 Found 295 hits with Last Name = 'orry' and Initial = 'a'
Found 295 hits with Last Name = 'orry' and Initial = 'a' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50329179
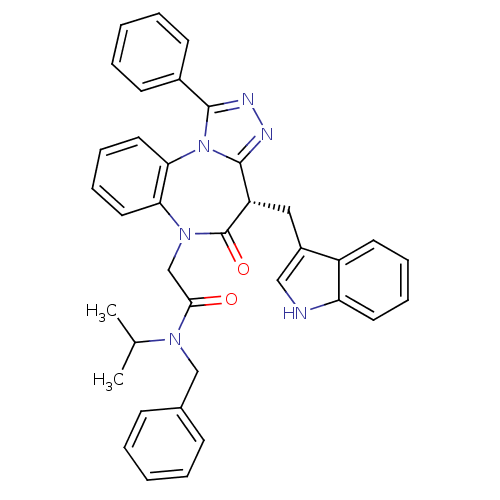
(2-((4S)-4-((1H-indol-3-yl)methyl)-5-oxo-1-phenyl-4...)Show SMILES CC(C)N(Cc1ccccc1)C(=O)CN1c2ccccc2-n2c(nnc2-c2ccccc2)[C@H](Cc2c[nH]c3ccccc23)C1=O |r| Show InChI InChI=1S/C37H34N6O2/c1-25(2)41(23-26-13-5-3-6-14-26)34(44)24-42-32-19-11-12-20-33(32)43-35(27-15-7-4-8-16-27)39-40-36(43)30(37(42)45)21-28-22-38-31-18-10-9-17-29(28)31/h3-20,22,25,30,38H,21,23-24H2,1-2H3/t30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic
Curated by ChEMBL
| Assay Description
Displacement of [125I-BDZ-2] from wild-type human CCK2R at allosteric site expressed in CHO cells after 60 mins by scintillation counter |
J Med Chem 58: 9562-77 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01110
BindingDB Entry DOI: 10.7270/Q29K4D33 |
More data for this
Ligand-Target Pair | |
Cholecystokinin receptor type A
(Homo sapiens (Human)) | BDBM50329179

(2-((4S)-4-((1H-indol-3-yl)methyl)-5-oxo-1-phenyl-4...)Show SMILES CC(C)N(Cc1ccccc1)C(=O)CN1c2ccccc2-n2c(nnc2-c2ccccc2)[C@H](Cc2c[nH]c3ccccc23)C1=O |r| Show InChI InChI=1S/C37H34N6O2/c1-25(2)41(23-26-13-5-3-6-14-26)34(44)24-42-32-19-11-12-20-33(32)43-35(27-15-7-4-8-16-27)39-40-36(43)30(37(42)45)21-28-22-38-31-18-10-9-17-29(28)31/h3-20,22,25,30,38H,21,23-24H2,1-2H3/t30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic
Curated by ChEMBL
| Assay Description
Displacement of [125I-CCK] from wild-type human CCK1R at allosteric site expressed in CHO cells after 60 mins by scintillation counter |
J Med Chem 58: 9562-77 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01110
BindingDB Entry DOI: 10.7270/Q29K4D33 |
More data for this
Ligand-Target Pair | |
Cholecystokinin receptor type A
(Homo sapiens (Human)) | BDBM50380728
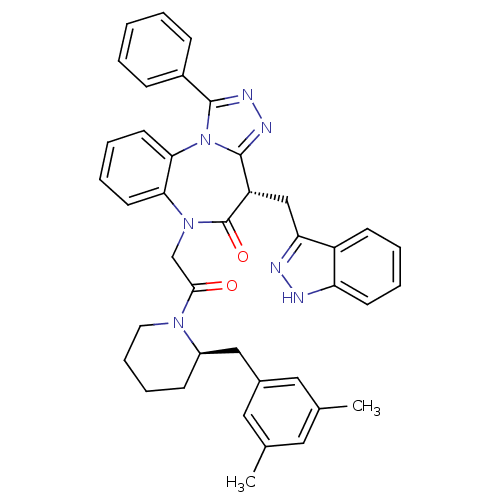
(CHEMBL2017835)Show SMILES Cc1cc(C)cc(C[C@H]2CCCCN2C(=O)CN2c3ccccc3-n3c(nnc3-c3ccccc3)[C@H](Cc3n[nH]c4ccccc34)C2=O)c1 |r| Show InChI InChI=1S/C40H39N7O2/c1-26-20-27(2)22-28(21-26)23-30-14-10-11-19-45(30)37(48)25-46-35-17-8-9-18-36(35)47-38(29-12-4-3-5-13-29)43-44-39(47)32(40(46)49)24-34-31-15-6-7-16-33(31)41-42-34/h3-9,12-13,15-18,20-22,30,32H,10-11,14,19,23-25H2,1-2H3,(H,41,42)/t30-,32+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 29 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic
Curated by ChEMBL
| Assay Description
Displacement of [125I-CCK] from wild-type human CCK1R at allosteric site expressed in CHO cells after 60 mins by scintillation counter |
J Med Chem 58: 9562-77 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01110
BindingDB Entry DOI: 10.7270/Q29K4D33 |
More data for this
Ligand-Target Pair | |
Lethal factor
(Bacillus anthracis) | BDBM8443

(2-[(5Z)-5-({5-[2-chloro-5-(trifluoromethyl)phenyl]...)Show SMILES OC(=O)CN1C(=S)S\C(=C/c2ccc(o2)-c2cc(ccc2Cl)C(F)(F)F)C1=O Show InChI InChI=1S/C17H9ClF3NO4S2/c18-11-3-1-8(17(19,20)21)5-10(11)12-4-2-9(26-12)6-13-15(25)22(7-14(23)24)16(27)28-13/h1-6H,7H2,(H,23,24)/b13-6- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 32 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Burnham Institute for Medical Research
| Assay Description
Fluorescence peptide cleavage assay was performed in a 96-well plate. Each reaction contained MAPKKide, LF, and the small-molecule inhibitor. The C-t... |
Proc Natl Acad Sci U S A 102: 9499-504 (2005)
Article DOI: 10.1073/pnas.0502733102
BindingDB Entry DOI: 10.7270/Q2TM78BQ |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50380728

(CHEMBL2017835)Show SMILES Cc1cc(C)cc(C[C@H]2CCCCN2C(=O)CN2c3ccccc3-n3c(nnc3-c3ccccc3)[C@H](Cc3n[nH]c4ccccc34)C2=O)c1 |r| Show InChI InChI=1S/C40H39N7O2/c1-26-20-27(2)22-28(21-26)23-30-14-10-11-19-45(30)37(48)25-46-35-17-8-9-18-36(35)47-38(29-12-4-3-5-13-29)43-44-39(47)32(40(46)49)24-34-31-15-6-7-16-33(31)41-42-34/h3-9,12-13,15-18,20-22,30,32H,10-11,14,19,23-25H2,1-2H3,(H,41,42)/t30-,32+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 69 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic
Curated by ChEMBL
| Assay Description
Displacement of [125I-BDZ-2] from wild-type human CCK2R at allosteric site expressed in CHO cells after 60 mins by scintillation counter |
J Med Chem 58: 9562-77 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01110
BindingDB Entry DOI: 10.7270/Q29K4D33 |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50329179

(2-((4S)-4-((1H-indol-3-yl)methyl)-5-oxo-1-phenyl-4...)Show SMILES CC(C)N(Cc1ccccc1)C(=O)CN1c2ccccc2-n2c(nnc2-c2ccccc2)[C@H](Cc2c[nH]c3ccccc23)C1=O |r| Show InChI InChI=1S/C37H34N6O2/c1-25(2)41(23-26-13-5-3-6-14-26)34(44)24-42-32-19-11-12-20-33(32)43-35(27-15-7-4-8-16-27)39-40-36(43)30(37(42)45)21-28-22-38-31-18-10-9-17-29(28)31/h3-20,22,25,30,38H,21,23-24H2,1-2H3/t30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 71 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic
Curated by ChEMBL
| Assay Description
Displacement of [125I-CCK] from wild-type human CCK2R at allosteric site expressed in CHO cells after 60 mins by scintillation counter |
J Med Chem 58: 9562-77 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01110
BindingDB Entry DOI: 10.7270/Q29K4D33 |
More data for this
Ligand-Target Pair | |
Cholecystokinin receptor type A
(Homo sapiens (Human)) | BDBM50329179

(2-((4S)-4-((1H-indol-3-yl)methyl)-5-oxo-1-phenyl-4...)Show SMILES CC(C)N(Cc1ccccc1)C(=O)CN1c2ccccc2-n2c(nnc2-c2ccccc2)[C@H](Cc2c[nH]c3ccccc23)C1=O |r| Show InChI InChI=1S/C37H34N6O2/c1-25(2)41(23-26-13-5-3-6-14-26)34(44)24-42-32-19-11-12-20-33(32)43-35(27-15-7-4-8-16-27)39-40-36(43)30(37(42)45)21-28-22-38-31-18-10-9-17-29(28)31/h3-20,22,25,30,38H,21,23-24H2,1-2H3/t30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 166 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic
Curated by ChEMBL
| Assay Description
Displacement of [125I-BDZ-1] from wild-type human CCK1R at allosteric site expressed in CHO cells after 60 mins by scintillation counter |
J Med Chem 58: 9562-77 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01110
BindingDB Entry DOI: 10.7270/Q29K4D33 |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50329179

(2-((4S)-4-((1H-indol-3-yl)methyl)-5-oxo-1-phenyl-4...)Show SMILES CC(C)N(Cc1ccccc1)C(=O)CN1c2ccccc2-n2c(nnc2-c2ccccc2)[C@H](Cc2c[nH]c3ccccc23)C1=O |r| Show InChI InChI=1S/C37H34N6O2/c1-25(2)41(23-26-13-5-3-6-14-26)34(44)24-42-32-19-11-12-20-33(32)43-35(27-15-7-4-8-16-27)39-40-36(43)30(37(42)45)21-28-22-38-31-18-10-9-17-29(28)31/h3-20,22,25,30,38H,21,23-24H2,1-2H3/t30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic
Curated by ChEMBL
| Assay Description
Displacement of [125I-BDZ-2] from wild-type human CCK2R expressed in CHO cells after 60 mins by scintillation counter |
J Med Chem 58: 9562-77 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01110
BindingDB Entry DOI: 10.7270/Q29K4D33 |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50380728

(CHEMBL2017835)Show SMILES Cc1cc(C)cc(C[C@H]2CCCCN2C(=O)CN2c3ccccc3-n3c(nnc3-c3ccccc3)[C@H](Cc3n[nH]c4ccccc34)C2=O)c1 |r| Show InChI InChI=1S/C40H39N7O2/c1-26-20-27(2)22-28(21-26)23-30-14-10-11-19-45(30)37(48)25-46-35-17-8-9-18-36(35)47-38(29-12-4-3-5-13-29)43-44-39(47)32(40(46)49)24-34-31-15-6-7-16-33(31)41-42-34/h3-9,12-13,15-18,20-22,30,32H,10-11,14,19,23-25H2,1-2H3,(H,41,42)/t30-,32+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 214 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic
Curated by ChEMBL
| Assay Description
Displacement of [125I-CCK] from wild-type human CCK2R at allosteric site expressed in CHO cells after 60 mins by scintillation counter |
J Med Chem 58: 9562-77 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01110
BindingDB Entry DOI: 10.7270/Q29K4D33 |
More data for this
Ligand-Target Pair | |
Cholecystokinin receptor type A
(Homo sapiens (Human)) | BDBM50380728

(CHEMBL2017835)Show SMILES Cc1cc(C)cc(C[C@H]2CCCCN2C(=O)CN2c3ccccc3-n3c(nnc3-c3ccccc3)[C@H](Cc3n[nH]c4ccccc34)C2=O)c1 |r| Show InChI InChI=1S/C40H39N7O2/c1-26-20-27(2)22-28(21-26)23-30-14-10-11-19-45(30)37(48)25-46-35-17-8-9-18-36(35)47-38(29-12-4-3-5-13-29)43-44-39(47)32(40(46)49)24-34-31-15-6-7-16-33(31)41-42-34/h3-9,12-13,15-18,20-22,30,32H,10-11,14,19,23-25H2,1-2H3,(H,41,42)/t30-,32+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 214 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic
Curated by ChEMBL
| Assay Description
Displacement of [125I-CCK] from wild-type human CCK1R expressed in CHO cells after 60 mins by scintillation counter |
J Med Chem 58: 9562-77 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01110
BindingDB Entry DOI: 10.7270/Q29K4D33 |
More data for this
Ligand-Target Pair | |
Cholecystokinin receptor type A
(Homo sapiens (Human)) | BDBM50329179

(2-((4S)-4-((1H-indol-3-yl)methyl)-5-oxo-1-phenyl-4...)Show SMILES CC(C)N(Cc1ccccc1)C(=O)CN1c2ccccc2-n2c(nnc2-c2ccccc2)[C@H](Cc2c[nH]c3ccccc23)C1=O |r| Show InChI InChI=1S/C37H34N6O2/c1-25(2)41(23-26-13-5-3-6-14-26)34(44)24-42-32-19-11-12-20-33(32)43-35(27-15-7-4-8-16-27)39-40-36(43)30(37(42)45)21-28-22-38-31-18-10-9-17-29(28)31/h3-20,22,25,30,38H,21,23-24H2,1-2H3/t30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 275 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic
Curated by ChEMBL
| Assay Description
Displacement of [125I-CCK] from wild-type human CCK1R expressed in CHO cells after 60 mins by scintillation counter |
J Med Chem 58: 9562-77 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01110
BindingDB Entry DOI: 10.7270/Q29K4D33 |
More data for this
Ligand-Target Pair | |
Cholecystokinin receptor type A
(Homo sapiens (Human)) | BDBM50329179

(2-((4S)-4-((1H-indol-3-yl)methyl)-5-oxo-1-phenyl-4...)Show SMILES CC(C)N(Cc1ccccc1)C(=O)CN1c2ccccc2-n2c(nnc2-c2ccccc2)[C@H](Cc2c[nH]c3ccccc23)C1=O |r| Show InChI InChI=1S/C37H34N6O2/c1-25(2)41(23-26-13-5-3-6-14-26)34(44)24-42-32-19-11-12-20-33(32)43-35(27-15-7-4-8-16-27)39-40-36(43)30(37(42)45)21-28-22-38-31-18-10-9-17-29(28)31/h3-20,22,25,30,38H,21,23-24H2,1-2H3/t30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 427 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic
Curated by ChEMBL
| Assay Description
Displacement of [125I-BDZ-1] from wild-type human CCK1R expressed in CHO cells after 60 mins by scintillation counter |
J Med Chem 58: 9562-77 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01110
BindingDB Entry DOI: 10.7270/Q29K4D33 |
More data for this
Ligand-Target Pair | |
Lethal factor
(Bacillus anthracis) | BDBM8435

(3-[(5Z)-5-{[5-(4-chlorophenyl)furan-2-yl]methylide...)Show SMILES OC(=O)CCN1C(=S)S\C(=C/c2ccc(o2)-c2ccc(Cl)cc2)C1=O Show InChI InChI=1S/C17H12ClNO4S2/c18-11-3-1-10(2-4-11)13-6-5-12(23-13)9-14-16(22)19(17(24)25-14)8-7-15(20)21/h1-6,9H,7-8H2,(H,20,21)/b14-9- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| 800 | n/a | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Burnham Institute for Medical Research
| Assay Description
Fluorescence peptide cleavage assay was performed in a 96-well plate. Each reaction contained MAPKKide, LF, and the small-molecule inhibitor. The C-t... |
Proc Natl Acad Sci U S A 102: 9499-504 (2005)
Article DOI: 10.1073/pnas.0502733102
BindingDB Entry DOI: 10.7270/Q2TM78BQ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cholecystokinin receptor type A
(Homo sapiens (Human)) | BDBM50380728

(CHEMBL2017835)Show SMILES Cc1cc(C)cc(C[C@H]2CCCCN2C(=O)CN2c3ccccc3-n3c(nnc3-c3ccccc3)[C@H](Cc3n[nH]c4ccccc34)C2=O)c1 |r| Show InChI InChI=1S/C40H39N7O2/c1-26-20-27(2)22-28(21-26)23-30-14-10-11-19-45(30)37(48)25-46-35-17-8-9-18-36(35)47-38(29-12-4-3-5-13-29)43-44-39(47)32(40(46)49)24-34-31-15-6-7-16-33(31)41-42-34/h3-9,12-13,15-18,20-22,30,32H,10-11,14,19,23-25H2,1-2H3,(H,41,42)/t30-,32+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 912 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic
Curated by ChEMBL
| Assay Description
Displacement of [125I-BDZ-1] from wild-type human CCK1R at allosteric site expressed in CHO cells after 60 mins by scintillation counter |
J Med Chem 58: 9562-77 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01110
BindingDB Entry DOI: 10.7270/Q29K4D33 |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50329179

(2-((4S)-4-((1H-indol-3-yl)methyl)-5-oxo-1-phenyl-4...)Show SMILES CC(C)N(Cc1ccccc1)C(=O)CN1c2ccccc2-n2c(nnc2-c2ccccc2)[C@H](Cc2c[nH]c3ccccc23)C1=O |r| Show InChI InChI=1S/C37H34N6O2/c1-25(2)41(23-26-13-5-3-6-14-26)34(44)24-42-32-19-11-12-20-33(32)43-35(27-15-7-4-8-16-27)39-40-36(43)30(37(42)45)21-28-22-38-31-18-10-9-17-29(28)31/h3-20,22,25,30,38H,21,23-24H2,1-2H3/t30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| <1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic
Curated by ChEMBL
| Assay Description
Displacement of [125I-CCK] from wild-type human CCK2R expressed in CHO cells after 60 mins by scintillation counter |
J Med Chem 58: 9562-77 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01110
BindingDB Entry DOI: 10.7270/Q29K4D33 |
More data for this
Ligand-Target Pair | |
Cholecystokinin receptor type A
(Homo sapiens (Human)) | BDBM50380728

(CHEMBL2017835)Show SMILES Cc1cc(C)cc(C[C@H]2CCCCN2C(=O)CN2c3ccccc3-n3c(nnc3-c3ccccc3)[C@H](Cc3n[nH]c4ccccc34)C2=O)c1 |r| Show InChI InChI=1S/C40H39N7O2/c1-26-20-27(2)22-28(21-26)23-30-14-10-11-19-45(30)37(48)25-46-35-17-8-9-18-36(35)47-38(29-12-4-3-5-13-29)43-44-39(47)32(40(46)49)24-34-31-15-6-7-16-33(31)41-42-34/h3-9,12-13,15-18,20-22,30,32H,10-11,14,19,23-25H2,1-2H3,(H,41,42)/t30-,32+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| <1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic
Curated by ChEMBL
| Assay Description
Displacement of [125I-BDZ-1] from wild-type human CCK1R expressed in CHO cells after 60 mins by scintillation counter |
J Med Chem 58: 9562-77 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01110
BindingDB Entry DOI: 10.7270/Q29K4D33 |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50380728

(CHEMBL2017835)Show SMILES Cc1cc(C)cc(C[C@H]2CCCCN2C(=O)CN2c3ccccc3-n3c(nnc3-c3ccccc3)[C@H](Cc3n[nH]c4ccccc34)C2=O)c1 |r| Show InChI InChI=1S/C40H39N7O2/c1-26-20-27(2)22-28(21-26)23-30-14-10-11-19-45(30)37(48)25-46-35-17-8-9-18-36(35)47-38(29-12-4-3-5-13-29)43-44-39(47)32(40(46)49)24-34-31-15-6-7-16-33(31)41-42-34/h3-9,12-13,15-18,20-22,30,32H,10-11,14,19,23-25H2,1-2H3,(H,41,42)/t30-,32+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.26E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic
Curated by ChEMBL
| Assay Description
Displacement of [125I-BDZ-2] from wild-type human CCK2R expressed in CHO cells after 60 mins by scintillation counter |
J Med Chem 58: 9562-77 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01110
BindingDB Entry DOI: 10.7270/Q29K4D33 |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50380728

(CHEMBL2017835)Show SMILES Cc1cc(C)cc(C[C@H]2CCCCN2C(=O)CN2c3ccccc3-n3c(nnc3-c3ccccc3)[C@H](Cc3n[nH]c4ccccc34)C2=O)c1 |r| Show InChI InChI=1S/C40H39N7O2/c1-26-20-27(2)22-28(21-26)23-30-14-10-11-19-45(30)37(48)25-46-35-17-8-9-18-36(35)47-38(29-12-4-3-5-13-29)43-44-39(47)32(40(46)49)24-34-31-15-6-7-16-33(31)41-42-34/h3-9,12-13,15-18,20-22,30,32H,10-11,14,19,23-25H2,1-2H3,(H,41,42)/t30-,32+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.41E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic
Curated by ChEMBL
| Assay Description
Displacement of [125I-CCK] from wild-type human CCK2R expressed in CHO cells after 60 mins by scintillation counter |
J Med Chem 58: 9562-77 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01110
BindingDB Entry DOI: 10.7270/Q29K4D33 |
More data for this
Ligand-Target Pair | |
Melanin-concentrating hormone receptor 1
(Homo sapiens (Human)) | BDBM50232469

(CHEMBL252145 | N,N-bis(tosylmethyl)hexan-1-amine)Show SMILES CCCCCCN(CS(=O)(=O)c1ccc(C)cc1)CS(=O)(=O)c1ccc(C)cc1 Show InChI InChI=1S/C22H31NO4S2/c1-4-5-6-7-16-23(17-28(24,25)21-12-8-19(2)9-13-21)18-29(26,27)22-14-10-20(3)11-15-22/h8-15H,4-7,16-18H2,1-3H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
MolSoft LLC
Curated by ChEMBL
| Assay Description
Displacement of [125I]MCH from human MCHR1 expressed in CHO cell membranes |
J Med Chem 51: 581-8 (2008)
Article DOI: 10.1021/jm070759m
BindingDB Entry DOI: 10.7270/Q2H994Z0 |
More data for this
Ligand-Target Pair | |
Melanin-concentrating hormone receptor 1
(Homo sapiens (Human)) | BDBM50232467

(CHEMBL399303 | phenyl-N,N-bis(tosylmethyl)methanam...)Show SMILES Cc1ccc(cc1)S(=O)(=O)CN(Cc1ccccc1)CS(=O)(=O)c1ccc(C)cc1 Show InChI InChI=1S/C23H25NO4S2/c1-19-8-12-22(13-9-19)29(25,26)17-24(16-21-6-4-3-5-7-21)18-30(27,28)23-14-10-20(2)11-15-23/h3-15H,16-18H2,1-2H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 7.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
MolSoft LLC
Curated by ChEMBL
| Assay Description
Displacement of [125I]MCH from human MCHR1 expressed in CHO cell membranes |
J Med Chem 51: 581-8 (2008)
Article DOI: 10.1021/jm070759m
BindingDB Entry DOI: 10.7270/Q2H994Z0 |
More data for this
Ligand-Target Pair | |
Melanin-concentrating hormone receptor 1
(Homo sapiens (Human)) | BDBM50232470
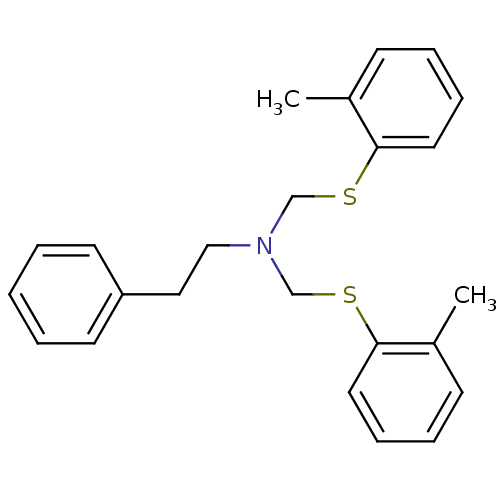
(2-phenyl-N,N-bis(o-tolylthiomethyl)ethanamine | CH...)Show InChI InChI=1S/C24H27NS2/c1-20-10-6-8-14-23(20)26-18-25(17-16-22-12-4-3-5-13-22)19-27-24-15-9-7-11-21(24)2/h3-15H,16-19H2,1-2H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 7.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
MolSoft LLC
Curated by ChEMBL
| Assay Description
Displacement of [125I]MCH from human MCHR1 expressed in CHO cell membranes |
J Med Chem 51: 581-8 (2008)
Article DOI: 10.1021/jm070759m
BindingDB Entry DOI: 10.7270/Q2H994Z0 |
More data for this
Ligand-Target Pair | |
Melanin-concentrating hormone receptor 1
(Homo sapiens (Human)) | BDBM50232468

(6-(((3-chlorobenzyl)(methyl)amino)methyl)-N2-o-tol...)Show InChI InChI=1S/C19H21ClN6/c1-13-6-3-4-9-16(13)22-19-24-17(23-18(21)25-19)12-26(2)11-14-7-5-8-15(20)10-14/h3-10H,11-12H2,1-2H3,(H3,21,22,23,24,25) | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
MolSoft LLC
Curated by ChEMBL
| Assay Description
Displacement of [125I]MCH from human MCHR1 expressed in CHO cell membranes |
J Med Chem 51: 581-8 (2008)
Article DOI: 10.1021/jm070759m
BindingDB Entry DOI: 10.7270/Q2H994Z0 |
More data for this
Ligand-Target Pair | |
Melanin-concentrating hormone receptor 1
(Homo sapiens (Human)) | BDBM50232466

(2-(dimethylamino)ethyl 4-(5-(4-methoxybenzylidene)...)Show SMILES COc1ccc(\C=C2\SC(=S)N(CCCC(=O)OCCN(C)C)C2=O)cc1 Show InChI InChI=1S/C19H24N2O4S2/c1-20(2)11-12-25-17(22)5-4-10-21-18(23)16(27-19(21)26)13-14-6-8-15(24-3)9-7-14/h6-9,13H,4-5,10-12H2,1-3H3/b16-13+ | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 8.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
MolSoft LLC
Curated by ChEMBL
| Assay Description
Displacement of [125I]MCH from human MCHR1 expressed in CHO cell membranes |
J Med Chem 51: 581-8 (2008)
Article DOI: 10.1021/jm070759m
BindingDB Entry DOI: 10.7270/Q2H994Z0 |
More data for this
Ligand-Target Pair | |
Melanin-concentrating hormone receptor 1
(Homo sapiens (Human)) | BDBM50232465
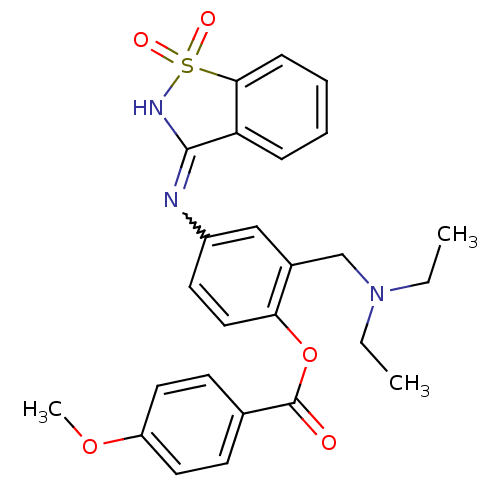
(4-methoxy-benzoic acid 2-diethylaminomethyl-4-(1,1...)Show SMILES CCN(CC)Cc1cc(ccc1OC(=O)c1ccc(OC)cc1)N=C1NS(=O)(=O)c2ccccc12 |w:23.24| Show InChI InChI=1S/C26H27N3O5S/c1-4-29(5-2)17-19-16-20(27-25-22-8-6-7-9-24(22)35(31,32)28-25)12-15-23(19)34-26(30)18-10-13-21(33-3)14-11-18/h6-16H,4-5,17H2,1-3H3,(H,27,28) | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 1.13E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
MolSoft LLC
Curated by ChEMBL
| Assay Description
Displacement of [125I]MCH from human MCHR1 expressed in CHO cell membranes |
J Med Chem 51: 581-8 (2008)
Article DOI: 10.1021/jm070759m
BindingDB Entry DOI: 10.7270/Q2H994Z0 |
More data for this
Ligand-Target Pair | |
Melanin-concentrating hormone receptor 1
(Homo sapiens (Human)) | BDBM50232464

(CHEMBL251947 | {3-[2-(4-bromo-phenyl)-benzo[d]imid...)Show SMILES CCN(CC)CCCn1c(cn2c1nc1ccccc21)-c1ccc(Br)cc1 Show InChI InChI=1S/C22H25BrN4/c1-3-25(4-2)14-7-15-26-21(17-10-12-18(23)13-11-17)16-27-20-9-6-5-8-19(20)24-22(26)27/h5-6,8-13,16H,3-4,7,14-15H2,1-2H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 1.19E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
MolSoft LLC
Curated by ChEMBL
| Assay Description
Displacement of [125I]MCH from human MCHR1 expressed in CHO cell membranes |
J Med Chem 51: 581-8 (2008)
Article DOI: 10.1021/jm070759m
BindingDB Entry DOI: 10.7270/Q2H994Z0 |
More data for this
Ligand-Target Pair | |
Melanin-concentrating hormone receptor 1
(Homo sapiens (Human)) | BDBM50232471

(CHEMBL251948 | N-(3-(benzyl(isopropyl)amino)propyl...)Show InChI InChI=1S/C21H27N3OS/c1-16(2)24(15-17-8-5-4-6-9-17)12-7-11-22-21(25)19-14-20-18(23(19)3)10-13-26-20/h4-6,8-10,13-14,16H,7,11-12,15H2,1-3H3,(H,22,25) | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 2.09E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
MolSoft LLC
Curated by ChEMBL
| Assay Description
Displacement of [125I]MCH from human MCHR1 expressed in CHO cell membranes |
J Med Chem 51: 581-8 (2008)
Article DOI: 10.1021/jm070759m
BindingDB Entry DOI: 10.7270/Q2H994Z0 |
More data for this
Ligand-Target Pair | |
Cholecystokinin receptor type A
(Homo sapiens (Human)) | BDBM50380728

(CHEMBL2017835)Show SMILES Cc1cc(C)cc(C[C@H]2CCCCN2C(=O)CN2c3ccccc3-n3c(nnc3-c3ccccc3)[C@H](Cc3n[nH]c4ccccc34)C2=O)c1 |r| Show InChI InChI=1S/C40H39N7O2/c1-26-20-27(2)22-28(21-26)23-30-14-10-11-19-45(30)37(48)25-46-35-17-8-9-18-36(35)47-38(29-12-4-3-5-13-29)43-44-39(47)32(40(46)49)24-34-31-15-6-7-16-33(31)41-42-34/h3-9,12-13,15-18,20-22,30,32H,10-11,14,19,23-25H2,1-2H3,(H,41,42)/t30-,32+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.813 | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic
Curated by ChEMBL
| Assay Description
Displacement of [125I-CCK] from human CCK1R N2.61T mutant expressed in CHO cells after 60 mins by scintillation counter |
J Med Chem 58: 9562-77 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01110
BindingDB Entry DOI: 10.7270/Q29K4D33 |
More data for this
Ligand-Target Pair | |
Cholecystokinin receptor type A
(Homo sapiens (Human)) | BDBM50380728

(CHEMBL2017835)Show SMILES Cc1cc(C)cc(C[C@H]2CCCCN2C(=O)CN2c3ccccc3-n3c(nnc3-c3ccccc3)[C@H](Cc3n[nH]c4ccccc34)C2=O)c1 |r| Show InChI InChI=1S/C40H39N7O2/c1-26-20-27(2)22-28(21-26)23-30-14-10-11-19-45(30)37(48)25-46-35-17-8-9-18-36(35)47-38(29-12-4-3-5-13-29)43-44-39(47)32(40(46)49)24-34-31-15-6-7-16-33(31)41-42-34/h3-9,12-13,15-18,20-22,30,32H,10-11,14,19,23-25H2,1-2H3,(H,41,42)/t30-,32+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic
Curated by ChEMBL
| Assay Description
Displacement of [125I-CCK] from human CCK1R W6.48A mutant expressed in CHO cells after 60 mins by scintillation counter |
J Med Chem 58: 9562-77 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01110
BindingDB Entry DOI: 10.7270/Q29K4D33 |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50329179

(2-((4S)-4-((1H-indol-3-yl)methyl)-5-oxo-1-phenyl-4...)Show SMILES CC(C)N(Cc1ccccc1)C(=O)CN1c2ccccc2-n2c(nnc2-c2ccccc2)[C@H](Cc2c[nH]c3ccccc23)C1=O |r| Show InChI InChI=1S/C37H34N6O2/c1-25(2)41(23-26-13-5-3-6-14-26)34(44)24-42-32-19-11-12-20-33(32)43-35(27-15-7-4-8-16-27)39-40-36(43)30(37(42)45)21-28-22-38-31-18-10-9-17-29(28)31/h3-20,22,25,30,38H,21,23-24H2,1-2H3/t30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic
Curated by ChEMBL
| Assay Description
Displacement of [125I-CCK] from human CCK2R H7.39L mutant expressed in CHO cells after 60 mins by scintillation counter |
J Med Chem 58: 9562-77 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01110
BindingDB Entry DOI: 10.7270/Q29K4D33 |
More data for this
Ligand-Target Pair | |
Cholecystokinin receptor type A
(Homo sapiens (Human)) | BDBM50329179

(2-((4S)-4-((1H-indol-3-yl)methyl)-5-oxo-1-phenyl-4...)Show SMILES CC(C)N(Cc1ccccc1)C(=O)CN1c2ccccc2-n2c(nnc2-c2ccccc2)[C@H](Cc2c[nH]c3ccccc23)C1=O |r| Show InChI InChI=1S/C37H34N6O2/c1-25(2)41(23-26-13-5-3-6-14-26)34(44)24-42-32-19-11-12-20-33(32)43-35(27-15-7-4-8-16-27)39-40-36(43)30(37(42)45)21-28-22-38-31-18-10-9-17-29(28)31/h3-20,22,25,30,38H,21,23-24H2,1-2H3/t30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic
Curated by ChEMBL
| Assay Description
Displacement of [125I-CCK] from human CCK1R N2.61T mutant expressed in CHO cells after 60 mins by scintillation counter |
J Med Chem 58: 9562-77 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01110
BindingDB Entry DOI: 10.7270/Q29K4D33 |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50329179

(2-((4S)-4-((1H-indol-3-yl)methyl)-5-oxo-1-phenyl-4...)Show SMILES CC(C)N(Cc1ccccc1)C(=O)CN1c2ccccc2-n2c(nnc2-c2ccccc2)[C@H](Cc2c[nH]c3ccccc23)C1=O |r| Show InChI InChI=1S/C37H34N6O2/c1-25(2)41(23-26-13-5-3-6-14-26)34(44)24-42-32-19-11-12-20-33(32)43-35(27-15-7-4-8-16-27)39-40-36(43)30(37(42)45)21-28-22-38-31-18-10-9-17-29(28)31/h3-20,22,25,30,38H,21,23-24H2,1-2H3/t30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic
Curated by ChEMBL
| Assay Description
Displacement of [125I-BDZ-2] from human CCK2R H7.39L mutant expressed in CHO cells after 60 mins by scintillation counter |
J Med Chem 58: 9562-77 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01110
BindingDB Entry DOI: 10.7270/Q29K4D33 |
More data for this
Ligand-Target Pair | |
Cholecystokinin receptor type A
(Homo sapiens (Human)) | BDBM50329179

(2-((4S)-4-((1H-indol-3-yl)methyl)-5-oxo-1-phenyl-4...)Show SMILES CC(C)N(Cc1ccccc1)C(=O)CN1c2ccccc2-n2c(nnc2-c2ccccc2)[C@H](Cc2c[nH]c3ccccc23)C1=O |r| Show InChI InChI=1S/C37H34N6O2/c1-25(2)41(23-26-13-5-3-6-14-26)34(44)24-42-32-19-11-12-20-33(32)43-35(27-15-7-4-8-16-27)39-40-36(43)30(37(42)45)21-28-22-38-31-18-10-9-17-29(28)31/h3-20,22,25,30,38H,21,23-24H2,1-2H3/t30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic
Curated by ChEMBL
| Assay Description
Agonist activity at human CCK1R V3.36A mutant expressed in CHO cells assessed as intracellular calcium response by fluorescence analysis |
J Med Chem 58: 9562-77 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01110
BindingDB Entry DOI: 10.7270/Q29K4D33 |
More data for this
Ligand-Target Pair | |
Cholecystokinin receptor type A
(Homo sapiens (Human)) | BDBM50329179

(2-((4S)-4-((1H-indol-3-yl)methyl)-5-oxo-1-phenyl-4...)Show SMILES CC(C)N(Cc1ccccc1)C(=O)CN1c2ccccc2-n2c(nnc2-c2ccccc2)[C@H](Cc2c[nH]c3ccccc23)C1=O |r| Show InChI InChI=1S/C37H34N6O2/c1-25(2)41(23-26-13-5-3-6-14-26)34(44)24-42-32-19-11-12-20-33(32)43-35(27-15-7-4-8-16-27)39-40-36(43)30(37(42)45)21-28-22-38-31-18-10-9-17-29(28)31/h3-20,22,25,30,38H,21,23-24H2,1-2H3/t30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic
Curated by ChEMBL
| Assay Description
Displacement of [125I-BDZ-1] from human CCK1R T3.28V, T3.29S mutant expressed in CHO cells after 60 mins by scintillation counter |
J Med Chem 58: 9562-77 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01110
BindingDB Entry DOI: 10.7270/Q29K4D33 |
More data for this
Ligand-Target Pair | |
Apoptosis regulator Bcl-2
(Homo sapiens (Human)) | BDBM608721
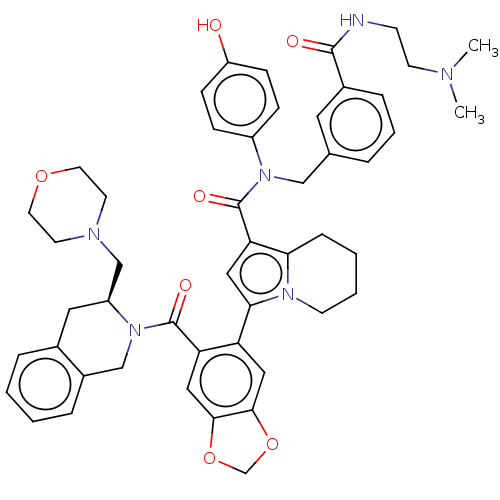
(US11697650, Compound 6 | US11697650, Example 31)Show SMILES CN(C)CCNC(=O)c1cccc(CN(C(=O)c2cc(-c3cc4OCOc4cc3C(=O)N3Cc4ccccc4C[C@H]3CN3CCOCC3)n3CCCCc23)c2ccc(O)cc2)c1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21V5K2C |
More data for this
Ligand-Target Pair | |
Apoptosis regulator Bcl-2
(Homo sapiens (Human)) | BDBM608720
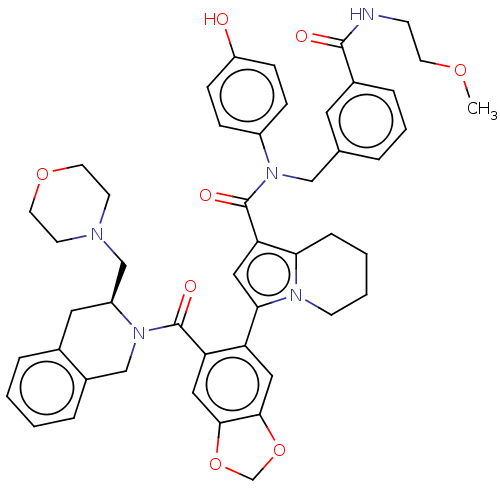
(US11697650, Compound 2 | US11697650, Example 30)Show SMILES COCCNC(=O)c1cccc(CN(C(=O)c2cc(-c3cc4OCOc4cc3C(=O)N3Cc4ccccc4C[C@H]3CN3CCOCC3)n3CCCCc23)c2ccc(O)cc2)c1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21V5K2C |
More data for this
Ligand-Target Pair | |
Apoptosis regulator Bcl-2
(Homo sapiens (Human)) | BDBM608719
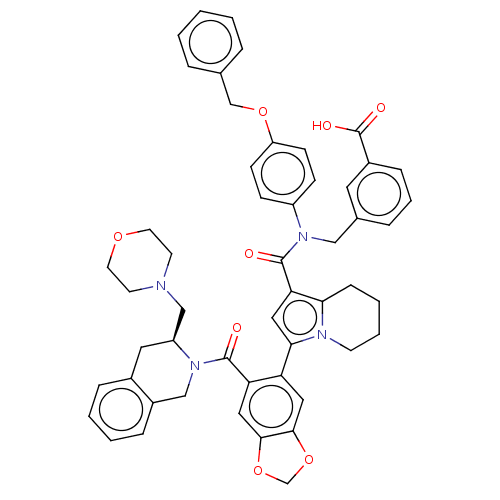
(US11697650, Compound 7 | US11697650, Example 28)Show SMILES OC(=O)c1cccc(CN(C(=O)c2cc(-c3cc4OCOc4cc3C(=O)N3Cc4ccccc4C[C@H]3CN3CCOCC3)n3CCCCc23)c2ccc(OCc3ccccc3)cc2)c1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21V5K2C |
More data for this
Ligand-Target Pair | |
Apoptosis regulator Bcl-2
(Homo sapiens (Human)) | BDBM608716
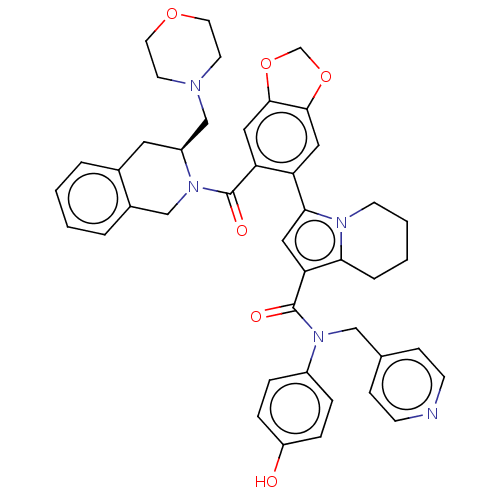
(US11697650, Compound 9 | US11697650, Example 25)Show SMILES Oc1ccc(cc1)N(Cc1ccncc1)C(=O)c1cc(-c2cc3OCOc3cc2C(=O)N2Cc3ccccc3C[C@H]2CN2CCOCC2)n2CCCCc12 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21V5K2C |
More data for this
Ligand-Target Pair | |
Apoptosis regulator Bcl-2
(Homo sapiens (Human)) | BDBM608706
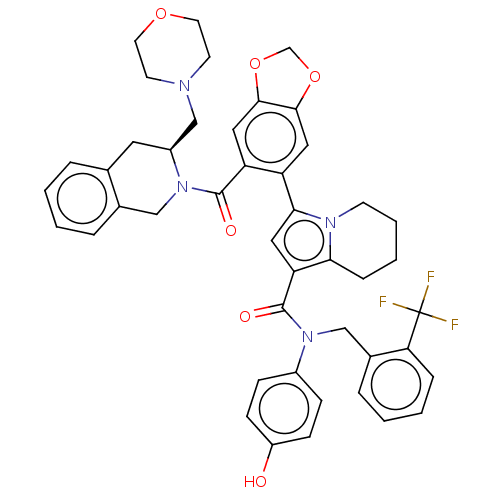
(US11697650, Compound 5 | US11697650, Example 15)Show SMILES Oc1ccc(cc1)N(Cc1ccccc1C(F)(F)F)C(=O)c1cc(-c2cc3OCOc3cc2C(=O)N2Cc3ccccc3C[C@H]2CN2CCOCC2)n2CCCCc12 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21V5K2C |
More data for this
Ligand-Target Pair | |
Apoptosis regulator Bcl-2
(Homo sapiens (Human)) | BDBM608704
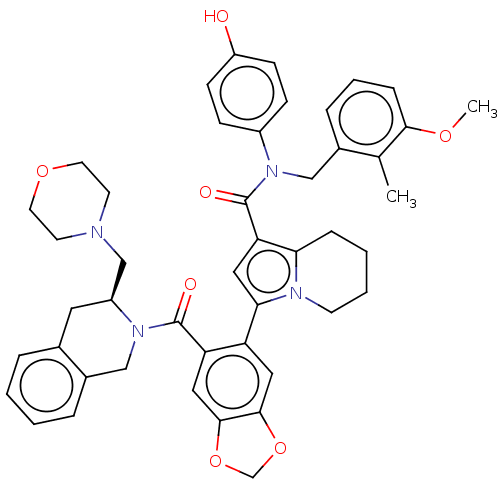
(US11697650, Compound 3 | US11697650, Example 13)Show SMILES COc1cccc(CN(C(=O)c2cc(-c3cc4OCOc4cc3C(=O)N3Cc4ccccc4C[C@H]3CN3CCOCC3)n3CCCCc23)c2ccc(O)cc2)c1C |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21V5K2C |
More data for this
Ligand-Target Pair | |
Apoptosis regulator Bcl-2
(Homo sapiens (Human)) | BDBM608702
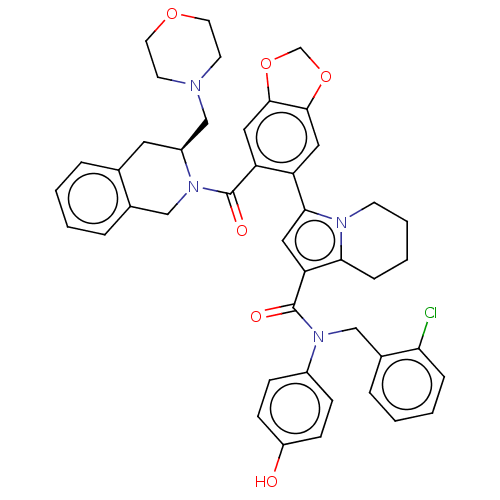
(US11697650, Compound 33 | US11697650, Example 11)Show SMILES Oc1ccc(cc1)N(Cc1ccccc1Cl)C(=O)c1cc(-c2cc3OCOc3cc2C(=O)N2Cc3ccccc3C[C@H]2CN2CCOCC2)n2CCCCc12 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21V5K2C |
More data for this
Ligand-Target Pair | |
Apoptosis regulator Bcl-2
(Homo sapiens (Human)) | BDBM608700

(N-(4-hydroxyphenyl)-N-(2-methoxybenzyl)-3-(6-{[(3S...)Show SMILES COc1ccccc1CN(C(=O)c1cc(-c2cc3OCOc3cc2C(=O)N2Cc3ccccc3C[C@H]2CN2CCOCC2)n2CCCCc12)c1ccc(O)cc1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21V5K2C |
More data for this
Ligand-Target Pair | |
Apoptosis regulator Bcl-2
(Homo sapiens (Human)) | BDBM608413
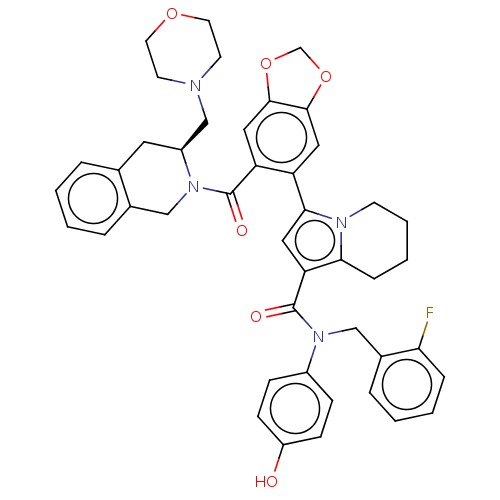
(N-(2-fluorobenzyl)-N-(4-hydroxyphenyl)-3-(6-{[(3S)...)Show SMILES Oc1ccc(cc1)N(Cc1ccccc1F)C(=O)c1cc(-c2cc3OCOc3cc2C(=O)N2Cc3ccccc3C[C@H]2CN2CCOCC2)n2CCCCc12 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21V5K2C |
More data for this
Ligand-Target Pair | |
Apoptosis regulator Bcl-2
(Homo sapiens (Human)) | BDBM608408
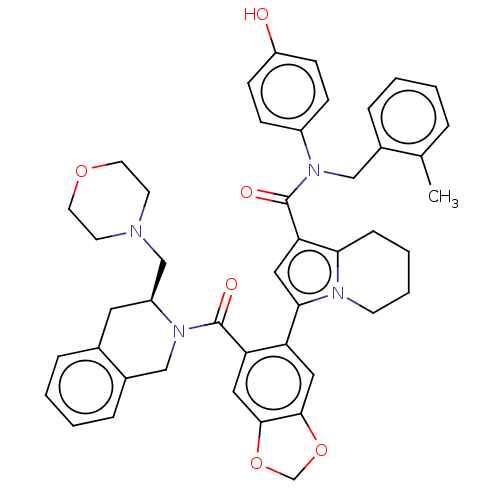
(N-(4-hydroxyphenyl)-N-(2-methylbenzyl)-3-(6-{[(3S)...)Show SMILES Cc1ccccc1CN(C(=O)c1cc(-c2cc3OCOc3cc2C(=O)N2Cc3ccccc3C[C@H]2CN2CCOCC2)n2CCCCc12)c1ccc(O)cc1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21V5K2C |
More data for this
Ligand-Target Pair | |
Apoptosis regulator Bcl-2
(Homo sapiens (Human)) | BDBM608731
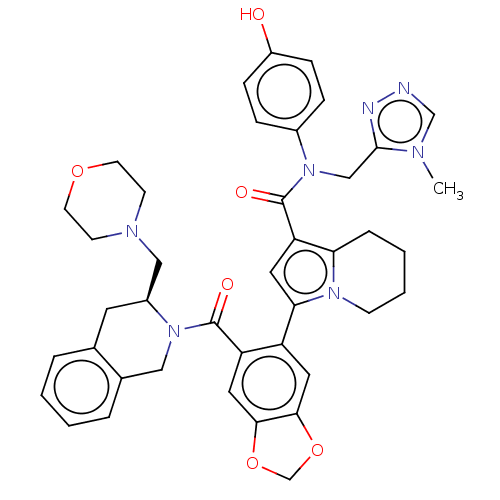
(US11697650, Compound 48 | US11697650, Example 42)Show SMILES Cn1cnnc1CN(C(=O)c1cc(-c2cc3OCOc3cc2C(=O)N2Cc3ccccc3C[C@H]2CN2CCOCC2)n2CCCCc12)c1ccc(O)cc1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21V5K2C |
More data for this
Ligand-Target Pair | |
Apoptosis regulator Bcl-2
(Homo sapiens (Human)) | BDBM608725
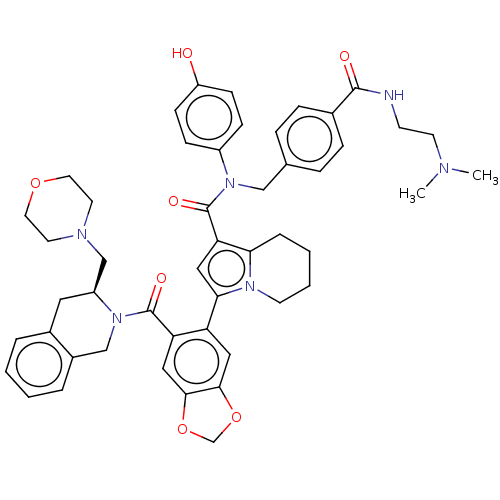
(US11697650, Compound 8 | US11697650, Example 37)Show SMILES CN(C)CCNC(=O)c1ccc(CN(C(=O)c2cc(-c3cc4OCOc4cc3C(=O)N3Cc4ccccc4C[C@H]3CN3CCOCC3)n3CCCCc23)c2ccc(O)cc2)cc1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21V5K2C |
More data for this
Ligand-Target Pair | |
Apoptosis regulator Bcl-2
(Homo sapiens (Human)) | BDBM608724

(US11697650, Compound 1 | US11697650, Example 35)Show SMILES COCCNC(=O)c1ccc(CN(C(=O)c2cc(-c3cc4OCOc4cc3C(=O)N3Cc4ccccc4C[C@H]3CN3CCOCC3)n3CCCCc23)c2ccc(O)cc2)cc1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21V5K2C |
More data for this
Ligand-Target Pair | |
Cholecystokinin receptor type A
(Homo sapiens (Human)) | BDBM50380728

(CHEMBL2017835)Show SMILES Cc1cc(C)cc(C[C@H]2CCCCN2C(=O)CN2c3ccccc3-n3c(nnc3-c3ccccc3)[C@H](Cc3n[nH]c4ccccc34)C2=O)c1 |r| Show InChI InChI=1S/C40H39N7O2/c1-26-20-27(2)22-28(21-26)23-30-14-10-11-19-45(30)37(48)25-46-35-17-8-9-18-36(35)47-38(29-12-4-3-5-13-29)43-44-39(47)32(40(46)49)24-34-31-15-6-7-16-33(31)41-42-34/h3-9,12-13,15-18,20-22,30,32H,10-11,14,19,23-25H2,1-2H3,(H,41,42)/t30-,32+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic
Curated by ChEMBL
| Assay Description
Displacement of [125I-BDZ-1] from human CCK1R T3.28V, T3.29S mutant expressed in CHO cells after 60 mins by scintillation counter |
J Med Chem 58: 9562-77 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01110
BindingDB Entry DOI: 10.7270/Q29K4D33 |
More data for this
Ligand-Target Pair | |
Apoptosis regulator Bcl-2
(Homo sapiens (Human)) | BDBM608723
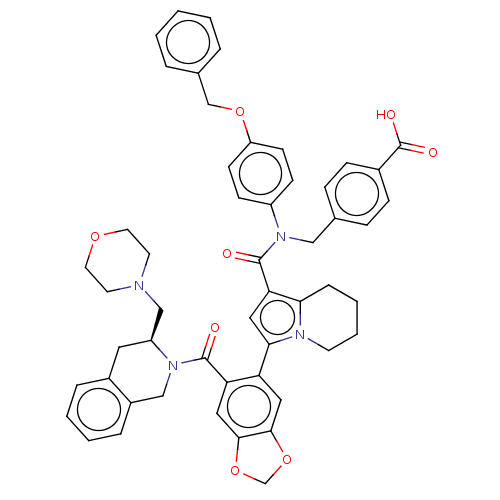
(US11697650, Compound 13 | US11697650, Example 33)Show SMILES OC(=O)c1ccc(CN(C(=O)c2cc(-c3cc4OCOc4cc3C(=O)N3Cc4ccccc4C[C@H]3CN3CCOCC3)n3CCCCc23)c2ccc(OCc3ccccc3)cc2)cc1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 7.5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21V5K2C |
More data for this
Ligand-Target Pair | |
Apoptosis regulator Bcl-2
(Homo sapiens (Human)) | BDBM608406
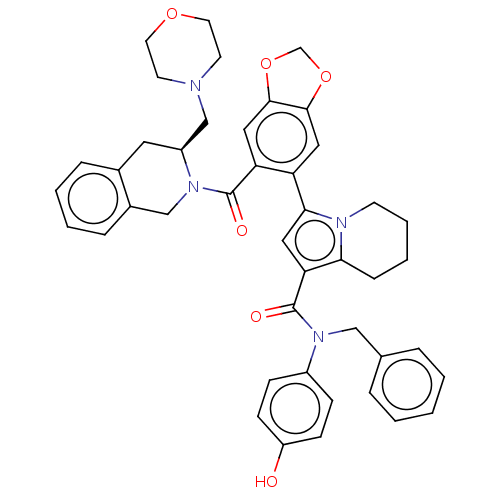
(N-benzyl-N-(4-hydroxyphenyl)-3-(6-{[(3S)-3-(morpho...)Show SMILES Oc1ccc(cc1)N(Cc1ccccc1)C(=O)c1cc(-c2cc3OCOc3cc2C(=O)N2Cc3ccccc3C[C@H]2CN2CCOCC2)n2CCCCc12 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 7.5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21V5K2C |
More data for this
Ligand-Target Pair | |
Apoptosis regulator Bcl-2
(Homo sapiens (Human)) | BDBM608703

(US11697650, Compound 19 | US11697650, Example 12)Show SMILES Oc1ccc(cc1)N(Cc1c(F)cccc1F)C(=O)c1cc(-c2cc3OCOc3cc2C(=O)N2Cc3ccccc3C[C@H]2CN2CCOCC2)n2CCCCc12 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 7.5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21V5K2C |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data