(44,45)31-16-14-30(47-5)15-17-31)21-33(41)32(18-26-10-7-6-8-11-26)38-35(42)27-12-9-13-28(19-27)36(43)39(3)22-34-37-29(23-46-4)24-48-34/h6-17,19,24-25,32-33,41H,18,20-23H2,1-5H3,(H,38,42)/t32-,33+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV-1 protease by fluorometric assay |
Bioorg Med Chem Lett 25: 4903-4909 (2015)
Article DOI: 10.1016/j.bmcl.2015.05.052
BindingDB Entry DOI: 10.7270/Q2P84FW4 |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM50498812
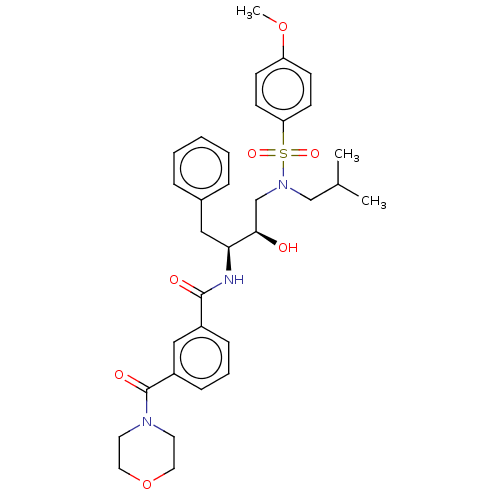
(CHEMBL3627855)Show SMILES COc1ccc(cc1)S(=O)(=O)N(CC(C)C)C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)c1cccc(c1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C33H41N3O7S/c1-24(2)22-36(44(40,41)29-14-12-28(42-3)13-15-29)23-31(37)30(20-25-8-5-4-6-9-25)34-32(38)26-10-7-11-27(21-26)33(39)35-16-18-43-19-17-35/h4-15,21,24,30-31,37H,16-20,22-23H2,1-3H3,(H,34,38)/t30-,31+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.420 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV-1 protease by fluorometric assay |
Bioorg Med Chem Lett 25: 4903-4909 (2015)
Article DOI: 10.1016/j.bmcl.2015.05.052
BindingDB Entry DOI: 10.7270/Q2P84FW4 |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM50498525
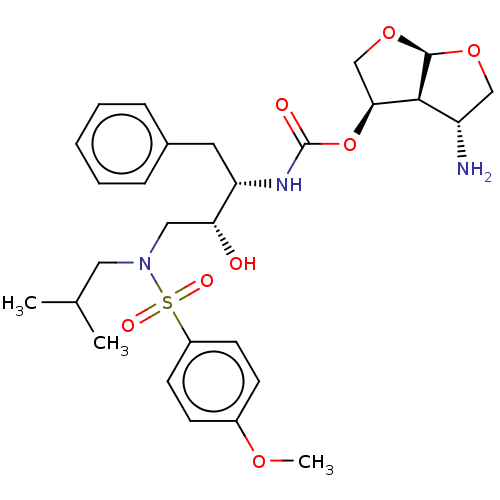
(CHEMBL3605639)Show SMILES [H][C@]12OC[C@H](N)[C@@]1([H])[C@H](CO2)OC(=O)N[C@@H](Cc1ccccc1)[C@H](O)CN(CC(C)C)S(=O)(=O)c1ccc(OC)cc1 |r| Show InChI InChI=1S/C28H39N3O8S/c1-18(2)14-31(40(34,35)21-11-9-20(36-3)10-12-21)15-24(32)23(13-19-7-5-4-6-8-19)30-28(33)39-25-17-38-27-26(25)22(29)16-37-27/h4-12,18,22-27,32H,13-17,29H2,1-3H3,(H,30,33)/t22-,23-,24+,25-,26-,27+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.530 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 protease |
J Med Chem 58: 6994-7006 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00900
BindingDB Entry DOI: 10.7270/Q2862KFS |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM50498807
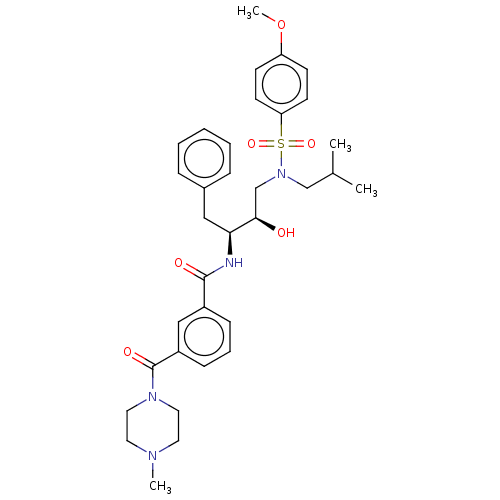
(CHEMBL3627856)Show SMILES COc1ccc(cc1)S(=O)(=O)N(CC(C)C)C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)c1cccc(c1)C(=O)N1CCN(C)CC1 |r| Show InChI InChI=1S/C34H44N4O6S/c1-25(2)23-38(45(42,43)30-15-13-29(44-4)14-16-30)24-32(39)31(21-26-9-6-5-7-10-26)35-33(40)27-11-8-12-28(22-27)34(41)37-19-17-36(3)18-20-37/h5-16,22,25,31-32,39H,17-21,23-24H2,1-4H3,(H,35,40)/t31-,32+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.540 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV-1 protease by fluorometric assay |
Bioorg Med Chem Lett 25: 4903-4909 (2015)
Article DOI: 10.1016/j.bmcl.2015.05.052
BindingDB Entry DOI: 10.7270/Q2P84FW4 |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM171371
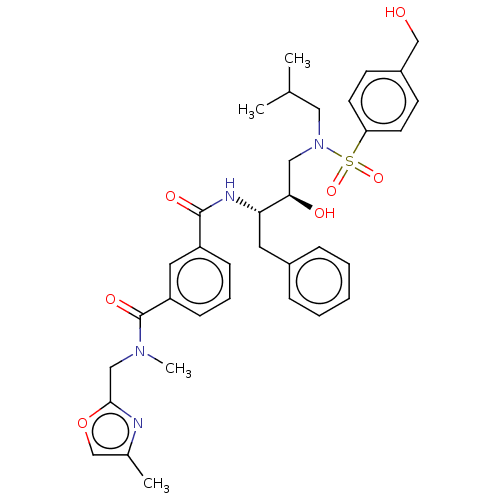
(US9085571, Table 1, Compound 19)Show SMILES CC(C)CN(C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)c1cccc(c1)C(=O)N(C)Cc1nc(C)co1)S(=O)(=O)c1ccc(CO)cc1 |r| Show InChI InChI=1S/C35H42N4O7S/c1-24(2)19-39(47(44,45)30-15-13-27(22-40)14-16-30)20-32(41)31(17-26-9-6-5-7-10-26)37-34(42)28-11-8-12-29(18-28)35(43)38(4)21-33-36-25(3)23-46-33/h5-16,18,23-24,31-32,40-41H,17,19-22H2,1-4H3,(H,37,42)/t31-,32+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.630 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV-1 protease by fluorometric assay |
Bioorg Med Chem Lett 25: 4903-4909 (2015)
Article DOI: 10.1016/j.bmcl.2015.05.052
BindingDB Entry DOI: 10.7270/Q2P84FW4 |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM50498813
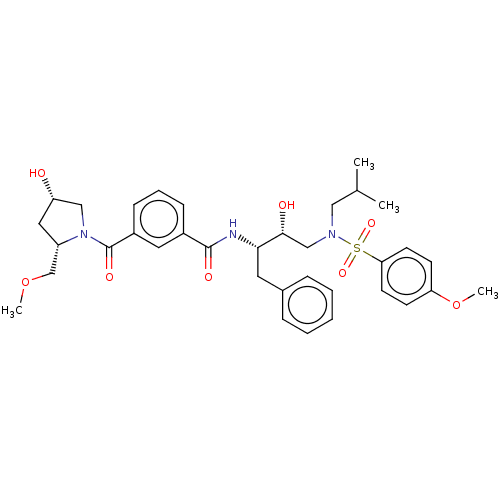
(CHEMBL3627875)Show SMILES COC[C@@H]1C[C@H](O)CN1C(=O)c1cccc(c1)C(=O)N[C@@H](Cc1ccccc1)[C@H](O)CN(CC(C)C)S(=O)(=O)c1ccc(OC)cc1 |r| Show InChI InChI=1S/C35H45N3O8S/c1-24(2)20-37(47(43,44)31-15-13-30(46-4)14-16-31)22-33(40)32(17-25-9-6-5-7-10-25)36-34(41)26-11-8-12-27(18-26)35(42)38-21-29(39)19-28(38)23-45-3/h5-16,18,24,28-29,32-33,39-40H,17,19-23H2,1-4H3,(H,36,41)/t28-,29-,32-,33+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV-1 protease by fluorometric assay |
Bioorg Med Chem Lett 25: 4903-4909 (2015)
Article DOI: 10.1016/j.bmcl.2015.05.052
BindingDB Entry DOI: 10.7270/Q2P84FW4 |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM50498810
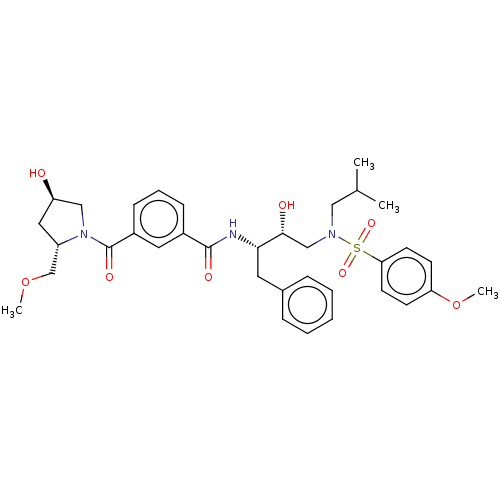
(CHEMBL3627857)Show SMILES COC[C@@H]1C[C@@H](O)CN1C(=O)c1cccc(c1)C(=O)N[C@@H](Cc1ccccc1)[C@H](O)CN(CC(C)C)S(=O)(=O)c1ccc(OC)cc1 |r| Show InChI InChI=1S/C35H45N3O8S/c1-24(2)20-37(47(43,44)31-15-13-30(46-4)14-16-31)22-33(40)32(17-25-9-6-5-7-10-25)36-34(41)26-11-8-12-27(18-26)35(42)38-21-29(39)19-28(38)23-45-3/h5-16,18,24,28-29,32-33,39-40H,17,19-23H2,1-4H3,(H,36,41)/t28-,29+,32-,33+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 2.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV-1 protease by fluorometric assay |
Bioorg Med Chem Lett 25: 4903-4909 (2015)
Article DOI: 10.1016/j.bmcl.2015.05.052
BindingDB Entry DOI: 10.7270/Q2P84FW4 |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM50498809
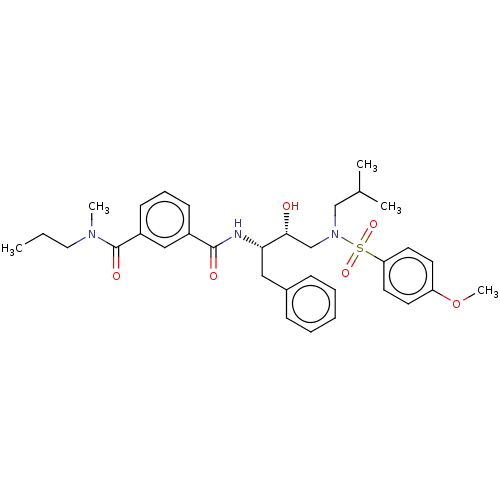
(CHEMBL3627852)Show SMILES CCCN(C)C(=O)c1cccc(c1)C(=O)N[C@@H](Cc1ccccc1)[C@H](O)CN(CC(C)C)S(=O)(=O)c1ccc(OC)cc1 |r| Show InChI InChI=1S/C33H43N3O6S/c1-6-19-35(4)33(39)27-14-10-13-26(21-27)32(38)34-30(20-25-11-8-7-9-12-25)31(37)23-36(22-24(2)3)43(40,41)29-17-15-28(42-5)16-18-29/h7-18,21,24,30-31,37H,6,19-20,22-23H2,1-5H3,(H,34,38)/t30-,31+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 5.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV-1 protease by fluorometric assay |
Bioorg Med Chem Lett 25: 4903-4909 (2015)
Article DOI: 10.1016/j.bmcl.2015.05.052
BindingDB Entry DOI: 10.7270/Q2P84FW4 |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM171367
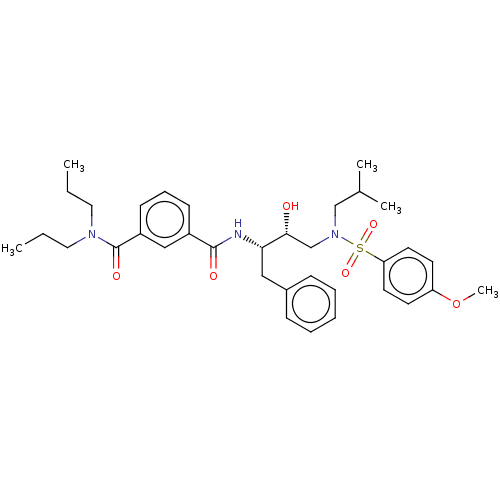
(US9085571, Table 1, Compound 15)Show SMILES CCCN(CCC)C(=O)c1cccc(c1)C(=O)N[C@@H](Cc1ccccc1)[C@H](O)CN(CC(C)C)S(=O)(=O)c1ccc(OC)cc1 |r| Show InChI InChI=1S/C35H47N3O6S/c1-6-20-37(21-7-2)35(41)29-15-11-14-28(23-29)34(40)36-32(22-27-12-9-8-10-13-27)33(39)25-38(24-26(3)4)45(42,43)31-18-16-30(44-5)17-19-31/h8-19,23,26,32-33,39H,6-7,20-22,24-25H2,1-5H3,(H,36,40)/t32-,33+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV-1 protease by fluorometric assay |
Bioorg Med Chem Lett 25: 4903-4909 (2015)
Article DOI: 10.1016/j.bmcl.2015.05.052
BindingDB Entry DOI: 10.7270/Q2P84FW4 |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM171369
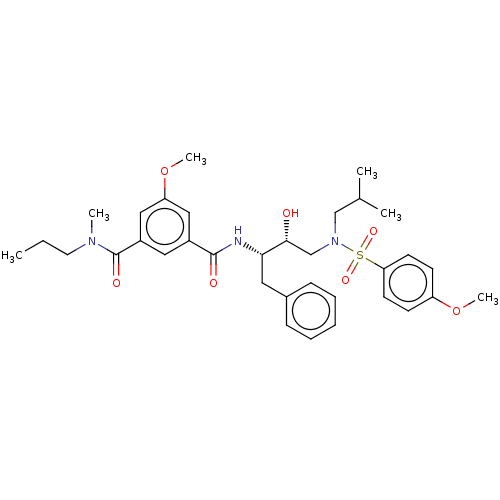
(US9085571, Table 1, Compound 17)Show SMILES CCCN(C)C(=O)c1cc(OC)cc(c1)C(=O)N[C@@H](Cc1ccccc1)[C@H](O)CN(CC(C)C)S(=O)(=O)c1ccc(OC)cc1 |r| Show InChI InChI=1S/C34H45N3O7S/c1-7-17-36(4)34(40)27-19-26(20-29(21-27)44-6)33(39)35-31(18-25-11-9-8-10-12-25)32(38)23-37(22-24(2)3)45(41,42)30-15-13-28(43-5)14-16-30/h8-16,19-21,24,31-32,38H,7,17-18,22-23H2,1-6H3,(H,35,39)/t31-,32+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV-1 protease by fluorometric assay |
Bioorg Med Chem Lett 25: 4903-4909 (2015)
Article DOI: 10.1016/j.bmcl.2015.05.052
BindingDB Entry DOI: 10.7270/Q2P84FW4 |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM50498811
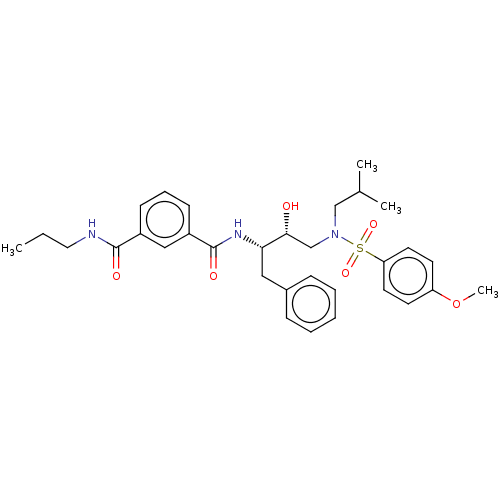
(CHEMBL3627851)Show SMILES CCCNC(=O)c1cccc(c1)C(=O)N[C@@H](Cc1ccccc1)[C@H](O)CN(CC(C)C)S(=O)(=O)c1ccc(OC)cc1 |r| Show InChI InChI=1S/C32H41N3O6S/c1-5-18-33-31(37)25-12-9-13-26(20-25)32(38)34-29(19-24-10-7-6-8-11-24)30(36)22-35(21-23(2)3)42(39,40)28-16-14-27(41-4)15-17-28/h6-17,20,23,29-30,36H,5,18-19,21-22H2,1-4H3,(H,33,37)(H,34,38)/t29-,30+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV-1 protease by fluorometric assay |
Bioorg Med Chem Lett 25: 4903-4909 (2015)
Article DOI: 10.1016/j.bmcl.2015.05.052
BindingDB Entry DOI: 10.7270/Q2P84FW4 |
More data for this
Ligand-Target Pair | |
Protease
(Human immunodeficiency virus 1 (HIV-1)) | BDBM171362
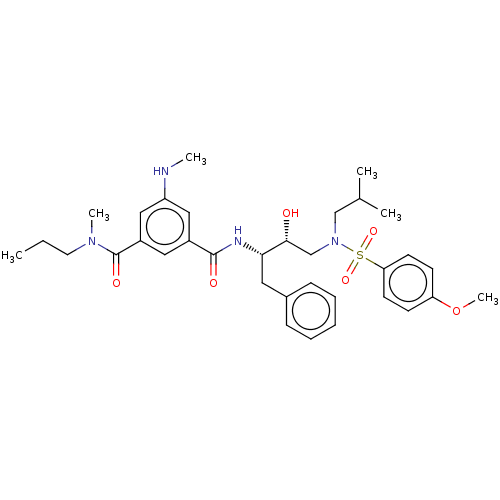
(US9085571, Table 1, Compound 10)Show SMILES CCCN(C)C(=O)c1cc(NC)cc(c1)C(=O)N[C@@H](Cc1ccccc1)[C@H](O)CN(CC(C)C)S(=O)(=O)c1ccc(OC)cc1 |r| Show InChI InChI=1S/C34H46N4O6S/c1-7-17-37(5)34(41)27-19-26(20-28(21-27)35-4)33(40)36-31(18-25-11-9-8-10-12-25)32(39)23-38(22-24(2)3)45(42,43)30-15-13-29(44-6)14-16-30/h8-16,19-21,24,31-32,35,39H,7,17-18,22-23H2,1-6H3,(H,36,40)/t31-,32+/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 158 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of HIV-1 protease by fluorometric assay |
Bioorg Med Chem Lett 25: 4903-4909 (2015)
Article DOI: 10.1016/j.bmcl.2015.05.052
BindingDB Entry DOI: 10.7270/Q2P84FW4 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data