 Found 692 hits with Last Name = 'nash' and Initial = 'k'
Found 692 hits with Last Name = 'nash' and Initial = 'k' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581191

(CHEMBL5070876)Show SMILES COc1ccc(cc1OC)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)c1ccc(CN(C(=O)O[C@H]2CN3CCC2CC3)c2ccccc2)s1 |r,wU:10.22,wD:33.34,(21.99,-25.56,;23.33,-26.33,;24.66,-25.56,;25.99,-26.33,;27.33,-25.56,;27.32,-24.01,;25.99,-23.25,;24.66,-24.02,;23.33,-23.25,;23.33,-21.71,;28.66,-23.24,;29.99,-24,;31.32,-23.23,;32.66,-24,;32.66,-25.54,;33.99,-23.22,;33.98,-21.68,;35.31,-20.91,;32.64,-20.92,;31.31,-21.7,;29.97,-20.94,;28.65,-21.7,;27.31,-20.93,;25.98,-21.71,;27.3,-19.39,;26.05,-18.5,;26.52,-17.03,;28.06,-17.02,;28.95,-15.77,;28.32,-14.37,;29.22,-13.12,;28.59,-11.71,;30.69,-13.23,;31.65,-12.02,;33.18,-12.26,;34.14,-11.06,;32.92,-10.1,;32.4,-11.6,;31.09,-10.59,;32.06,-9.39,;33.58,-9.63,;26.79,-14.22,;26.16,-12.81,;24.63,-12.66,;23.73,-13.91,;24.37,-15.32,;25.9,-15.47,;28.54,-18.49,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581204

(CHEMBL5076637)Show SMILES COc1ccc(cc1OC)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)c1ccc(CNC(C(=O)O[C@H]2CN3CCC2CC3)c2ccccc2F)s1 |r,wU:10.22,wD:34.35,(20.54,-23.22,;21.88,-23.99,;23.21,-23.22,;24.54,-23.99,;25.88,-23.22,;25.87,-21.67,;24.54,-20.9,;23.21,-21.68,;21.88,-20.91,;21.88,-19.37,;27.21,-20.89,;28.54,-21.66,;29.87,-20.88,;31.21,-21.65,;31.21,-23.19,;32.54,-20.88,;32.53,-19.34,;33.86,-18.56,;31.19,-18.57,;29.86,-19.35,;28.52,-18.59,;27.2,-19.35,;25.86,-18.59,;24.53,-19.36,;25.85,-17.05,;24.61,-16.14,;25.08,-14.68,;26.65,-14.7,;27.52,-13.42,;26.84,-12.04,;27.7,-10.76,;29.24,-10.88,;29.91,-12.26,;30.1,-9.59,;31.63,-9.71,;32.31,-11.1,;33.83,-11.22,;32.55,-10.34,;33.76,-9.31,;32.5,-8.44,;34.04,-8.56,;34.7,-9.95,;27.02,-9.38,;25.5,-9.27,;24.82,-7.9,;25.69,-6.61,;27.22,-6.72,;27.9,-8.11,;29.43,-8.22,;27.1,-16.14,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581204

(CHEMBL5076637)Show SMILES COc1ccc(cc1OC)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)c1ccc(CNC(C(=O)O[C@H]2CN3CCC2CC3)c2ccccc2F)s1 |r,wU:10.22,wD:34.35,(20.54,-23.22,;21.88,-23.99,;23.21,-23.22,;24.54,-23.99,;25.88,-23.22,;25.87,-21.67,;24.54,-20.9,;23.21,-21.68,;21.88,-20.91,;21.88,-19.37,;27.21,-20.89,;28.54,-21.66,;29.87,-20.88,;31.21,-21.65,;31.21,-23.19,;32.54,-20.88,;32.53,-19.34,;33.86,-18.56,;31.19,-18.57,;29.86,-19.35,;28.52,-18.59,;27.2,-19.35,;25.86,-18.59,;24.53,-19.36,;25.85,-17.05,;24.61,-16.14,;25.08,-14.68,;26.65,-14.7,;27.52,-13.42,;26.84,-12.04,;27.7,-10.76,;29.24,-10.88,;29.91,-12.26,;30.1,-9.59,;31.63,-9.71,;32.31,-11.1,;33.83,-11.22,;32.55,-10.34,;33.76,-9.31,;32.5,-8.44,;34.04,-8.56,;34.7,-9.95,;27.02,-9.38,;25.5,-9.27,;24.82,-7.9,;25.69,-6.61,;27.22,-6.72,;27.9,-8.11,;29.43,-8.22,;27.1,-16.14,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581209
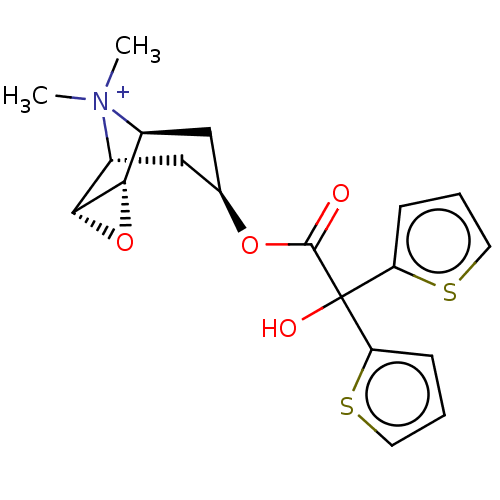
(CHEMBL4650755)Show SMILES [H][C@]12O[C@@]1([H])[C@]1([H])C[C@H](C[C@@]2([H])[N+]1(C)C)OC(=O)C(O)(c1cccs1)c1cccs1 |r| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.126 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581192

(CHEMBL5091461)Show SMILES COc1ccc(cc1OC)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)c1ccc(CN(C(=O)O[C@H]2CN3CCC2CC3)c2ccccc2F)s1 |r,wU:10.22,wD:33.34,(21.47,-24.81,;22.8,-25.58,;24.14,-24.81,;25.47,-25.58,;26.8,-24.81,;26.8,-23.26,;25.47,-22.49,;24.14,-23.26,;22.81,-22.49,;22.81,-20.95,;28.14,-22.48,;29.47,-23.25,;30.8,-22.47,;32.14,-23.24,;32.14,-24.78,;33.47,-22.47,;33.46,-20.93,;34.79,-20.15,;32.12,-20.16,;30.79,-20.94,;29.45,-20.18,;28.13,-20.94,;26.79,-20.18,;25.46,-20.95,;26.78,-18.64,;25.53,-17.74,;25.99,-16.27,;27.53,-16.27,;28.43,-15.02,;27.8,-13.61,;28.7,-12.36,;28.06,-10.96,;30.17,-12.47,;31.13,-11.27,;32.65,-11.5,;33.62,-10.31,;32.4,-9.34,;31.88,-10.85,;30.57,-9.84,;31.53,-8.64,;33.06,-8.87,;26.27,-13.46,;25.38,-14.71,;23.85,-14.57,;23.21,-13.16,;24.11,-11.9,;25.64,-12.06,;26.54,-10.81,;28.02,-17.73,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.126 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581203
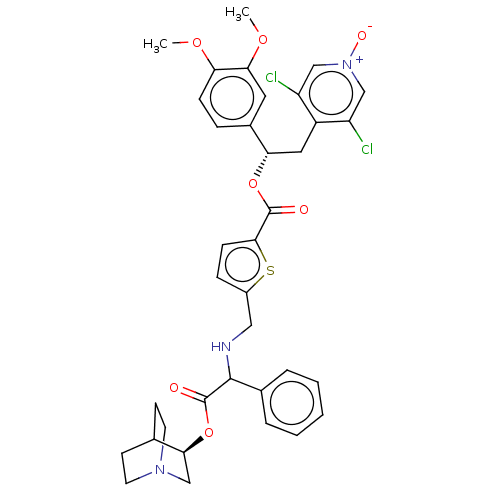
(CHEMBL5074599)Show SMILES COc1ccc(cc1OC)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)c1ccc(CNC(C(=O)O[C@H]2CN3CCC2CC3)c2ccccc2)s1 |r,wU:10.22,wD:34.35,(20.54,-23.22,;21.88,-23.99,;23.21,-23.22,;24.54,-23.99,;25.88,-23.22,;25.87,-21.67,;24.54,-20.9,;23.21,-21.68,;21.88,-20.91,;21.88,-19.37,;27.21,-20.89,;28.54,-21.66,;29.87,-20.88,;31.21,-21.65,;31.21,-23.19,;32.54,-20.88,;32.53,-19.34,;33.86,-18.56,;31.19,-18.57,;29.86,-19.35,;28.52,-18.59,;27.2,-19.35,;25.86,-18.59,;24.53,-19.36,;25.85,-17.05,;24.61,-16.14,;25.08,-14.68,;26.65,-14.7,;27.52,-13.42,;26.84,-12.04,;27.7,-10.76,;29.24,-10.88,;29.91,-12.26,;30.1,-9.59,;31.63,-9.71,;32.31,-11.1,;33.83,-11.22,;32.55,-10.34,;33.76,-9.31,;32.5,-8.44,;34.04,-8.56,;34.7,-9.95,;27.02,-9.38,;27.9,-8.11,;27.22,-6.72,;25.69,-6.61,;24.82,-7.9,;25.5,-9.27,;27.1,-16.14,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 0.126 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581203

(CHEMBL5074599)Show SMILES COc1ccc(cc1OC)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)c1ccc(CNC(C(=O)O[C@H]2CN3CCC2CC3)c2ccccc2)s1 |r,wU:10.22,wD:34.35,(20.54,-23.22,;21.88,-23.99,;23.21,-23.22,;24.54,-23.99,;25.88,-23.22,;25.87,-21.67,;24.54,-20.9,;23.21,-21.68,;21.88,-20.91,;21.88,-19.37,;27.21,-20.89,;28.54,-21.66,;29.87,-20.88,;31.21,-21.65,;31.21,-23.19,;32.54,-20.88,;32.53,-19.34,;33.86,-18.56,;31.19,-18.57,;29.86,-19.35,;28.52,-18.59,;27.2,-19.35,;25.86,-18.59,;24.53,-19.36,;25.85,-17.05,;24.61,-16.14,;25.08,-14.68,;26.65,-14.7,;27.52,-13.42,;26.84,-12.04,;27.7,-10.76,;29.24,-10.88,;29.91,-12.26,;30.1,-9.59,;31.63,-9.71,;32.31,-11.1,;33.83,-11.22,;32.55,-10.34,;33.76,-9.31,;32.5,-8.44,;34.04,-8.56,;34.7,-9.95,;27.02,-9.38,;27.9,-8.11,;27.22,-6.72,;25.69,-6.61,;24.82,-7.9,;25.5,-9.27,;27.1,-16.14,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 0.126 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581185

(CHEMBL5076558)Show SMILES COc1ccc(cc1OC)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)c1cccc(CNC(C(=O)O[C@H]2CN3CCC2CC3)c2ccccc2)c1 |r,wU:10.22,wD:35.36,(49.9,-16.29,;51.24,-17.06,;52.57,-16.29,;53.91,-17.06,;55.24,-16.29,;55.24,-14.74,;53.9,-13.98,;52.57,-14.75,;51.24,-13.98,;51.24,-12.44,;56.57,-13.96,;57.91,-14.73,;59.24,-13.95,;59.22,-12.42,;57.89,-11.66,;60.55,-11.65,;61.89,-12.41,;63.22,-11.63,;61.9,-13.95,;60.57,-14.73,;60.57,-16.27,;56.56,-12.42,;55.23,-11.66,;53.9,-12.43,;55.22,-10.12,;53.89,-9.36,;53.88,-7.83,;55.21,-7.04,;56.55,-7.81,;57.88,-7.04,;57.88,-5.5,;59.21,-4.72,;60.54,-5.49,;60.55,-7.03,;61.87,-4.71,;63.21,-5.48,;63.21,-7.03,;64.54,-7.79,;65.87,-7.02,;65.87,-5.48,;64.54,-4.71,;65.29,-6.04,;63.75,-6.45,;59.2,-3.18,;60.54,-2.41,;60.53,-.87,;59.19,-.1,;57.86,-.89,;57.87,-2.42,;56.55,-9.35,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.158 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581187

(CHEMBL5077161)Show SMILES COc1ccc(cc1OC)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)c1ccc(CN(C(=O)O[C@H]2CN3CCC2CC3)c2ccccc2)cc1 |r,wU:10.22,wD:33.34,(3.32,-53.1,;4.65,-53.87,;5.98,-53.1,;7.32,-53.87,;8.66,-53.1,;8.65,-51.55,;7.31,-50.79,;5.99,-51.56,;4.65,-50.79,;4.65,-49.25,;9.98,-50.78,;11.32,-51.54,;12.65,-50.77,;12.64,-49.24,;11.3,-48.48,;13.96,-48.46,;15.31,-49.22,;16.64,-48.45,;15.31,-50.76,;13.98,-51.54,;13.98,-53.08,;9.98,-49.24,;8.64,-48.47,;7.31,-49.25,;8.63,-46.93,;9.97,-46.16,;9.96,-44.62,;8.62,-43.85,;8.64,-42.32,;9.99,-41.56,;11.31,-42.35,;12.66,-41.6,;11.29,-43.89,;12.61,-44.68,;12.59,-46.22,;13.9,-47.01,;15.25,-46.26,;15.28,-44.72,;13.95,-43.92,;13.13,-45.36,;14.7,-45.67,;10.01,-40.02,;11.36,-39.28,;11.38,-37.74,;10.05,-36.95,;8.71,-37.71,;8.69,-39.25,;7.29,-44.64,;7.3,-46.17,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.158 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581189
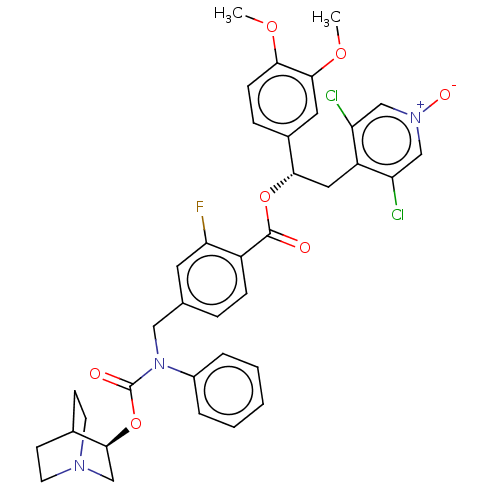
(CHEMBL5075132)Show SMILES COc1ccc(cc1OC)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)c1ccc(CN(C(=O)O[C@H]2CN3CCC2CC3)c2ccccc2)cc1F |r,wU:10.22,wD:33.34,(45.59,-53.85,;46.93,-54.62,;48.26,-53.85,;49.6,-54.62,;50.93,-53.85,;50.93,-52.3,;49.59,-51.54,;48.26,-52.31,;46.93,-51.54,;46.93,-50,;52.26,-51.53,;53.59,-52.29,;54.93,-51.52,;54.91,-49.99,;53.57,-49.23,;56.24,-49.21,;57.58,-49.97,;58.91,-49.2,;57.59,-51.52,;56.26,-52.29,;56.26,-53.83,;52.25,-49.99,;50.92,-49.22,;49.59,-50,;50.91,-47.68,;52.24,-46.91,;52.24,-45.37,;50.9,-44.61,;50.92,-43.07,;52.27,-42.32,;53.59,-43.1,;54.93,-42.35,;53.57,-44.64,;54.89,-45.43,;54.86,-46.97,;56.18,-47.76,;57.53,-47.01,;57.55,-45.47,;56.23,-44.68,;55.41,-46.11,;56.97,-46.42,;52.29,-40.78,;53.63,-40.03,;53.66,-38.49,;52.33,-37.7,;50.98,-38.46,;50.97,-40,;49.57,-45.39,;49.58,-46.93,;48.25,-47.71,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.158 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50581209

(CHEMBL4650755)Show SMILES [H][C@]12O[C@@]1([H])[C@]1([H])C[C@H](C[C@@]2([H])[N+]1(C)C)OC(=O)C(O)(c1cccs1)c1cccs1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.158 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M2 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581190
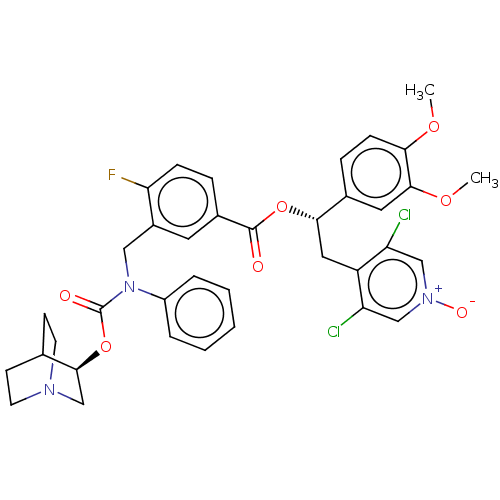
(CHEMBL5076266)Show SMILES COc1ccc(cc1OC)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)c1ccc(F)c(CN(C(=O)O[C@H]2CN3CCC2CC3)c2ccccc2)c1 |r,wU:10.22,wD:35.36,(65.48,-54.2,;66.82,-54.98,;68.15,-54.21,;69.48,-54.98,;70.82,-54.21,;70.82,-52.65,;69.48,-51.89,;68.15,-52.66,;66.82,-51.89,;66.82,-50.35,;72.15,-51.88,;73.48,-52.64,;74.81,-51.87,;74.8,-50.34,;73.46,-49.58,;76.13,-49.56,;77.47,-50.33,;78.8,-49.55,;77.48,-51.87,;76.15,-52.64,;76.15,-54.18,;72.14,-50.34,;70.8,-49.58,;69.47,-50.35,;70.8,-48.04,;69.47,-47.28,;69.45,-45.74,;70.79,-44.96,;69.92,-43.68,;72.13,-45.73,;73.46,-44.95,;73.45,-43.41,;74.78,-42.64,;74.78,-41.1,;76.12,-43.4,;77.45,-42.63,;78.79,-43.4,;80.11,-42.63,;80.12,-41.09,;78.78,-40.32,;77.44,-41.09,;78.3,-42.5,;79.33,-41.29,;72.12,-42.65,;72.12,-41.1,;70.79,-40.34,;69.45,-41.11,;69.46,-42.66,;70.8,-43.42,;72.13,-47.26,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581202

(CHEMBL5090464)Show SMILES [O-][n+]1cc(Cl)c(C[C@H](OC(=O)c2ccc(CN(C(=O)O[C@H]3CN4CCC3CC4)c3ccccc3F)s2)c2ccc(OC(F)F)c(OCC3CC3)c2)c(Cl)c1 |r,wU:7.7,wD:20.19,(41.59,-14.5,;40.26,-15.27,;40.26,-16.81,;38.94,-17.59,;38.94,-19.13,;37.6,-16.82,;36.27,-17.59,;34.94,-16.83,;34.93,-15.29,;33.59,-14.52,;32.26,-15.3,;33.58,-12.98,;32.33,-12.09,;32.8,-10.62,;34.34,-10.62,;35.24,-9.36,;34.6,-7.96,;35.5,-6.71,;34.87,-5.31,;36.97,-6.82,;37.93,-5.62,;39.45,-5.85,;40.42,-4.65,;39.2,-3.69,;38.68,-5.2,;37.37,-4.19,;38.33,-2.99,;39.86,-3.22,;33.06,-7.82,;32.18,-9.07,;30.65,-8.94,;30,-7.53,;30.89,-6.27,;32.43,-6.41,;33.32,-5.16,;34.82,-12.08,;33.6,-17.6,;33.61,-19.15,;32.27,-19.92,;30.94,-19.15,;29.61,-19.92,;28.27,-19.15,;28.27,-17.61,;26.94,-19.92,;30.94,-17.61,;29.61,-16.84,;29.61,-15.3,;28.28,-14.53,;27.5,-13.2,;26.73,-14.54,;32.27,-16.84,;37.59,-15.29,;36.25,-14.53,;38.92,-14.51,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50581203

(CHEMBL5074599)Show SMILES COc1ccc(cc1OC)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)c1ccc(CNC(C(=O)O[C@H]2CN3CCC2CC3)c2ccccc2)s1 |r,wU:10.22,wD:34.35,(20.54,-23.22,;21.88,-23.99,;23.21,-23.22,;24.54,-23.99,;25.88,-23.22,;25.87,-21.67,;24.54,-20.9,;23.21,-21.68,;21.88,-20.91,;21.88,-19.37,;27.21,-20.89,;28.54,-21.66,;29.87,-20.88,;31.21,-21.65,;31.21,-23.19,;32.54,-20.88,;32.53,-19.34,;33.86,-18.56,;31.19,-18.57,;29.86,-19.35,;28.52,-18.59,;27.2,-19.35,;25.86,-18.59,;24.53,-19.36,;25.85,-17.05,;24.61,-16.14,;25.08,-14.68,;26.65,-14.7,;27.52,-13.42,;26.84,-12.04,;27.7,-10.76,;29.24,-10.88,;29.91,-12.26,;30.1,-9.59,;31.63,-9.71,;32.31,-11.1,;33.83,-11.22,;32.55,-10.34,;33.76,-9.31,;32.5,-8.44,;34.04,-8.56,;34.7,-9.95,;27.02,-9.38,;27.9,-8.11,;27.22,-6.72,;25.69,-6.61,;24.82,-7.9,;25.5,-9.27,;27.1,-16.14,)| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M2 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581193
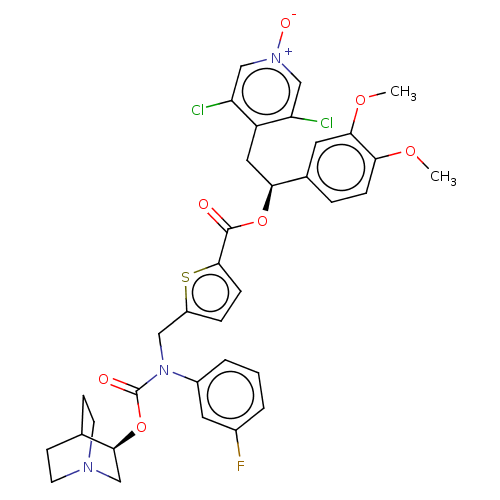
(CHEMBL5084383)Show SMILES COc1ccc(cc1OC)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)c1ccc(CN(C(=O)O[C@H]2CN3CCC2CC3)c2cccc(F)c2)s1 |r,wU:10.22,wD:33.34,(21.47,-24.81,;22.8,-25.58,;24.14,-24.81,;25.47,-25.58,;26.8,-24.81,;26.8,-23.26,;25.47,-22.49,;24.14,-23.26,;22.81,-22.49,;22.81,-20.95,;28.14,-22.48,;29.47,-23.25,;30.8,-22.47,;32.14,-23.24,;32.14,-24.78,;33.47,-22.47,;33.46,-20.93,;34.79,-20.15,;32.12,-20.16,;30.79,-20.94,;29.45,-20.18,;28.13,-20.94,;26.79,-20.18,;25.46,-20.95,;26.78,-18.64,;25.53,-17.74,;25.99,-16.27,;27.53,-16.27,;28.43,-15.02,;27.8,-13.61,;28.7,-12.36,;28.06,-10.96,;30.17,-12.47,;31.13,-11.27,;32.65,-11.5,;33.62,-10.31,;32.4,-9.34,;31.88,-10.85,;30.57,-9.84,;31.53,-8.64,;33.06,-8.87,;26.27,-13.46,;25.38,-14.71,;23.85,-14.57,;23.21,-13.16,;24.11,-11.9,;23.48,-10.5,;25.64,-12.06,;28.02,-17.73,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581199

(CHEMBL5090179)Show SMILES COc1cc(ccc1OC(F)F)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)c1ccc(CN(C(=O)O[C@H]2CN3CCC2CC3)c2ccccc2F)s1 |r,wU:12.24,wD:35.36,(22.8,-20.95,;22.8,-22.49,;24.13,-23.26,;25.46,-22.49,;26.79,-23.25,;26.79,-24.8,;25.46,-25.57,;24.13,-24.8,;22.8,-25.57,;21.46,-24.8,;21.46,-23.26,;20.13,-25.57,;28.12,-22.47,;29.46,-23.24,;30.79,-22.46,;32.13,-23.23,;32.13,-24.77,;33.45,-22.46,;33.45,-20.92,;34.77,-20.14,;32.1,-20.16,;30.77,-20.93,;29.44,-20.18,;28.12,-20.93,;26.78,-20.17,;25.45,-20.94,;26.77,-18.63,;25.52,-17.73,;25.99,-16.27,;27.52,-16.26,;28.42,-15.01,;27.79,-13.61,;28.69,-12.36,;28.05,-10.95,;30.16,-12.47,;31.12,-11.26,;32.64,-11.5,;33.6,-10.3,;32.39,-9.34,;31.86,-10.85,;30.56,-9.84,;31.52,-8.63,;33.04,-8.87,;26.25,-13.46,;25.37,-14.72,;23.84,-14.58,;23.19,-13.17,;24.08,-11.92,;25.62,-12.06,;26.51,-10.81,;28.01,-17.72,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.251 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581201

(CHEMBL5085717)Show SMILES CC(C)Oc1cc(ccc1OC(F)F)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)c1ccc(CN(C(=O)O[C@H]2CN3CCC2CC3)c2ccccc2F)s1 |r,wU:14.26,wD:37.38,(8.35,-14.65,;9.68,-15.42,;11.02,-14.65,;9.68,-16.95,;11.02,-17.72,;12.34,-16.95,;13.67,-17.72,;13.68,-19.27,;12.34,-20.04,;11.01,-19.27,;9.68,-20.04,;8.34,-19.27,;8.34,-17.73,;7.01,-20.04,;15.01,-16.94,;16.34,-17.71,;17.67,-16.93,;19.01,-17.7,;19.01,-19.24,;20.34,-16.93,;20.33,-15.39,;21.66,-14.61,;18.99,-14.63,;17.66,-15.4,;16.32,-14.64,;15,-15.4,;13.66,-14.64,;12.33,-15.41,;13.66,-13.1,;12.4,-12.2,;12.87,-10.74,;14.41,-10.73,;15.31,-9.48,;14.67,-8.08,;15.57,-6.83,;14.94,-5.42,;17.05,-6.94,;18,-5.73,;19.53,-5.97,;20.49,-4.77,;19.27,-3.81,;18.75,-5.31,;17.44,-4.31,;18.41,-3.1,;19.93,-3.34,;13.14,-7.93,;12.25,-9.19,;10.72,-9.05,;10.07,-7.64,;10.97,-6.38,;12.5,-6.53,;13.4,-5.28,;14.9,-12.19,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.316 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581186
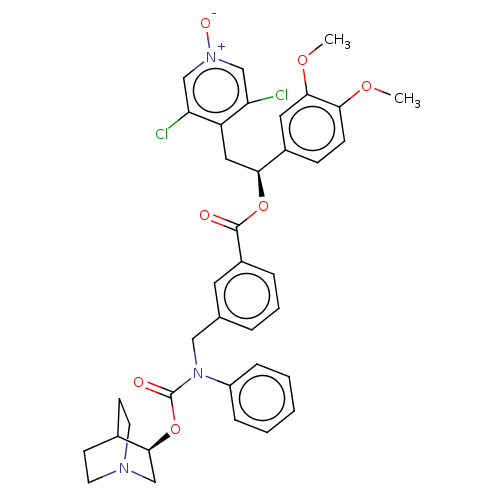
(CHEMBL5088742)Show SMILES COc1ccc(cc1OC)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)c1cccc(CN(C(=O)O[C@H]2CN3CCC2CC3)c2ccccc2)c1 |r,wU:10.22,wD:34.35,(67,-31.8,;68.33,-32.57,;69.66,-31.81,;71,-32.58,;72.34,-31.8,;72.33,-30.25,;70.99,-29.49,;69.67,-30.26,;68.33,-29.49,;68.33,-27.95,;73.66,-29.48,;75,-30.24,;76.33,-29.47,;76.32,-27.94,;74.98,-27.18,;77.64,-27.16,;78.99,-27.93,;80.32,-27.15,;78.99,-29.47,;77.66,-30.24,;77.66,-31.78,;73.66,-27.94,;72.32,-27.17,;70.99,-27.95,;72.31,-25.63,;70.98,-24.88,;70.97,-23.34,;72.3,-22.56,;73.64,-23.33,;74.97,-22.55,;74.97,-21.01,;76.3,-20.24,;76.29,-18.7,;77.63,-21,;78.97,-20.23,;80.3,-21,;81.63,-20.23,;81.63,-18.69,;80.3,-17.92,;78.96,-18.69,;79.81,-20.1,;80.84,-18.89,;73.63,-20.25,;73.64,-18.7,;72.3,-17.94,;70.97,-18.71,;70.98,-20.26,;72.31,-21.02,;73.65,-24.86,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.316 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581188

(CHEMBL5076680)Show SMILES COc1ccc(cc1OC)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)Cc1ccc(CN(C(=O)O[C@H]2CN3CCC2CC3)c2ccccc2)cc1 |r,wU:10.22,wD:34.35,(26.37,-52.58,;27.7,-53.35,;29.04,-52.58,;30.37,-53.35,;31.71,-52.58,;31.7,-51.03,;30.37,-50.26,;29.04,-51.03,;27.7,-50.26,;27.7,-48.72,;33.03,-50.25,;34.37,-51.02,;35.7,-50.24,;35.69,-48.71,;34.35,-47.95,;37.01,-47.93,;38.36,-48.7,;39.69,-47.92,;38.36,-50.24,;37.03,-51.01,;37.04,-52.55,;33.03,-48.71,;31.69,-47.95,;30.36,-48.72,;31.68,-46.41,;30.34,-45.64,;30.28,-44.1,;28.92,-43.39,;27.62,-44.21,;26.27,-43.48,;26.23,-41.94,;27.54,-41.14,;27.51,-39.6,;28.89,-41.87,;30.21,-41.07,;31.56,-41.82,;32.87,-41.02,;32.84,-39.48,;31.49,-38.74,;30.17,-39.54,;31.05,-40.93,;32.06,-39.69,;24.88,-41.2,;24.85,-39.66,;23.5,-38.92,;22.18,-39.72,;22.22,-41.27,;23.58,-42,;27.69,-45.76,;29.05,-46.47,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.316 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581198
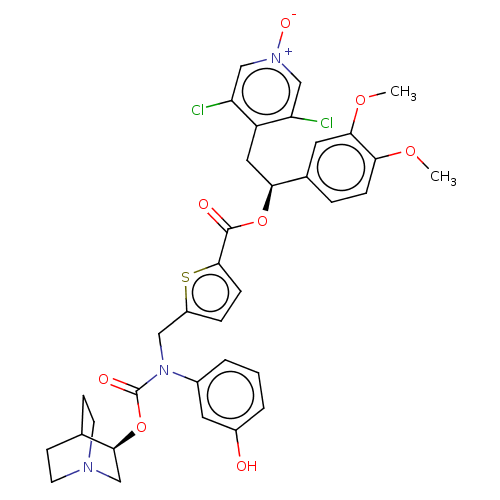
(CHEMBL5086769)Show SMILES COc1ccc(cc1OC)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)c1ccc(CN(C(=O)O[C@H]2CN3CCC2CC3)c2cccc(O)c2)s1 |r,wU:10.22,wD:33.34,(21.46,-24.8,;22.8,-25.57,;24.13,-24.8,;25.46,-25.57,;26.79,-24.8,;26.79,-23.25,;25.46,-22.49,;24.13,-23.26,;22.8,-22.49,;22.8,-20.95,;28.12,-22.47,;29.46,-23.24,;30.79,-22.46,;32.13,-23.23,;32.13,-24.77,;33.45,-22.46,;33.45,-20.92,;34.77,-20.14,;32.1,-20.16,;30.77,-20.93,;29.44,-20.18,;28.12,-20.93,;26.78,-20.17,;25.45,-20.94,;26.77,-18.63,;25.52,-17.73,;25.99,-16.27,;27.52,-16.26,;28.42,-15.01,;27.79,-13.61,;28.69,-12.36,;28.05,-10.95,;30.16,-12.47,;31.12,-11.26,;32.64,-11.5,;33.6,-10.3,;32.39,-9.34,;31.86,-10.85,;30.56,-9.84,;31.52,-8.63,;33.04,-8.87,;26.25,-13.46,;25.37,-14.72,;23.84,-14.58,;23.19,-13.17,;24.08,-11.92,;23.44,-10.51,;25.62,-12.06,;28.01,-17.72,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.316 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581200
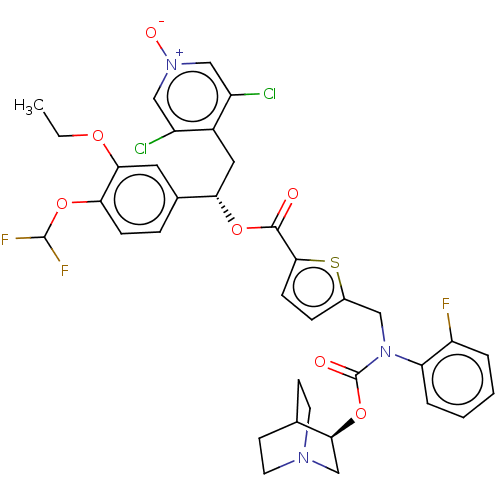
(CHEMBL5084829)Show SMILES CCOc1cc(ccc1OC(F)F)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)c1ccc(CN(C(=O)O[C@H]2CN3CCC2CC3)c2ccccc2F)s1 |r,wU:13.25,wD:36.37,(8.35,-14.65,;9.68,-15.42,;9.68,-16.95,;11.02,-17.72,;12.34,-16.95,;13.67,-17.72,;13.68,-19.27,;12.34,-20.04,;11.01,-19.27,;9.68,-20.04,;8.34,-19.27,;8.34,-17.73,;7.01,-20.04,;15.01,-16.94,;16.34,-17.71,;17.67,-16.93,;19.01,-17.7,;19.01,-19.24,;20.34,-16.93,;20.33,-15.39,;21.66,-14.61,;18.99,-14.63,;17.66,-15.4,;16.32,-14.64,;15,-15.4,;13.66,-14.64,;12.33,-15.41,;13.66,-13.1,;12.4,-12.2,;12.87,-10.74,;14.41,-10.73,;15.31,-9.48,;14.67,-8.08,;15.57,-6.83,;14.94,-5.42,;17.05,-6.94,;18,-5.73,;19.53,-5.97,;20.49,-4.77,;19.27,-3.81,;18.75,-5.31,;17.44,-4.31,;18.41,-3.1,;19.93,-3.34,;13.14,-7.93,;12.25,-9.19,;10.72,-9.05,;10.07,-7.64,;10.97,-6.38,;12.5,-6.53,;13.4,-5.28,;14.9,-12.19,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.316 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581185

(CHEMBL5076558)Show SMILES COc1ccc(cc1OC)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)c1cccc(CNC(C(=O)O[C@H]2CN3CCC2CC3)c2ccccc2)c1 |r,wU:10.22,wD:35.36,(49.9,-16.29,;51.24,-17.06,;52.57,-16.29,;53.91,-17.06,;55.24,-16.29,;55.24,-14.74,;53.9,-13.98,;52.57,-14.75,;51.24,-13.98,;51.24,-12.44,;56.57,-13.96,;57.91,-14.73,;59.24,-13.95,;59.22,-12.42,;57.89,-11.66,;60.55,-11.65,;61.89,-12.41,;63.22,-11.63,;61.9,-13.95,;60.57,-14.73,;60.57,-16.27,;56.56,-12.42,;55.23,-11.66,;53.9,-12.43,;55.22,-10.12,;53.89,-9.36,;53.88,-7.83,;55.21,-7.04,;56.55,-7.81,;57.88,-7.04,;57.88,-5.5,;59.21,-4.72,;60.54,-5.49,;60.55,-7.03,;61.87,-4.71,;63.21,-5.48,;63.21,-7.03,;64.54,-7.79,;65.87,-7.02,;65.87,-5.48,;64.54,-4.71,;65.29,-6.04,;63.75,-6.45,;59.2,-3.18,;60.54,-2.41,;60.53,-.87,;59.19,-.1,;57.86,-.89,;57.87,-2.42,;56.55,-9.35,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.398 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581183

(CHEMBL5087564)Show SMILES COc1ccc(cc1OC)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)c1cccc(NC(C(=O)O[C@H]2CN3CCC2CC3)c2ccccc2)c1 |r,wU:10.22,wD:34.35,(2.97,-32.16,;4.31,-32.93,;5.64,-32.16,;6.98,-32.94,;8.31,-32.16,;8.31,-30.61,;6.97,-29.85,;5.64,-30.62,;4.31,-29.85,;4.31,-28.31,;9.64,-29.84,;10.98,-30.6,;12.31,-29.83,;12.29,-28.3,;10.95,-27.54,;13.62,-27.52,;14.96,-28.28,;16.29,-27.51,;14.97,-29.83,;13.64,-30.6,;13.64,-32.14,;9.63,-28.3,;8.3,-27.53,;6.97,-28.31,;8.29,-25.99,;6.96,-25.24,;6.95,-23.7,;8.28,-22.92,;9.62,-23.68,;10.95,-22.91,;10.95,-21.37,;12.28,-20.59,;12.27,-19.06,;13.61,-21.36,;14.94,-20.59,;16.28,-21.36,;17.61,-20.59,;17.61,-19.05,;16.27,-18.28,;14.93,-19.05,;15.79,-20.46,;16.82,-19.25,;9.61,-20.6,;9.61,-19.06,;8.28,-18.3,;6.95,-19.07,;6.96,-20.62,;8.29,-21.38,;9.62,-25.22,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.398 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581185

(CHEMBL5076558)Show SMILES COc1ccc(cc1OC)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)c1cccc(CNC(C(=O)O[C@H]2CN3CCC2CC3)c2ccccc2)c1 |r,wU:10.22,wD:35.36,(49.9,-16.29,;51.24,-17.06,;52.57,-16.29,;53.91,-17.06,;55.24,-16.29,;55.24,-14.74,;53.9,-13.98,;52.57,-14.75,;51.24,-13.98,;51.24,-12.44,;56.57,-13.96,;57.91,-14.73,;59.24,-13.95,;59.22,-12.42,;57.89,-11.66,;60.55,-11.65,;61.89,-12.41,;63.22,-11.63,;61.9,-13.95,;60.57,-14.73,;60.57,-16.27,;56.56,-12.42,;55.23,-11.66,;53.9,-12.43,;55.22,-10.12,;53.89,-9.36,;53.88,-7.83,;55.21,-7.04,;56.55,-7.81,;57.88,-7.04,;57.88,-5.5,;59.21,-4.72,;60.54,-5.49,;60.55,-7.03,;61.87,-4.71,;63.21,-5.48,;63.21,-7.03,;64.54,-7.79,;65.87,-7.02,;65.87,-5.48,;64.54,-4.71,;65.29,-6.04,;63.75,-6.45,;59.2,-3.18,;60.54,-2.41,;60.53,-.87,;59.19,-.1,;57.86,-.89,;57.87,-2.42,;56.55,-9.35,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.398 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581206

(CHEMBL5076886)Show SMILES [H][C@@]12CC[C@]([H])(CC(C1)OC(=O)C(NCc1ccc(s1)C(=O)O[C@@H](Cc1c(Cl)c[n+]([O-])cc1Cl)c1ccc(OC)c(OC)c1)c1ccccc1)N2C |r| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.501 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581195
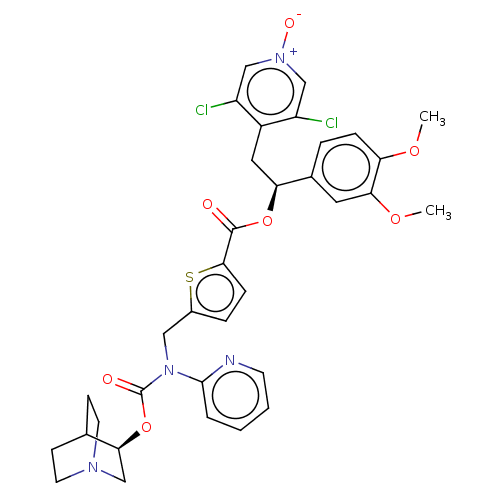
(CHEMBL5085166)Show SMILES COc1ccc(cc1OC)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)c1ccc(CN(C(=O)O[C@H]2CN3CCC2CC3)c2ccccn2)s1 |r,wU:10.22,wD:33.34,(21.46,-24.8,;22.8,-25.57,;24.13,-24.8,;25.46,-25.57,;26.79,-24.8,;26.79,-23.25,;25.46,-22.49,;24.13,-23.26,;22.8,-22.49,;22.8,-20.95,;28.12,-22.47,;29.46,-23.24,;30.79,-22.46,;32.13,-23.23,;32.13,-24.77,;33.45,-22.46,;33.45,-20.92,;34.77,-20.14,;32.1,-20.16,;30.77,-20.93,;29.44,-20.18,;28.12,-20.93,;26.78,-20.17,;25.45,-20.94,;26.77,-18.63,;25.52,-17.73,;25.99,-16.27,;27.52,-16.26,;28.42,-15.01,;27.79,-13.61,;28.69,-12.36,;28.05,-10.95,;30.16,-12.47,;31.12,-11.26,;32.64,-11.5,;33.6,-10.3,;32.39,-9.34,;31.86,-10.85,;30.56,-9.84,;31.52,-8.63,;33.04,-8.87,;26.26,-13.46,;25.37,-14.71,;23.84,-14.56,;23.2,-13.15,;24.1,-11.9,;25.63,-12.05,;28.01,-17.72,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.501 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581206

(CHEMBL5076886)Show SMILES [H][C@@]12CC[C@]([H])(CC(C1)OC(=O)C(NCc1ccc(s1)C(=O)O[C@@H](Cc1c(Cl)c[n+]([O-])cc1Cl)c1ccc(OC)c(OC)c1)c1ccccc1)N2C |r| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.501 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581205
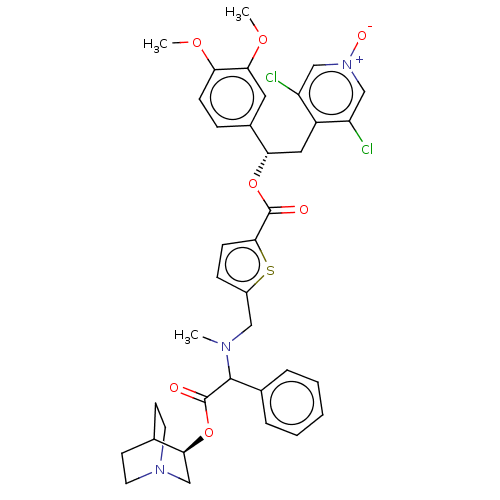
(CHEMBL5077424)Show SMILES COc1ccc(cc1OC)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)c1ccc(CN(C)C(C(=O)O[C@H]2CN3CCC2CC3)c2ccccc2)s1 |r,wU:10.22,wD:35.36,(20.59,-23.22,;21.92,-23.99,;23.26,-23.22,;24.59,-23.99,;25.92,-23.22,;25.92,-21.67,;24.59,-20.9,;23.26,-21.68,;21.93,-20.91,;21.93,-19.37,;27.26,-20.89,;28.59,-21.66,;29.92,-20.88,;31.26,-21.65,;31.26,-23.19,;32.59,-20.88,;32.58,-19.34,;33.91,-18.56,;31.24,-18.57,;29.91,-19.35,;28.57,-18.59,;27.25,-19.35,;25.91,-18.59,;24.58,-19.36,;25.9,-17.05,;24.65,-16.14,;25.12,-14.68,;26.7,-14.7,;27.56,-13.42,;26.89,-12.04,;25.35,-11.93,;27.75,-10.76,;29.28,-10.88,;29.96,-12.26,;30.14,-9.59,;31.68,-9.71,;32.35,-11.1,;33.88,-11.22,;32.59,-10.34,;33.8,-9.31,;32.54,-8.44,;34.08,-8.56,;34.75,-9.95,;27.07,-9.38,;27.94,-8.11,;27.27,-6.72,;25.73,-6.61,;24.87,-7.9,;25.54,-9.27,;27.15,-16.14,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.631 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581205

(CHEMBL5077424)Show SMILES COc1ccc(cc1OC)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)c1ccc(CN(C)C(C(=O)O[C@H]2CN3CCC2CC3)c2ccccc2)s1 |r,wU:10.22,wD:35.36,(20.59,-23.22,;21.92,-23.99,;23.26,-23.22,;24.59,-23.99,;25.92,-23.22,;25.92,-21.67,;24.59,-20.9,;23.26,-21.68,;21.93,-20.91,;21.93,-19.37,;27.26,-20.89,;28.59,-21.66,;29.92,-20.88,;31.26,-21.65,;31.26,-23.19,;32.59,-20.88,;32.58,-19.34,;33.91,-18.56,;31.24,-18.57,;29.91,-19.35,;28.57,-18.59,;27.25,-19.35,;25.91,-18.59,;24.58,-19.36,;25.9,-17.05,;24.65,-16.14,;25.12,-14.68,;26.7,-14.7,;27.56,-13.42,;26.89,-12.04,;25.35,-11.93,;27.75,-10.76,;29.28,-10.88,;29.96,-12.26,;30.14,-9.59,;31.68,-9.71,;32.35,-11.1,;33.88,-11.22,;32.59,-10.34,;33.8,-9.31,;32.54,-8.44,;34.08,-8.56,;34.75,-9.95,;27.07,-9.38,;27.94,-8.11,;27.27,-6.72,;25.73,-6.61,;24.87,-7.9,;25.54,-9.27,;27.15,-16.14,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.631 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581184
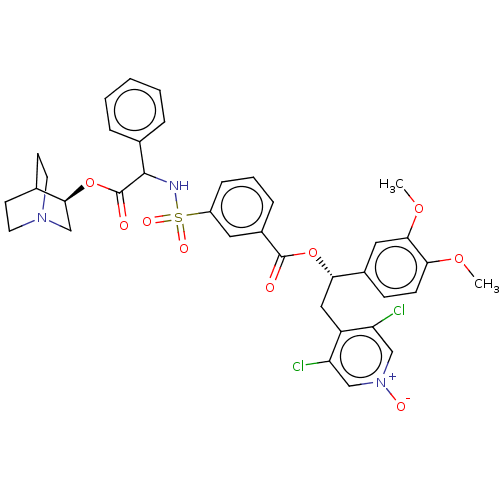
(CHEMBL5094110)Show SMILES COc1ccc(cc1OC)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)c1cccc(c1)S(=O)(=O)NC(C(=O)O[C@H]1CN2CCC1CC2)c1ccccc1 |r,wU:10.22,wD:38.40,(23.58,-31.52,;24.91,-32.29,;26.24,-31.52,;27.58,-32.29,;28.92,-31.52,;28.91,-29.97,;27.58,-29.2,;26.25,-29.97,;24.91,-29.21,;24.91,-27.67,;30.24,-29.19,;31.58,-29.96,;32.91,-29.18,;32.9,-27.65,;31.56,-26.89,;34.22,-26.87,;35.57,-27.64,;36.9,-26.86,;35.57,-29.18,;34.24,-29.95,;34.24,-31.49,;30.24,-27.65,;28.9,-26.89,;27.57,-27.66,;28.89,-25.35,;27.56,-24.59,;27.55,-23.06,;28.88,-22.27,;30.22,-23.04,;30.23,-24.58,;31.55,-22.26,;33.09,-22.26,;32.32,-23.59,;31.55,-20.72,;32.88,-19.95,;34.22,-20.72,;34.22,-22.26,;35.55,-19.94,;36.88,-20.71,;36.88,-22.25,;38.21,-23.02,;39.55,-22.25,;39.55,-20.71,;38.21,-19.94,;38.91,-21.44,;37.43,-21.68,;32.87,-18.41,;34.22,-17.63,;34.21,-16.1,;32.87,-15.33,;31.54,-16.11,;31.55,-17.65,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.631 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581181

(CHEMBL5086895)Show SMILES [O-][n+]1cc(Cl)c(C[C@H](OC(=O)c2cccc(CNC(C(=O)O[C@H]3CN4CCC3CC4)c3ccccc3)c2)c2ccc(OC(F)F)c(OCC3CC3)c2)c(Cl)c1 |r,wU:7.7,wD:22.21,(59.38,-8.9,;58.05,-9.67,;56.71,-8.91,;55.38,-9.69,;54.04,-8.93,;55.39,-11.22,;54.06,-11.99,;52.73,-11.23,;52.72,-9.69,;51.38,-8.92,;50.05,-9.7,;51.38,-7.38,;50.05,-6.62,;50.04,-5.09,;51.37,-4.31,;52.71,-5.07,;54.03,-4.29,;54.03,-2.76,;55.36,-1.98,;56.7,-2.75,;56.71,-4.29,;58.03,-1.97,;59.37,-2.74,;59.37,-4.29,;60.7,-5.05,;62.03,-4.29,;62.03,-2.74,;60.69,-1.97,;61.4,-3.48,;59.91,-3.71,;55.36,-.44,;56.7,.34,;56.7,1.88,;55.36,2.65,;54.02,1.86,;54.04,.33,;52.71,-6.61,;51.4,-12,;51.4,-13.55,;50.06,-14.32,;48.73,-13.55,;47.4,-14.32,;46.06,-13.55,;44.73,-14.32,;46.06,-12.01,;48.73,-12.01,;47.4,-11.24,;47.4,-9.7,;46.06,-8.93,;45.3,-7.6,;44.53,-8.93,;50.06,-11.24,;56.73,-11.99,;56.73,-13.53,;58.06,-11.21,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.631 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Leucine-rich repeat serine/threonine-protein kinase 2
(Homo sapiens (Human)) | BDBM50448118
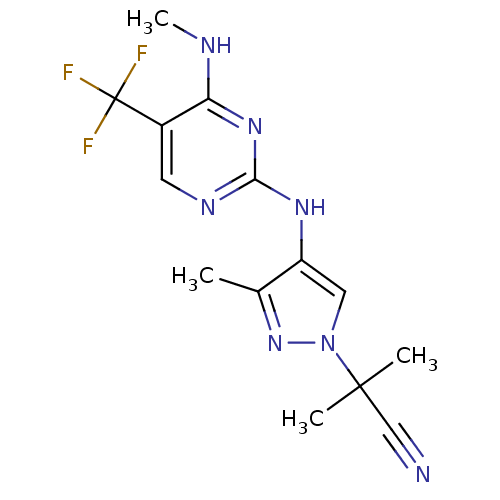
(CHEMBL3122113 | US10590114, No. 80 | US11111235, N...)Show InChI InChI=1S/C14H16F3N7/c1-8-10(6-24(23-8)13(2,3)7-18)21-12-20-5-9(14(15,16)17)11(19-4)22-12/h5-6H,1-4H3,(H2,19,20,21,22) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of LRRK2 (unknown origin) |
J Med Chem 57: 921-36 (2014)
Article DOI: 10.1021/jm401654j
BindingDB Entry DOI: 10.7270/Q2N0181N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Leucine-rich repeat serine/threonine-protein kinase 2
(Homo sapiens (Human)) | BDBM50448127
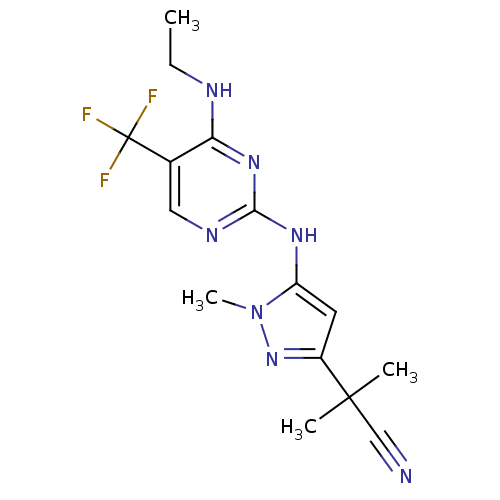
(CHEMBL3122119 | US9212173, 44)Show InChI InChI=1S/C15H18F3N7/c1-5-20-12-9(15(16,17)18)7-21-13(23-12)22-11-6-10(24-25(11)4)14(2,3)8-19/h6-7H,5H2,1-4H3,(H2,20,21,22,23) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of LRRK2 (unknown origin) |
J Med Chem 57: 921-36 (2014)
Article DOI: 10.1021/jm401654j
BindingDB Entry DOI: 10.7270/Q2N0181N |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581179
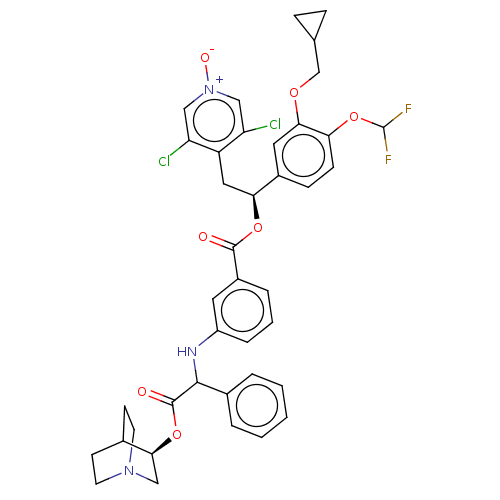
(CHEMBL5078680)Show SMILES [O-][n+]1cc(Cl)c(C[C@H](OC(=O)c2cccc(NC(C(=O)O[C@H]3CN4CCC3CC4)c3ccccc3)c2)c2ccc(OC(F)F)c(OCC3CC3)c2)c(Cl)c1 |r,wU:7.7,wD:21.20,(17.64,-7.86,;16.31,-8.64,;14.96,-7.87,;13.64,-8.65,;12.3,-7.89,;13.65,-10.18,;12.32,-10.96,;10.98,-10.19,;10.98,-8.65,;9.64,-7.89,;8.31,-8.66,;9.64,-6.35,;8.3,-5.59,;8.29,-4.05,;9.63,-3.27,;10.96,-4.04,;12.3,-3.26,;12.29,-1.72,;13.62,-.95,;13.62,.59,;14.96,-1.71,;16.29,-.94,;17.62,-1.71,;18.95,-.95,;18.95,.6,;17.62,1.37,;16.28,.6,;17.13,-.82,;18.17,.4,;10.95,-.96,;10.96,.58,;9.62,1.36,;8.29,.58,;8.3,-.97,;9.64,-1.73,;10.97,-5.58,;9.65,-10.97,;9.66,-12.52,;8.32,-13.29,;6.99,-12.52,;5.65,-13.29,;4.32,-12.52,;2.99,-13.29,;4.32,-10.98,;6.99,-10.97,;5.66,-10.2,;5.66,-8.66,;4.32,-7.89,;3.55,-6.57,;2.78,-7.9,;8.32,-10.2,;14.98,-10.95,;14.99,-12.49,;16.31,-10.18,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.794 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581210
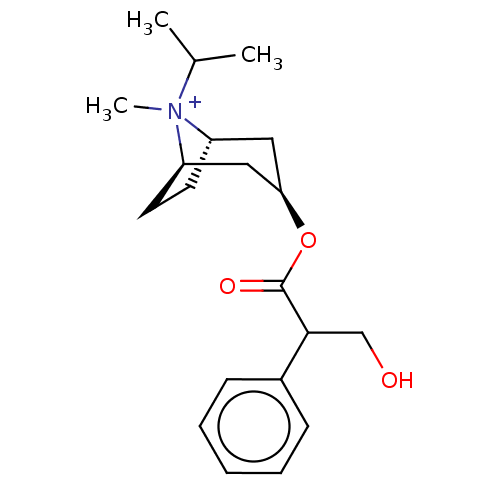
(IPRATROPIUM | Ipratropium | Ipratropium cation | I...)Show SMILES [H][C@]12CC[C@]([H])(C[C@@H](C1)OC(=O)C(CO)c1ccccc1)[N+]2(C)C(C)C |r,TLB:9:7:21:3.2,22:21:7.6.8:3.2,THB:23:21:7.6.8:3.2| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 0.794 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50581210

(IPRATROPIUM | Ipratropium | Ipratropium cation | I...)Show SMILES [H][C@]12CC[C@]([H])(C[C@@H](C1)OC(=O)C(CO)c1ccccc1)[N+]2(C)C(C)C |r,TLB:9:7:21:3.2,22:21:7.6.8:3.2,THB:23:21:7.6.8:3.2| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 0.794 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M2 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581196
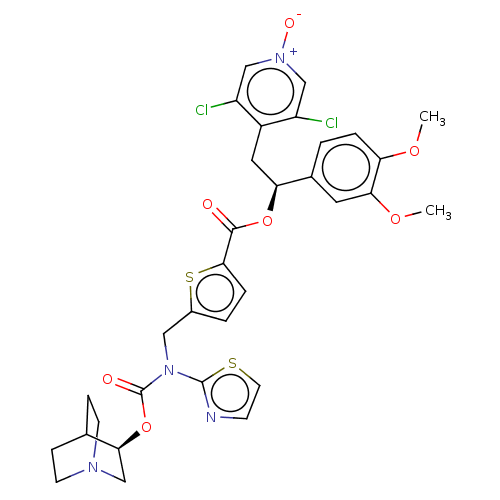
(CHEMBL5086272)Show SMILES COc1ccc(cc1OC)[C@H](Cc1c(Cl)c[n+]([O-])cc1Cl)OC(=O)c1ccc(CN(C(=O)O[C@H]2CN3CCC2CC3)c2nccs2)s1 |r,wU:10.22,wD:33.34,(21.46,-24.8,;22.8,-25.57,;24.13,-24.8,;25.46,-25.57,;26.79,-24.8,;26.79,-23.25,;25.45,-22.48,;24.13,-23.25,;22.8,-22.49,;22.8,-20.95,;28.12,-22.47,;29.46,-23.24,;30.79,-22.46,;32.12,-23.23,;32.13,-24.77,;33.45,-22.46,;33.45,-20.92,;34.77,-20.14,;32.1,-20.16,;30.77,-20.93,;29.44,-20.17,;28.12,-20.93,;26.78,-20.17,;25.45,-20.94,;26.77,-18.63,;25.52,-17.73,;25.98,-16.27,;27.52,-16.26,;28.42,-15.01,;27.79,-13.61,;28.69,-12.36,;28.05,-10.95,;30.16,-12.47,;31.12,-11.26,;32.64,-11.5,;33.6,-10.3,;32.39,-9.34,;31.86,-10.84,;30.56,-9.84,;31.52,-8.63,;33.04,-8.87,;26.25,-13.46,;25.23,-14.61,;23.82,-13.99,;23.97,-12.46,;25.47,-12.13,;28.01,-17.72,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.794 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Leucine-rich repeat serine/threonine-protein kinase 2
(Homo sapiens (Human)) | BDBM50448126
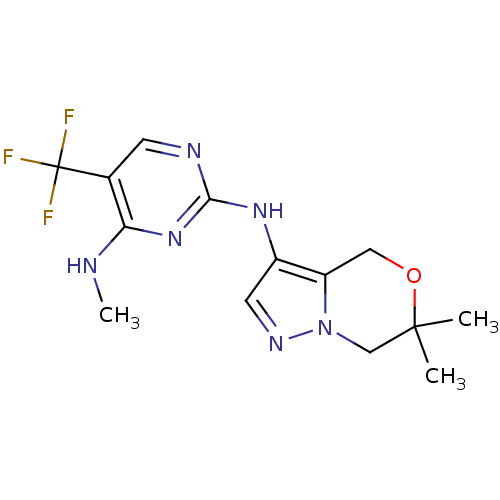
(CHEMBL3122105 | US9212186, 22)Show InChI InChI=1S/C14H17F3N6O/c1-13(2)7-23-10(6-24-13)9(5-20-23)21-12-19-4-8(14(15,16)17)11(18-3)22-12/h4-5H,6-7H2,1-3H3,(H2,18,19,21,22) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of LRRK2 (unknown origin) |
J Med Chem 57: 921-36 (2014)
Article DOI: 10.1021/jm401654j
BindingDB Entry DOI: 10.7270/Q2N0181N |
More data for this
Ligand-Target Pair | |
Leucine-rich repeat serine/threonine-protein kinase 2
(Homo sapiens (Human)) | BDBM50398676
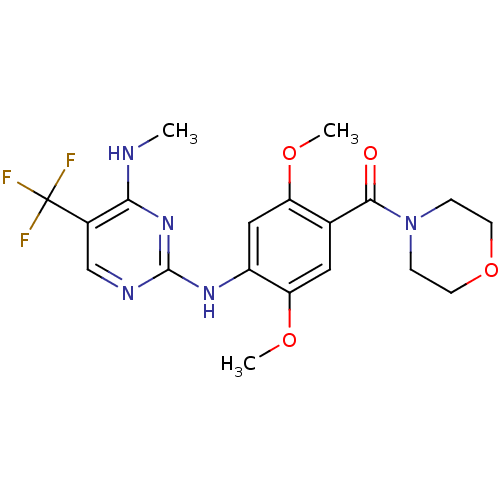
(CHEMBL2178125)Show SMILES CNc1nc(Nc2cc(OC)c(cc2OC)C(=O)N2CCOCC2)ncc1C(F)(F)F Show InChI InChI=1S/C19H22F3N5O4/c1-23-16-12(19(20,21)22)10-24-18(26-16)25-13-9-14(29-2)11(8-15(13)30-3)17(28)27-4-6-31-7-5-27/h8-10H,4-7H2,1-3H3,(H2,23,24,25,26) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of LRRK2 using FAM-LRRKtide as substrate after 120 mins by microfluidic capillary electrophoresis assay |
J Med Chem 55: 9416-33 (2012)
Article DOI: 10.1021/jm301020q
BindingDB Entry DOI: 10.7270/Q2P55PN7 |
More data for this
Ligand-Target Pair | |
Leucine-rich repeat serine/threonine-protein kinase 2
(Homo sapiens (Human)) | BDBM50398668
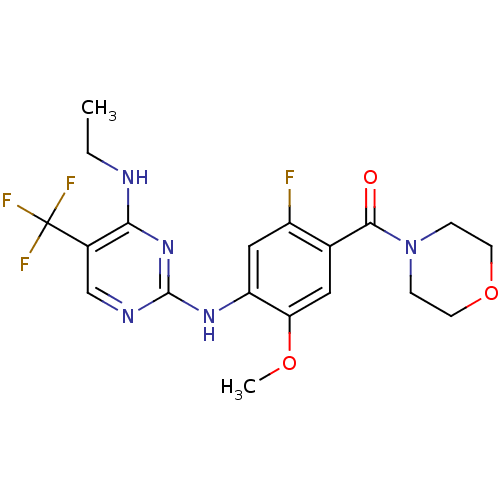
(CHEMBL2178134 | US8802674, 256)Show SMILES CCNc1nc(Nc2cc(F)c(cc2OC)C(=O)N2CCOCC2)ncc1C(F)(F)F Show InChI InChI=1S/C19H21F4N5O3/c1-3-24-16-12(19(21,22)23)10-25-18(27-16)26-14-9-13(20)11(8-15(14)30-2)17(29)28-4-6-31-7-5-28/h8-10H,3-7H2,1-2H3,(H2,24,25,26,27) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of LRRK2 using FAM-LRRKtide as substrate after 120 mins by microfluidic capillary electrophoresis assay |
J Med Chem 55: 9416-33 (2012)
Article DOI: 10.1021/jm301020q
BindingDB Entry DOI: 10.7270/Q2P55PN7 |
More data for this
Ligand-Target Pair | |
Leucine-rich repeat serine/threonine-protein kinase 2
(Homo sapiens (Human)) | BDBM50398667

(CHEMBL2178135)Show SMILES COc1cc(C(=O)N2CCOCC2)c(F)cc1Nc1ncc(c(NC2CC2)n1)C(F)(F)F Show InChI InChI=1S/C20H21F4N5O3/c1-31-16-8-12(18(30)29-4-6-32-7-5-29)14(21)9-15(16)27-19-25-10-13(20(22,23)24)17(28-19)26-11-2-3-11/h8-11H,2-7H2,1H3,(H2,25,26,27,28) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of LRRK2 using FAM-LRRKtide as substrate after 120 mins by microfluidic capillary electrophoresis assay |
J Med Chem 55: 9416-33 (2012)
Article DOI: 10.1021/jm301020q
BindingDB Entry DOI: 10.7270/Q2P55PN7 |
More data for this
Ligand-Target Pair | |
Leucine-rich repeat serine/threonine-protein kinase 2
(Homo sapiens (Human)) | BDBM50398662
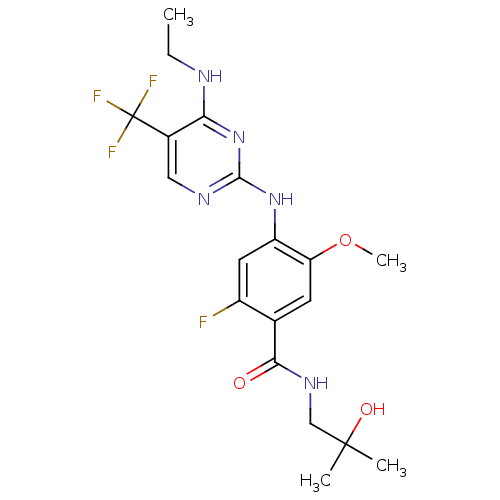
(CHEMBL2178140)Show SMILES CCNc1nc(Nc2cc(F)c(cc2OC)C(=O)NCC(C)(C)O)ncc1C(F)(F)F Show InChI InChI=1S/C19H23F4N5O3/c1-5-24-15-11(19(21,22)23)8-25-17(28-15)27-13-7-12(20)10(6-14(13)31-4)16(29)26-9-18(2,3)30/h6-8,30H,5,9H2,1-4H3,(H,26,29)(H2,24,25,27,28) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of LRRK2 using FAM-LRRKtide as substrate after 120 mins by microfluidic capillary electrophoresis assay |
J Med Chem 55: 9416-33 (2012)
Article DOI: 10.1021/jm301020q
BindingDB Entry DOI: 10.7270/Q2P55PN7 |
More data for this
Ligand-Target Pair | |
Leucine-rich repeat serine/threonine-protein kinase 2
(Homo sapiens (Human)) | BDBM50448117
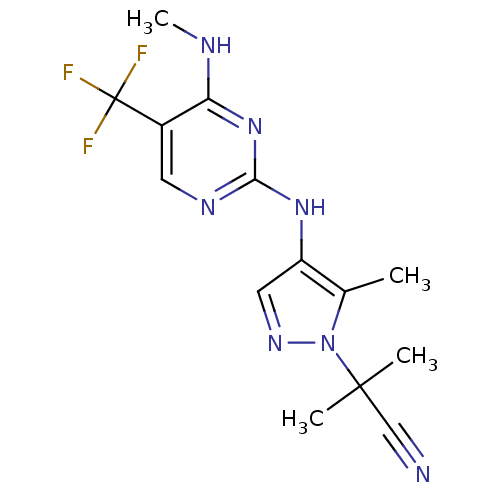
(CHEMBL3122114)Show InChI InChI=1S/C14H16F3N7/c1-8-10(6-21-24(8)13(2,3)7-18)22-12-20-5-9(14(15,16)17)11(19-4)23-12/h5-6H,1-4H3,(H2,19,20,22,23) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of LRRK2 (unknown origin) |
J Med Chem 57: 921-36 (2014)
Article DOI: 10.1021/jm401654j
BindingDB Entry DOI: 10.7270/Q2N0181N |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581182

(CHEMBL5080391)Show SMILES [O-][n+]1cc(Cl)c(C[C@H](OC(=O)c2cccc(CN(C(=O)O[C@H]3CN4CCC3CC4)c3ccccc3)c2)c2ccc(OC(F)F)c(OCC3CC3)c2)c(Cl)c1 |r,wU:7.7,wD:21.20,(80.86,-8.1,;79.53,-8.88,;78.19,-8.11,;76.86,-8.89,;75.52,-8.13,;76.88,-10.42,;75.54,-11.2,;74.21,-10.43,;74.2,-8.89,;72.87,-8.13,;71.53,-8.9,;72.86,-6.59,;71.53,-5.83,;71.52,-4.3,;72.85,-3.51,;74.19,-4.28,;75.52,-3.5,;75.51,-1.96,;76.85,-1.19,;76.84,.35,;78.18,-1.96,;79.51,-1.18,;80.85,-1.95,;82.18,-1.19,;82.18,.36,;80.84,1.13,;79.5,.36,;80.36,-1.06,;81.39,.16,;74.18,-1.2,;74.18,.34,;72.85,1.12,;71.52,.34,;71.53,-1.21,;72.86,-1.97,;74.19,-5.82,;72.88,-11.21,;72.88,-12.76,;71.55,-13.53,;70.21,-12.76,;68.88,-13.53,;67.54,-12.76,;66.21,-13.53,;67.54,-11.22,;70.21,-11.22,;68.88,-10.45,;68.88,-8.91,;67.55,-8.14,;66.78,-6.81,;66.01,-8.14,;71.54,-10.44,;78.21,-11.19,;78.21,-12.73,;79.54,-10.42,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50581180

(CHEMBL5081214)Show SMILES [O-][n+]1cc(Cl)c(C[C@H](OC(=O)c2cccc(c2)S(=O)(=O)NC(C(=O)O[C@H]2CN3CCC2CC3)c2ccccc2)c2ccc(OC(F)F)c(OCC3CC3)c2)c(Cl)c1 |r,wU:7.7,wD:25.25,(36.66,-8.55,;35.33,-9.33,;33.99,-8.57,;32.66,-9.34,;31.32,-8.58,;32.68,-10.87,;31.35,-11.65,;30.01,-10.88,;30,-9.34,;28.67,-8.58,;27.34,-9.35,;28.66,-7.04,;27.33,-6.28,;27.32,-4.75,;28.65,-3.96,;29.99,-4.73,;29.99,-6.27,;31.31,-3.95,;32.85,-3.95,;32.08,-5.28,;31.32,-2.42,;32.65,-1.64,;33.98,-2.41,;33.99,-3.95,;35.31,-1.63,;36.65,-2.4,;36.65,-3.95,;37.98,-4.71,;39.31,-3.94,;39.31,-2.4,;37.98,-1.63,;38.68,-3.13,;37.19,-3.37,;32.64,-.1,;33.98,.68,;33.98,2.22,;32.64,2.99,;31.31,2.2,;31.32,.68,;28.68,-11.66,;28.68,-13.21,;27.35,-13.98,;26.01,-13.21,;24.68,-13.98,;23.34,-13.21,;22.01,-13.98,;23.35,-11.67,;26.01,-11.67,;24.68,-10.9,;24.68,-9.36,;23.35,-8.59,;22.58,-7.26,;21.81,-8.59,;27.34,-10.9,;34.01,-11.65,;34.01,-13.19,;35.34,-10.87,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-N-methyl Scopolamine Chloride from human M3 receptor membranes incubated for 2 hrs by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00204
BindingDB Entry DOI: 10.7270/Q2DN48WC |
More data for this
Ligand-Target Pair | |
Leucine-rich repeat serine/threonine-protein kinase 2
(Homo sapiens (Human)) | BDBM50398665
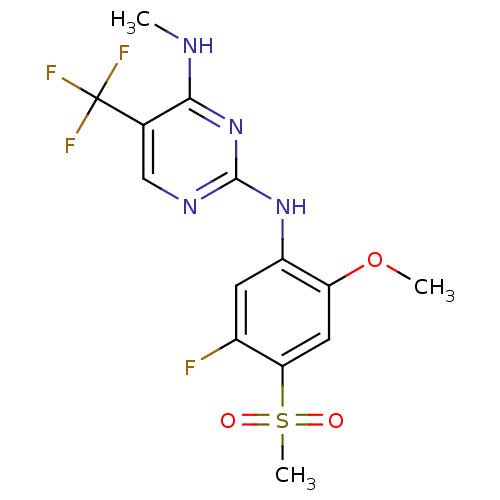
(CHEMBL2178137 | US9145402, 15)Show SMILES CNc1nc(Nc2cc(F)c(cc2OC)S(C)(=O)=O)ncc1C(F)(F)F Show InChI InChI=1S/C14H14F4N4O3S/c1-19-12-7(14(16,17)18)6-20-13(22-12)21-9-4-8(15)11(26(3,23)24)5-10(9)25-2/h4-6H,1-3H3,(H2,19,20,21,22) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of LRRK2 using FAM-LRRKtide as substrate after 120 mins by microfluidic capillary electrophoresis assay |
J Med Chem 55: 9416-33 (2012)
Article DOI: 10.1021/jm301020q
BindingDB Entry DOI: 10.7270/Q2P55PN7 |
More data for this
Ligand-Target Pair | |
Leucine-rich repeat serine/threonine-protein kinase 2
(Homo sapiens (Human)) | BDBM50396150
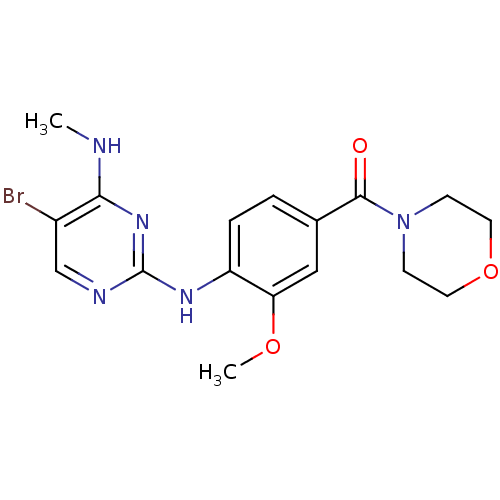
(CHEMBL2171743)Show InChI InChI=1S/C17H20BrN5O3/c1-19-15-12(18)10-20-17(22-15)21-13-4-3-11(9-14(13)25-2)16(24)23-5-7-26-8-6-23/h3-4,9-10H,5-8H2,1-2H3,(H2,19,20,21,22) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of LRRK2 using FAM-LRRKtide as substrate after 120 mins by microfluidic capillary electrophoresis assay |
J Med Chem 55: 9416-33 (2012)
Article DOI: 10.1021/jm301020q
BindingDB Entry DOI: 10.7270/Q2P55PN7 |
More data for this
Ligand-Target Pair | |
Leucine-rich repeat serine/threonine-protein kinase 2
(Homo sapiens (Human)) | BDBM50448111
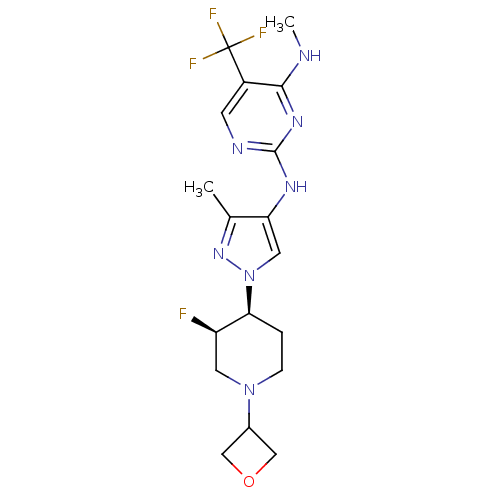
(CHEMBL3122116)Show SMILES CNc1nc(Nc2cn(nc2C)[C@H]2CCN(C[C@H]2F)C2COC2)ncc1C(F)(F)F |r| Show InChI InChI=1S/C18H23F4N7O/c1-10-14(25-17-24-5-12(18(20,21)22)16(23-2)26-17)7-29(27-10)15-3-4-28(6-13(15)19)11-8-30-9-11/h5,7,11,13,15H,3-4,6,8-9H2,1-2H3,(H2,23,24,25,26)/t13-,15+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of LRRK2 (unknown origin) |
J Med Chem 57: 921-36 (2014)
Article DOI: 10.1021/jm401654j
BindingDB Entry DOI: 10.7270/Q2N0181N |
More data for this
Ligand-Target Pair | |
Leucine-rich repeat serine/threonine-protein kinase 2
(Homo sapiens (Human)) | BDBM50396148

(CHEMBL2171745 | US8802674, 50)Show SMILES CNc1nc(Nc2ccc(cc2OC)C(=O)N2CCOCC2)ncc1C(F)(F)F Show InChI InChI=1S/C18H20F3N5O3/c1-22-15-12(18(19,20)21)10-23-17(25-15)24-13-4-3-11(9-14(13)28-2)16(27)26-5-7-29-8-6-26/h3-4,9-10H,5-8H2,1-2H3,(H2,22,23,24,25) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of LRRK2 using FAM-LRRKtide as substrate after 120 mins by microfluidic capillary electrophoresis assay |
J Med Chem 55: 9416-33 (2012)
Article DOI: 10.1021/jm301020q
BindingDB Entry DOI: 10.7270/Q2P55PN7 |
More data for this
Ligand-Target Pair | |
Leucine-rich repeat serine/threonine-protein kinase 2
(Homo sapiens (Human)) | BDBM50398677
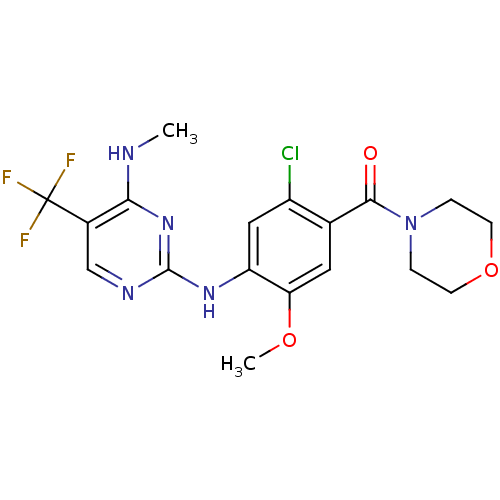
(CHEMBL2178124 | US8802674, 292)Show SMILES CNc1nc(Nc2cc(Cl)c(cc2OC)C(=O)N2CCOCC2)ncc1C(F)(F)F Show InChI InChI=1S/C18H19ClF3N5O3/c1-23-15-11(18(20,21)22)9-24-17(26-15)25-13-8-12(19)10(7-14(13)29-2)16(28)27-3-5-30-6-4-27/h7-9H,3-6H2,1-2H3,(H2,23,24,25,26) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of LRRK2 using FAM-LRRKtide as substrate after 120 mins by microfluidic capillary electrophoresis assay |
J Med Chem 55: 9416-33 (2012)
Article DOI: 10.1021/jm301020q
BindingDB Entry DOI: 10.7270/Q2P55PN7 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data