 Found 223 hits with Last Name = 'stamper' and Initial = 'm'
Found 223 hits with Last Name = 'stamper' and Initial = 'm' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Protein kinase C delta type
(Homo sapiens (Human)) | BDBM3153
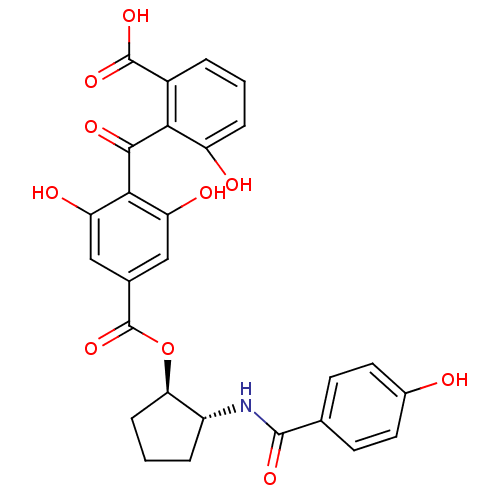
(2-{[2,6-dihydroxy-4-({[(1R,2R)-2-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C27H23NO10/c29-15-9-7-13(8-10-15)25(34)28-17-4-2-6-21(17)38-27(37)14-11-19(31)23(20(32)12-14)24(33)22-16(26(35)36)3-1-5-18(22)30/h1,3,5,7-12,17,21,29-32H,2,4,6H2,(H,28,34)(H,35,36)/t17-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 0.900 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Human Protein kinase C delta |
Bioorg Med Chem Lett 5: 1839-1842 (1995)
Article DOI: 10.1016/0960-894X(95)00303-B
BindingDB Entry DOI: 10.7270/Q2QN66R9 |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM50285233

(4-(2-Carboxy-6-hydroxy-benzoyl)-3,5-dihydroxy-benz...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCOC[C@@H]1NC(=O)c1ccc(O)cc1 Show InChI InChI=1S/C28H25NO11/c30-16-8-6-14(7-9-16)26(35)29-18-13-39-10-2-5-22(18)40-28(38)15-11-20(32)24(21(33)12-15)25(34)23-17(27(36)37)3-1-4-19(23)31/h1,3-4,6-9,11-12,18,22,30-33H,2,5,10,13H2,(H,29,35)(H,36,37)/t18-,22+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human protein kinase C eta |
Bioorg Med Chem Lett 5: 2147-2150 (1995)
Article DOI: 10.1016/0960-894X(95)00364-Y
BindingDB Entry DOI: 10.7270/Q2RF5TZ9 |
More data for this
Ligand-Target Pair | |
Protein kinase C epsilon type
(Homo sapiens (Human)) | BDBM3152
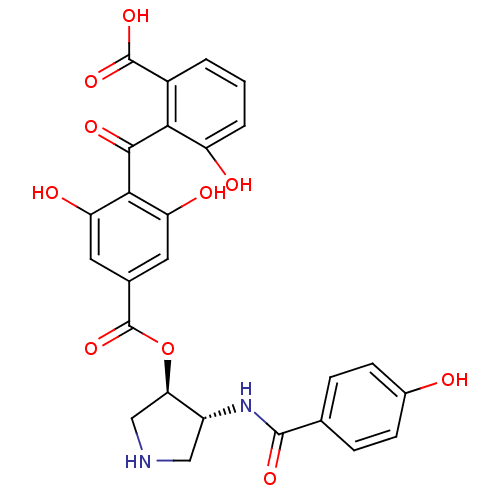
(2-{[2,6-dihydroxy-4-({[(3R,4R)-4-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CNC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C26H22N2O10/c29-14-6-4-12(5-7-14)24(34)28-16-10-27-11-20(16)38-26(37)13-8-18(31)22(19(32)9-13)23(33)21-15(25(35)36)2-1-3-17(21)30/h1-9,16,20,27,29-32H,10-11H2,(H,28,34)(H,35,36)/t16-,20-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
PKC was assayed by quantitating the incorporation of 32P from [gamma-32P]ATP into histone type IIIs. |
J Med Chem 45: 2624-43 (2002)
Article DOI: 10.1021/jm020018f
BindingDB Entry DOI: 10.7270/Q2BG2M50 |
More data for this
Ligand-Target Pair | |
cAMP-dependent protein kinase catalytic subunit alpha
(Bos taurus (bovine)) | BDBM3153

(2-{[2,6-dihydroxy-4-({[(1R,2R)-2-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C27H23NO10/c29-15-9-7-13(8-10-15)25(34)28-17-4-2-6-21(17)38-27(37)14-11-19(31)23(20(32)12-14)24(33)22-16(26(35)36)3-1-5-18(22)30/h1,3,5,7-12,17,21,29-32H,2,4,6H2,(H,28,34)(H,35,36)/t17-,21-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
The activity of PKA, activated by cAMP, is measured by its ability to transfer phosphate from [gamma-32P]ATP to histone. |
J Med Chem 45: 2624-43 (2002)
Article DOI: 10.1021/jm020018f
BindingDB Entry DOI: 10.7270/Q2BG2M50 |
More data for this
Ligand-Target Pair | |
Protein kinase C delta type
(Homo sapiens (Human)) | BDBM3239
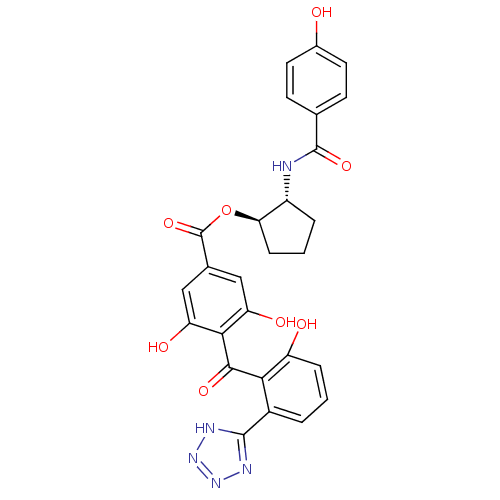
((1R,2R)-2-[(4-hydroxybenzene)amido]cyclopentyl 3,5...)Show SMILES Oc1ccc(cc1)C(=O)N[C@@H]1CCC[C@H]1OC(=O)c1cc(O)c(C(=O)c2c(O)cccc2-c2nnn[nH]2)c(O)c1 |r| Show InChI InChI=1S/C27H23N5O8/c33-15-9-7-13(8-10-15)26(38)28-17-4-2-6-21(17)40-27(39)14-11-19(35)23(20(36)12-14)24(37)22-16(3-1-5-18(22)34)25-29-31-32-30-25/h1,3,5,7-12,17,21,33-36H,2,4,6H2,(H,28,38)(H,29,30,31,32)/t17-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Human Protein kinase C delta |
Bioorg Med Chem Lett 5: 1839-1842 (1995)
Article DOI: 10.1016/0960-894X(95)00303-B
BindingDB Entry DOI: 10.7270/Q2QN66R9 |
More data for this
Ligand-Target Pair | |
Protein kinase C alpha type
(Homo sapiens (Human)) | BDBM3152

(2-{[2,6-dihydroxy-4-({[(3R,4R)-4-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CNC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C26H22N2O10/c29-14-6-4-12(5-7-14)24(34)28-16-10-27-11-20(16)38-26(37)13-8-18(31)22(19(32)9-13)23(33)21-15(25(35)36)2-1-3-17(21)30/h1-9,16,20,27,29-32H,10-11H2,(H,28,34)(H,35,36)/t16-,20-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
PKC was assayed by quantitating the incorporation of 32P from [gamma-32P]ATP into histone type IIIs. |
J Med Chem 45: 2624-43 (2002)
Article DOI: 10.1021/jm020018f
BindingDB Entry DOI: 10.7270/Q2BG2M50 |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM50285232
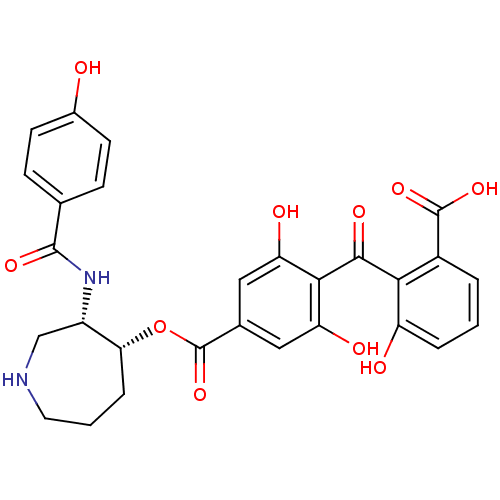
(4-(2-Carboxy-6-hydroxy-benzoyl)-3,5-dihydroxy-benz...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCNC[C@@H]1NC(=O)c1ccc(O)cc1 Show InChI InChI=1S/C28H26N2O10/c31-16-8-6-14(7-9-16)26(36)30-18-13-29-10-2-5-22(18)40-28(39)15-11-20(33)24(21(34)12-15)25(35)23-17(27(37)38)3-1-4-19(23)32/h1,3-4,6-9,11-12,18,22,29,31-34H,2,5,10,13H2,(H,30,36)(H,37,38)/t18-,22+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| Article
| n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human protein kinase C eta |
Bioorg Med Chem Lett 5: 2147-2150 (1995)
Article DOI: 10.1016/0960-894X(95)00364-Y
BindingDB Entry DOI: 10.7270/Q2RF5TZ9 |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM3149
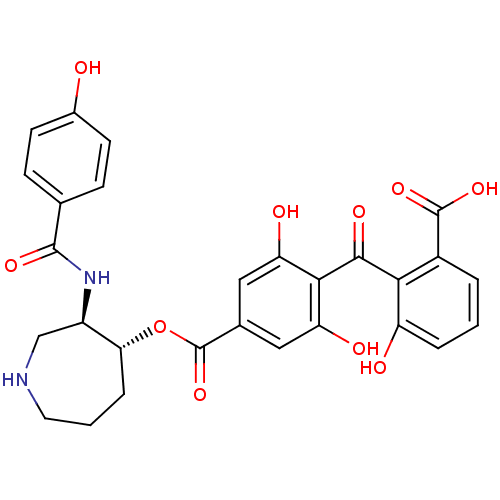
(2-{[2,6-dihydroxy-4-({[(3R,4R)-3-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCNC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C28H26N2O10/c31-16-8-6-14(7-9-16)26(36)30-18-13-29-10-2-5-22(18)40-28(39)15-11-20(33)24(21(34)12-15)25(35)23-17(27(37)38)3-1-4-19(23)32/h1,3-4,6-9,11-12,18,22,29,31-34H,2,5,10,13H2,(H,30,36)(H,37,38)/t18-,22-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
PKC was assayed by quantitating the incorporation of 32P from [gamma-32P]ATP into histone type IIIs. |
J Med Chem 45: 2624-43 (2002)
Article DOI: 10.1021/jm020018f
BindingDB Entry DOI: 10.7270/Q2BG2M50 |
More data for this
Ligand-Target Pair | |
Protein kinase C delta type
(Homo sapiens (Human)) | BDBM3236
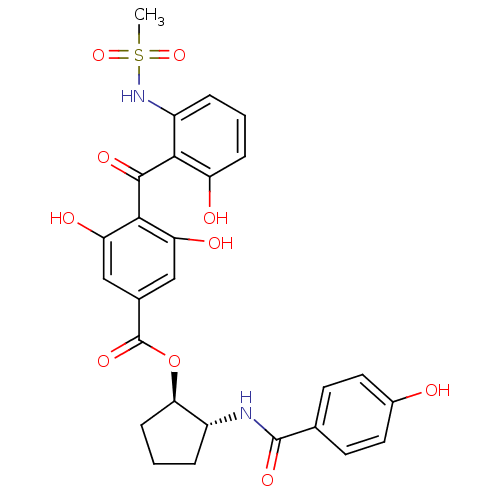
((1R,2R)-2-[(4-hydroxybenzene)amido]cyclopentyl 3,5...)Show SMILES CS(=O)(=O)Nc1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C27H26N2O10S/c1-40(37,38)29-18-5-2-6-19(31)23(18)25(34)24-20(32)12-15(13-21(24)33)27(36)39-22-7-3-4-17(22)28-26(35)14-8-10-16(30)11-9-14/h2,5-6,8-13,17,22,29-33H,3-4,7H2,1H3,(H,28,35)/t17-,22-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Human Protein kinase C delta |
Bioorg Med Chem Lett 5: 1839-1842 (1995)
Article DOI: 10.1016/0960-894X(95)00303-B
BindingDB Entry DOI: 10.7270/Q2QN66R9 |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM50285232

(4-(2-Carboxy-6-hydroxy-benzoyl)-3,5-dihydroxy-benz...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCNC[C@@H]1NC(=O)c1ccc(O)cc1 Show InChI InChI=1S/C28H26N2O10/c31-16-8-6-14(7-9-16)26(36)30-18-13-29-10-2-5-22(18)40-28(39)15-11-20(33)24(21(34)12-15)25(35)23-17(27(37)38)3-1-4-19(23)32/h1,3-4,6-9,11-12,18,22,29,31-34H,2,5,10,13H2,(H,30,36)(H,37,38)/t18-,22+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| Article
| n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human Protein kinase C beta 1 |
Bioorg Med Chem Lett 5: 2147-2150 (1995)
Article DOI: 10.1016/0960-894X(95)00364-Y
BindingDB Entry DOI: 10.7270/Q2RF5TZ9 |
More data for this
Ligand-Target Pair | |
Protein kinase C delta type
(Homo sapiens (Human)) | BDBM50285232

(4-(2-Carboxy-6-hydroxy-benzoyl)-3,5-dihydroxy-benz...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCNC[C@@H]1NC(=O)c1ccc(O)cc1 Show InChI InChI=1S/C28H26N2O10/c31-16-8-6-14(7-9-16)26(36)30-18-13-29-10-2-5-22(18)40-28(39)15-11-20(33)24(21(34)12-15)25(35)23-17(27(37)38)3-1-4-19(23)32/h1,3-4,6-9,11-12,18,22,29,31-34H,2,5,10,13H2,(H,30,36)(H,37,38)/t18-,22+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| Article
| n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human Protein kinase C delta |
Bioorg Med Chem Lett 5: 2147-2150 (1995)
Article DOI: 10.1016/0960-894X(95)00364-Y
BindingDB Entry DOI: 10.7270/Q2RF5TZ9 |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM3152

(2-{[2,6-dihydroxy-4-({[(3R,4R)-4-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CNC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C26H22N2O10/c29-14-6-4-12(5-7-14)24(34)28-16-10-27-11-20(16)38-26(37)13-8-18(31)22(19(32)9-13)23(33)21-15(25(35)36)2-1-3-17(21)30/h1-9,16,20,27,29-32H,10-11H2,(H,28,34)(H,35,36)/t16-,20-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 33 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
PKC was assayed by quantitating the incorporation of 32P from [gamma-32P]ATP into histone type IIIs. |
J Med Chem 45: 2624-43 (2002)
Article DOI: 10.1021/jm020018f
BindingDB Entry DOI: 10.7270/Q2BG2M50 |
More data for this
Ligand-Target Pair | |
Protein kinase C gamma type
(Homo sapiens (Human)) | BDBM50285232

(4-(2-Carboxy-6-hydroxy-benzoyl)-3,5-dihydroxy-benz...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCNC[C@@H]1NC(=O)c1ccc(O)cc1 Show InChI InChI=1S/C28H26N2O10/c31-16-8-6-14(7-9-16)26(36)30-18-13-29-10-2-5-22(18)40-28(39)15-11-20(33)24(21(34)12-15)25(35)23-17(27(37)38)3-1-4-19(23)32/h1,3-4,6-9,11-12,18,22,29,31-34H,2,5,10,13H2,(H,30,36)(H,37,38)/t18-,22+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| Article
| n/a | n/a | 34 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human Protein kinase C gamma |
Bioorg Med Chem Lett 5: 2147-2150 (1995)
Article DOI: 10.1016/0960-894X(95)00364-Y
BindingDB Entry DOI: 10.7270/Q2RF5TZ9 |
More data for this
Ligand-Target Pair | |
Protein kinase C epsilon type
(Homo sapiens (Human)) | BDBM3149

(2-{[2,6-dihydroxy-4-({[(3R,4R)-3-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCNC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C28H26N2O10/c31-16-8-6-14(7-9-16)26(36)30-18-13-29-10-2-5-22(18)40-28(39)15-11-20(33)24(21(34)12-15)25(35)23-17(27(37)38)3-1-4-19(23)32/h1,3-4,6-9,11-12,18,22,29,31-34H,2,5,10,13H2,(H,30,36)(H,37,38)/t18-,22-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 38 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
PKC was assayed by quantitating the incorporation of 32P from [gamma-32P]ATP into histone type IIIs. |
J Med Chem 45: 2624-43 (2002)
Article DOI: 10.1021/jm020018f
BindingDB Entry DOI: 10.7270/Q2BG2M50 |
More data for this
Ligand-Target Pair | |
Protein kinase C alpha type
(Homo sapiens (Human)) | BDBM3153

(2-{[2,6-dihydroxy-4-({[(1R,2R)-2-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C27H23NO10/c29-15-9-7-13(8-10-15)25(34)28-17-4-2-6-21(17)38-27(37)14-11-19(31)23(20(32)12-14)24(33)22-16(26(35)36)3-1-5-18(22)30/h1,3,5,7-12,17,21,29-32H,2,4,6H2,(H,28,34)(H,35,36)/t17-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
PKC was assayed by quantitating the incorporation of 32P from [gamma-32P]ATP into histone type IIIs. |
J Med Chem 45: 2624-43 (2002)
Article DOI: 10.1021/jm020018f
BindingDB Entry DOI: 10.7270/Q2BG2M50 |
More data for this
Ligand-Target Pair | |
Protein kinase C alpha type
(Homo sapiens (Human)) | BDBM3153

(2-{[2,6-dihydroxy-4-({[(1R,2R)-2-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C27H23NO10/c29-15-9-7-13(8-10-15)25(34)28-17-4-2-6-21(17)38-27(37)14-11-19(31)23(20(32)12-14)24(33)22-16(26(35)36)3-1-5-18(22)30/h1,3,5,7-12,17,21,29-32H,2,4,6H2,(H,28,34)(H,35,36)/t17-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Human Protein kinase C alpha |
Bioorg Med Chem Lett 5: 1839-1842 (1995)
Article DOI: 10.1016/0960-894X(95)00303-B
BindingDB Entry DOI: 10.7270/Q2QN66R9 |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM50285232

(4-(2-Carboxy-6-hydroxy-benzoyl)-3,5-dihydroxy-benz...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCNC[C@@H]1NC(=O)c1ccc(O)cc1 Show InChI InChI=1S/C28H26N2O10/c31-16-8-6-14(7-9-16)26(36)30-18-13-29-10-2-5-22(18)40-28(39)15-11-20(33)24(21(34)12-15)25(35)23-17(27(37)38)3-1-4-19(23)32/h1,3-4,6-9,11-12,18,22,29,31-34H,2,5,10,13H2,(H,30,36)(H,37,38)/t18-,22+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| Article
| n/a | n/a | 44 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human Protein kinase C beta 2 |
Bioorg Med Chem Lett 5: 2147-2150 (1995)
Article DOI: 10.1016/0960-894X(95)00364-Y
BindingDB Entry DOI: 10.7270/Q2RF5TZ9 |
More data for this
Ligand-Target Pair | |
Protein kinase C epsilon type
(Homo sapiens (Human)) | BDBM50285232

(4-(2-Carboxy-6-hydroxy-benzoyl)-3,5-dihydroxy-benz...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCNC[C@@H]1NC(=O)c1ccc(O)cc1 Show InChI InChI=1S/C28H26N2O10/c31-16-8-6-14(7-9-16)26(36)30-18-13-29-10-2-5-22(18)40-28(39)15-11-20(33)24(21(34)12-15)25(35)23-17(27(37)38)3-1-4-19(23)32/h1,3-4,6-9,11-12,18,22,29,31-34H,2,5,10,13H2,(H,30,36)(H,37,38)/t18-,22+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| Article
| n/a | n/a | 49 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human Protein kinase C epsilon |
Bioorg Med Chem Lett 5: 2147-2150 (1995)
Article DOI: 10.1016/0960-894X(95)00364-Y
BindingDB Entry DOI: 10.7270/Q2RF5TZ9 |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM3153

(2-{[2,6-dihydroxy-4-({[(1R,2R)-2-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C27H23NO10/c29-15-9-7-13(8-10-15)25(34)28-17-4-2-6-21(17)38-27(37)14-11-19(31)23(20(32)12-14)24(33)22-16(26(35)36)3-1-5-18(22)30/h1,3,5,7-12,17,21,29-32H,2,4,6H2,(H,28,34)(H,35,36)/t17-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Human Protein kinase C beta 2 |
Bioorg Med Chem Lett 5: 1839-1842 (1995)
Article DOI: 10.1016/0960-894X(95)00303-B
BindingDB Entry DOI: 10.7270/Q2QN66R9 |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM3153

(2-{[2,6-dihydroxy-4-({[(1R,2R)-2-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C27H23NO10/c29-15-9-7-13(8-10-15)25(34)28-17-4-2-6-21(17)38-27(37)14-11-19(31)23(20(32)12-14)24(33)22-16(26(35)36)3-1-5-18(22)30/h1,3,5,7-12,17,21,29-32H,2,4,6H2,(H,28,34)(H,35,36)/t17-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
PKC was assayed by quantitating the incorporation of 32P from [gamma-32P]ATP into histone type IIIs. |
J Med Chem 45: 2624-43 (2002)
Article DOI: 10.1021/jm020018f
BindingDB Entry DOI: 10.7270/Q2BG2M50 |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM3235

((1R,2R)-2-[(4-hydroxybenzene)amido]cyclopentyl 3,5...)Show SMILES Oc1ccc(cc1)C(=O)N[C@@H]1CCC[C@H]1OC(=O)c1cc(O)c(C(=O)c2c(O)cccc2NS(=O)(=O)C(F)(F)F)c(O)c1 |r| Show InChI InChI=1S/C27H23F3N2O10S/c28-27(29,30)43(40,41)32-17-4-1-5-18(34)22(17)24(37)23-19(35)11-14(12-20(23)36)26(39)42-21-6-2-3-16(21)31-25(38)13-7-9-15(33)10-8-13/h1,4-5,7-12,16,21,32-36H,2-3,6H2,(H,31,38)/t16-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
PKC was assayed by quantitating the incorporation of 32P from [gamma-32P]ATP into histone type IIIs. |
J Med Chem 45: 2624-43 (2002)
Article DOI: 10.1021/jm020018f
BindingDB Entry DOI: 10.7270/Q2BG2M50 |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM3235

((1R,2R)-2-[(4-hydroxybenzene)amido]cyclopentyl 3,5...)Show SMILES Oc1ccc(cc1)C(=O)N[C@@H]1CCC[C@H]1OC(=O)c1cc(O)c(C(=O)c2c(O)cccc2NS(=O)(=O)C(F)(F)F)c(O)c1 |r| Show InChI InChI=1S/C27H23F3N2O10S/c28-27(29,30)43(40,41)32-17-4-1-5-18(34)22(17)24(37)23-19(35)11-14(12-20(23)36)26(39)42-21-6-2-3-16(21)31-25(38)13-7-9-15(33)10-8-13/h1,4-5,7-12,16,21,32-36H,2-3,6H2,(H,31,38)/t16-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Human Protein kinase C beta 2 |
Bioorg Med Chem Lett 5: 1839-1842 (1995)
Article DOI: 10.1016/0960-894X(95)00303-B
BindingDB Entry DOI: 10.7270/Q2QN66R9 |
More data for this
Ligand-Target Pair | |
cAMP-dependent protein kinase catalytic subunit alpha
(Bos taurus (bovine)) | BDBM3230

((3R,4R)-4-[(4-hydroxybenzene)amido]pyrrolidin-3-yl...)Show SMILES Oc1ccc(cc1)C(=O)N[C@@H]1CNC[C@H]1OC(=O)c1cc(O)c(C(=O)c2c(O)ccc3CCCCc23)c(O)c1 |r| Show InChI InChI=1S/C29H28N2O8/c32-18-8-5-16(6-9-18)28(37)31-20-13-30-14-24(20)39-29(38)17-11-22(34)26(23(35)12-17)27(36)25-19-4-2-1-3-15(19)7-10-21(25)33/h5-12,20,24,30,32-35H,1-4,13-14H2,(H,31,37)/t20-,24-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
The activity of PKA, activated by cAMP, is measured by its ability to transfer phosphate from [gamma-32P]ATP to histone. |
J Med Chem 45: 2624-43 (2002)
Article DOI: 10.1021/jm020018f
BindingDB Entry DOI: 10.7270/Q2BG2M50 |
More data for this
Ligand-Target Pair | |
Protein kinase C epsilon type
(Homo sapiens (Human)) | BDBM3153

(2-{[2,6-dihydroxy-4-({[(1R,2R)-2-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C27H23NO10/c29-15-9-7-13(8-10-15)25(34)28-17-4-2-6-21(17)38-27(37)14-11-19(31)23(20(32)12-14)24(33)22-16(26(35)36)3-1-5-18(22)30/h1,3,5,7-12,17,21,29-32H,2,4,6H2,(H,28,34)(H,35,36)/t17-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
PKC was assayed by quantitating the incorporation of 32P from [gamma-32P]ATP into histone type IIIs. |
J Med Chem 45: 2624-43 (2002)
Article DOI: 10.1021/jm020018f
BindingDB Entry DOI: 10.7270/Q2BG2M50 |
More data for this
Ligand-Target Pair | |
Protein kinase C delta type
(Homo sapiens (Human)) | BDBM3235

((1R,2R)-2-[(4-hydroxybenzene)amido]cyclopentyl 3,5...)Show SMILES Oc1ccc(cc1)C(=O)N[C@@H]1CCC[C@H]1OC(=O)c1cc(O)c(C(=O)c2c(O)cccc2NS(=O)(=O)C(F)(F)F)c(O)c1 |r| Show InChI InChI=1S/C27H23F3N2O10S/c28-27(29,30)43(40,41)32-17-4-1-5-18(34)22(17)24(37)23-19(35)11-14(12-20(23)36)26(39)42-21-6-2-3-16(21)31-25(38)13-7-9-15(33)10-8-13/h1,4-5,7-12,16,21,32-36H,2-3,6H2,(H,31,38)/t16-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | <50 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Human Protein kinase C delta |
Bioorg Med Chem Lett 5: 1839-1842 (1995)
Article DOI: 10.1016/0960-894X(95)00303-B
BindingDB Entry DOI: 10.7270/Q2QN66R9 |
More data for this
Ligand-Target Pair | |
Protein kinase C epsilon type
(Homo sapiens (Human)) | BDBM3153

(2-{[2,6-dihydroxy-4-({[(1R,2R)-2-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C27H23NO10/c29-15-9-7-13(8-10-15)25(34)28-17-4-2-6-21(17)38-27(37)14-11-19(31)23(20(32)12-14)24(33)22-16(26(35)36)3-1-5-18(22)30/h1,3,5,7-12,17,21,29-32H,2,4,6H2,(H,28,34)(H,35,36)/t17-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Human Protein kinase C epsilon |
Bioorg Med Chem Lett 5: 1839-1842 (1995)
Article DOI: 10.1016/0960-894X(95)00303-B
BindingDB Entry DOI: 10.7270/Q2QN66R9 |
More data for this
Ligand-Target Pair | |
Protein kinase C gamma type
(Homo sapiens (Human)) | BDBM50285236
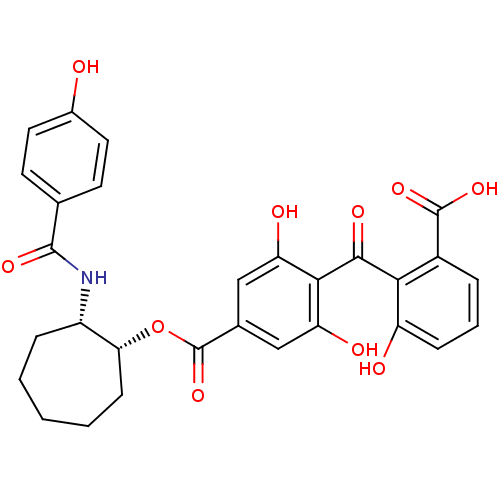
(4-(2-Carboxy-6-hydroxy-benzoyl)-3,5-dihydroxy-benz...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCCC[C@@H]1NC(=O)c1ccc(O)cc1 Show InChI InChI=1S/C29H27NO10/c31-17-11-9-15(10-12-17)27(36)30-19-6-2-1-3-8-23(19)40-29(39)16-13-21(33)25(22(34)14-16)26(35)24-18(28(37)38)5-4-7-20(24)32/h4-5,7,9-14,19,23,31-34H,1-3,6,8H2,(H,30,36)(H,37,38)/t19-,23+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 60 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human Protein kinase C gamma |
Bioorg Med Chem Lett 5: 2147-2150 (1995)
Article DOI: 10.1016/0960-894X(95)00364-Y
BindingDB Entry DOI: 10.7270/Q2RF5TZ9 |
More data for this
Ligand-Target Pair | |
cAMP-dependent protein kinase catalytic subunit alpha
(Bos taurus (bovine)) | BDBM3152

(2-{[2,6-dihydroxy-4-({[(3R,4R)-4-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CNC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C26H22N2O10/c29-14-6-4-12(5-7-14)24(34)28-16-10-27-11-20(16)38-26(37)13-8-18(31)22(19(32)9-13)23(33)21-15(25(35)36)2-1-3-17(21)30/h1-9,16,20,27,29-32H,10-11H2,(H,28,34)(H,35,36)/t16-,20-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 60 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
The activity of PKA, activated by cAMP, is measured by its ability to transfer phosphate from [gamma-32P]ATP to histone. |
J Med Chem 45: 2624-43 (2002)
Article DOI: 10.1021/jm020018f
BindingDB Entry DOI: 10.7270/Q2BG2M50 |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM50285236

(4-(2-Carboxy-6-hydroxy-benzoyl)-3,5-dihydroxy-benz...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCCC[C@@H]1NC(=O)c1ccc(O)cc1 Show InChI InChI=1S/C29H27NO10/c31-17-11-9-15(10-12-17)27(36)30-19-6-2-1-3-8-23(19)40-29(39)16-13-21(33)25(22(34)14-16)26(35)24-18(28(37)38)5-4-7-20(24)32/h4-5,7,9-14,19,23,31-34H,1-3,6,8H2,(H,30,36)(H,37,38)/t19-,23+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 60 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human protein kinase C eta |
Bioorg Med Chem Lett 5: 2147-2150 (1995)
Article DOI: 10.1016/0960-894X(95)00364-Y
BindingDB Entry DOI: 10.7270/Q2RF5TZ9 |
More data for this
Ligand-Target Pair | |
Protein kinase C alpha type
(Homo sapiens (Human)) | BDBM3149

(2-{[2,6-dihydroxy-4-({[(3R,4R)-3-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCNC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C28H26N2O10/c31-16-8-6-14(7-9-16)26(36)30-18-13-29-10-2-5-22(18)40-28(39)15-11-20(33)24(21(34)12-15)25(35)23-17(27(37)38)3-1-4-19(23)32/h1,3-4,6-9,11-12,18,22,29,31-34H,2,5,10,13H2,(H,30,36)(H,37,38)/t18-,22-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 67 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
PKC was assayed by quantitating the incorporation of 32P from [gamma-32P]ATP into histone type IIIs. |
J Med Chem 45: 2624-43 (2002)
Article DOI: 10.1021/jm020018f
BindingDB Entry DOI: 10.7270/Q2BG2M50 |
More data for this
Ligand-Target Pair | |
Protein kinase C alpha type
(Homo sapiens (Human)) | BDBM50285232

(4-(2-Carboxy-6-hydroxy-benzoyl)-3,5-dihydroxy-benz...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCNC[C@@H]1NC(=O)c1ccc(O)cc1 Show InChI InChI=1S/C28H26N2O10/c31-16-8-6-14(7-9-16)26(36)30-18-13-29-10-2-5-22(18)40-28(39)15-11-20(33)24(21(34)12-15)25(35)23-17(27(37)38)3-1-4-19(23)32/h1,3-4,6-9,11-12,18,22,29,31-34H,2,5,10,13H2,(H,30,36)(H,37,38)/t18-,22+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| Article
| n/a | n/a | 74 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human Protein kinase C alpha |
Bioorg Med Chem Lett 5: 2147-2150 (1995)
Article DOI: 10.1016/0960-894X(95)00364-Y
BindingDB Entry DOI: 10.7270/Q2RF5TZ9 |
More data for this
Ligand-Target Pair | |
Protein kinase C delta type
(Homo sapiens (Human)) | BDBM50285233

(4-(2-Carboxy-6-hydroxy-benzoyl)-3,5-dihydroxy-benz...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCOC[C@@H]1NC(=O)c1ccc(O)cc1 Show InChI InChI=1S/C28H25NO11/c30-16-8-6-14(7-9-16)26(35)29-18-13-39-10-2-5-22(18)40-28(38)15-11-20(32)24(21(33)12-15)25(34)23-17(27(36)37)3-1-4-19(23)31/h1,3-4,6-9,11-12,18,22,30-33H,2,5,10,13H2,(H,29,35)(H,36,37)/t18-,22+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human Protein kinase C delta |
Bioorg Med Chem Lett 5: 2147-2150 (1995)
Article DOI: 10.1016/0960-894X(95)00364-Y
BindingDB Entry DOI: 10.7270/Q2RF5TZ9 |
More data for this
Ligand-Target Pair | |
Protein kinase C delta type
(Homo sapiens (Human)) | BDBM50285236

(4-(2-Carboxy-6-hydroxy-benzoyl)-3,5-dihydroxy-benz...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCCC[C@@H]1NC(=O)c1ccc(O)cc1 Show InChI InChI=1S/C29H27NO10/c31-17-11-9-15(10-12-17)27(36)30-19-6-2-1-3-8-23(19)40-29(39)16-13-21(33)25(22(34)14-16)26(35)24-18(28(37)38)5-4-7-20(24)32/h4-5,7,9-14,19,23,31-34H,1-3,6,8H2,(H,30,36)(H,37,38)/t19-,23+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human Protein kinase C delta |
Bioorg Med Chem Lett 5: 2147-2150 (1995)
Article DOI: 10.1016/0960-894X(95)00364-Y
BindingDB Entry DOI: 10.7270/Q2RF5TZ9 |
More data for this
Ligand-Target Pair | |
Protein kinase C delta type
(Homo sapiens (Human)) | BDBM3240
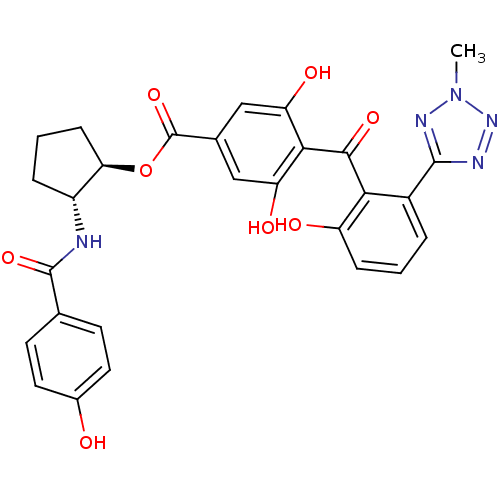
((1R,2R)-2-[(4-hydroxybenzene)amido]cyclopentyl 3,5...)Show SMILES Cn1nnc(n1)-c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C28H25N5O8/c1-33-31-26(30-32-33)17-4-2-6-19(35)23(17)25(38)24-20(36)12-15(13-21(24)37)28(40)41-22-7-3-5-18(22)29-27(39)14-8-10-16(34)11-9-14/h2,4,6,8-13,18,22,34-37H,3,5,7H2,1H3,(H,29,39)/t18-,22-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Human Protein kinase C delta |
Bioorg Med Chem Lett 5: 1839-1842 (1995)
Article DOI: 10.1016/0960-894X(95)00303-B
BindingDB Entry DOI: 10.7270/Q2QN66R9 |
More data for this
Ligand-Target Pair | |
Protein kinase C delta type
(Homo sapiens (Human)) | BDBM50285234
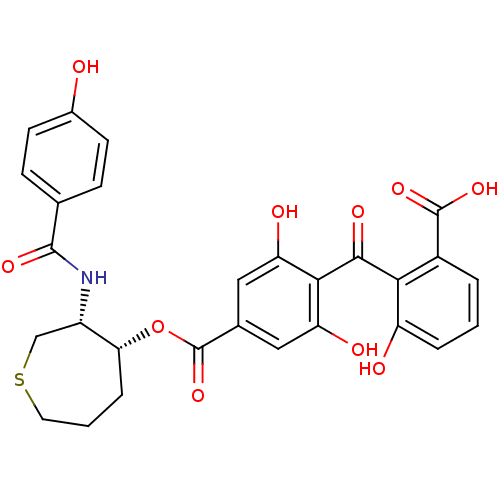
(4-(2-Carboxy-6-hydroxy-benzoyl)-3,5-dihydroxy-benz...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCSC[C@@H]1NC(=O)c1ccc(O)cc1 Show InChI InChI=1S/C28H25NO10S/c30-16-8-6-14(7-9-16)26(35)29-18-13-40-10-2-5-22(18)39-28(38)15-11-20(32)24(21(33)12-15)25(34)23-17(27(36)37)3-1-4-19(23)31/h1,3-4,6-9,11-12,18,22,30-33H,2,5,10,13H2,(H,29,35)(H,36,37)/t18-,22+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human Protein kinase C delta |
Bioorg Med Chem Lett 5: 2147-2150 (1995)
Article DOI: 10.1016/0960-894X(95)00364-Y
BindingDB Entry DOI: 10.7270/Q2RF5TZ9 |
More data for this
Ligand-Target Pair | |
Protein kinase C alpha type
(Homo sapiens (Human)) | BDBM3235

((1R,2R)-2-[(4-hydroxybenzene)amido]cyclopentyl 3,5...)Show SMILES Oc1ccc(cc1)C(=O)N[C@@H]1CCC[C@H]1OC(=O)c1cc(O)c(C(=O)c2c(O)cccc2NS(=O)(=O)C(F)(F)F)c(O)c1 |r| Show InChI InChI=1S/C27H23F3N2O10S/c28-27(29,30)43(40,41)32-17-4-1-5-18(34)22(17)24(37)23-19(35)11-14(12-20(23)36)26(39)42-21-6-2-3-16(21)31-25(38)13-7-9-15(33)10-8-13/h1,4-5,7-12,16,21,32-36H,2-3,6H2,(H,31,38)/t16-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
PKC was assayed by quantitating the incorporation of 32P from [gamma-32P]ATP into histone type IIIs. |
J Med Chem 45: 2624-43 (2002)
Article DOI: 10.1021/jm020018f
BindingDB Entry DOI: 10.7270/Q2BG2M50 |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM50285234

(4-(2-Carboxy-6-hydroxy-benzoyl)-3,5-dihydroxy-benz...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCSC[C@@H]1NC(=O)c1ccc(O)cc1 Show InChI InChI=1S/C28H25NO10S/c30-16-8-6-14(7-9-16)26(35)29-18-13-40-10-2-5-22(18)39-28(38)15-11-20(32)24(21(33)12-15)25(34)23-17(27(36)37)3-1-4-19(23)31/h1,3-4,6-9,11-12,18,22,30-33H,2,5,10,13H2,(H,29,35)(H,36,37)/t18-,22+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human protein kinase C eta |
Bioorg Med Chem Lett 5: 2147-2150 (1995)
Article DOI: 10.1016/0960-894X(95)00364-Y
BindingDB Entry DOI: 10.7270/Q2RF5TZ9 |
More data for this
Ligand-Target Pair | |
Protein kinase C alpha type
(Homo sapiens (Human)) | BDBM3235

((1R,2R)-2-[(4-hydroxybenzene)amido]cyclopentyl 3,5...)Show SMILES Oc1ccc(cc1)C(=O)N[C@@H]1CCC[C@H]1OC(=O)c1cc(O)c(C(=O)c2c(O)cccc2NS(=O)(=O)C(F)(F)F)c(O)c1 |r| Show InChI InChI=1S/C27H23F3N2O10S/c28-27(29,30)43(40,41)32-17-4-1-5-18(34)22(17)24(37)23-19(35)11-14(12-20(23)36)26(39)42-21-6-2-3-16(21)31-25(38)13-7-9-15(33)10-8-13/h1,4-5,7-12,16,21,32-36H,2-3,6H2,(H,31,38)/t16-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Human Protein kinase C alpha |
Bioorg Med Chem Lett 5: 1839-1842 (1995)
Article DOI: 10.1016/0960-894X(95)00303-B
BindingDB Entry DOI: 10.7270/Q2QN66R9 |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM3236

((1R,2R)-2-[(4-hydroxybenzene)amido]cyclopentyl 3,5...)Show SMILES CS(=O)(=O)Nc1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C27H26N2O10S/c1-40(37,38)29-18-5-2-6-19(31)23(18)25(34)24-20(32)12-15(13-21(24)33)27(36)39-22-7-3-4-17(22)28-26(35)14-8-10-16(30)11-9-14/h2,5-6,8-13,17,22,29-33H,3-4,7H2,1H3,(H,28,35)/t17-,22-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
PKC was assayed by quantitating the incorporation of 32P from [gamma-32P]ATP into histone type IIIs. |
J Med Chem 45: 2624-43 (2002)
Article DOI: 10.1021/jm020018f
BindingDB Entry DOI: 10.7270/Q2BG2M50 |
More data for this
Ligand-Target Pair | |
Protein kinase C delta type
(Homo sapiens (Human)) | BDBM3241

((1R,2R)-2-[(4-hydroxybenzene)amido]cyclopentyl 3,5...)Show SMILES Cn1nnnc1-c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C28H25N5O8/c1-33-26(30-31-32-33)17-4-2-6-19(35)23(17)25(38)24-20(36)12-15(13-21(24)37)28(40)41-22-7-3-5-18(22)29-27(39)14-8-10-16(34)11-9-14/h2,4,6,8-13,18,22,34-37H,3,5,7H2,1H3,(H,29,39)/t18-,22-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 130 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Human Protein kinase C delta |
Bioorg Med Chem Lett 5: 1839-1842 (1995)
Article DOI: 10.1016/0960-894X(95)00303-B
BindingDB Entry DOI: 10.7270/Q2QN66R9 |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM3236

((1R,2R)-2-[(4-hydroxybenzene)amido]cyclopentyl 3,5...)Show SMILES CS(=O)(=O)Nc1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C27H26N2O10S/c1-40(37,38)29-18-5-2-6-19(31)23(18)25(34)24-20(32)12-15(13-21(24)33)27(36)39-22-7-3-4-17(22)28-26(35)14-8-10-16(30)11-9-14/h2,5-6,8-13,17,22,29-33H,3-4,7H2,1H3,(H,28,35)/t17-,22-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 150 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Human Protein kinase C beta 2 |
Bioorg Med Chem Lett 5: 1839-1842 (1995)
Article DOI: 10.1016/0960-894X(95)00303-B
BindingDB Entry DOI: 10.7270/Q2QN66R9 |
More data for this
Ligand-Target Pair | |
cAMP-dependent protein kinase catalytic subunit alpha
(Bos taurus (bovine)) | BDBM3239

((1R,2R)-2-[(4-hydroxybenzene)amido]cyclopentyl 3,5...)Show SMILES Oc1ccc(cc1)C(=O)N[C@@H]1CCC[C@H]1OC(=O)c1cc(O)c(C(=O)c2c(O)cccc2-c2nnn[nH]2)c(O)c1 |r| Show InChI InChI=1S/C27H23N5O8/c33-15-9-7-13(8-10-15)26(38)28-17-4-2-6-21(17)40-27(39)14-11-19(35)23(20(36)12-14)24(37)22-16(3-1-5-18(22)34)25-29-31-32-30-25/h1,3,5,7-12,17,21,33-36H,2,4,6H2,(H,28,38)(H,29,30,31,32)/t17-,21-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 210 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
The activity of PKA, activated by cAMP, is measured by its ability to transfer phosphate from [gamma-32P]ATP to histone. |
J Med Chem 45: 2624-43 (2002)
Article DOI: 10.1021/jm020018f
BindingDB Entry DOI: 10.7270/Q2BG2M50 |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM50285236

(4-(2-Carboxy-6-hydroxy-benzoyl)-3,5-dihydroxy-benz...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCCC[C@@H]1NC(=O)c1ccc(O)cc1 Show InChI InChI=1S/C29H27NO10/c31-17-11-9-15(10-12-17)27(36)30-19-6-2-1-3-8-23(19)40-29(39)16-13-21(33)25(22(34)14-16)26(35)24-18(28(37)38)5-4-7-20(24)32/h4-5,7,9-14,19,23,31-34H,1-3,6,8H2,(H,30,36)(H,37,38)/t19-,23+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 220 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human Protein kinase C beta 1 |
Bioorg Med Chem Lett 5: 2147-2150 (1995)
Article DOI: 10.1016/0960-894X(95)00364-Y
BindingDB Entry DOI: 10.7270/Q2RF5TZ9 |
More data for this
Ligand-Target Pair | |
cAMP-dependent protein kinase catalytic subunit alpha
(Bos taurus (bovine)) | BDBM3231

((3R,4R)-3-[(4-hydroxybenzene)amido]azepan-4-yl 3,5...)Show SMILES COc1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCNC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C28H28N2O9/c1-38-23-5-2-4-19(32)25(23)26(35)24-20(33)12-16(13-21(24)34)28(37)39-22-6-3-11-29-14-18(22)30-27(36)15-7-9-17(31)10-8-15/h2,4-5,7-10,12-13,18,22,29,31-34H,3,6,11,14H2,1H3,(H,30,36)/t18-,22-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 250 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
The activity of PKA, activated by cAMP, is measured by its ability to transfer phosphate from [gamma-32P]ATP to histone. |
J Med Chem 45: 2624-43 (2002)
Article DOI: 10.1021/jm020018f
BindingDB Entry DOI: 10.7270/Q2BG2M50 |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM3239

((1R,2R)-2-[(4-hydroxybenzene)amido]cyclopentyl 3,5...)Show SMILES Oc1ccc(cc1)C(=O)N[C@@H]1CCC[C@H]1OC(=O)c1cc(O)c(C(=O)c2c(O)cccc2-c2nnn[nH]2)c(O)c1 |r| Show InChI InChI=1S/C27H23N5O8/c33-15-9-7-13(8-10-15)26(38)28-17-4-2-6-21(17)40-27(39)14-11-19(35)23(20(36)12-14)24(37)22-16(3-1-5-18(22)34)25-29-31-32-30-25/h1,3,5,7-12,17,21,33-36H,2,4,6H2,(H,28,38)(H,29,30,31,32)/t17-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 260 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
PKC was assayed by quantitating the incorporation of 32P from [gamma-32P]ATP into histone type IIIs. |
J Med Chem 45: 2624-43 (2002)
Article DOI: 10.1021/jm020018f
BindingDB Entry DOI: 10.7270/Q2BG2M50 |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM3230

((3R,4R)-4-[(4-hydroxybenzene)amido]pyrrolidin-3-yl...)Show SMILES Oc1ccc(cc1)C(=O)N[C@@H]1CNC[C@H]1OC(=O)c1cc(O)c(C(=O)c2c(O)ccc3CCCCc23)c(O)c1 |r| Show InChI InChI=1S/C29H28N2O8/c32-18-8-5-16(6-9-18)28(37)31-20-13-30-14-24(20)39-29(38)17-11-22(34)26(23(35)12-17)27(36)25-19-4-2-1-3-15(19)7-10-21(25)33/h5-12,20,24,30,32-35H,1-4,13-14H2,(H,31,37)/t20-,24-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 260 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
PKC was assayed by quantitating the incorporation of 32P from [gamma-32P]ATP into histone type IIIs. |
J Med Chem 45: 2624-43 (2002)
Article DOI: 10.1021/jm020018f
BindingDB Entry DOI: 10.7270/Q2BG2M50 |
More data for this
Ligand-Target Pair | |
Protein kinase C alpha type
(Homo sapiens (Human)) | BDBM50285236

(4-(2-Carboxy-6-hydroxy-benzoyl)-3,5-dihydroxy-benz...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCCC[C@@H]1NC(=O)c1ccc(O)cc1 Show InChI InChI=1S/C29H27NO10/c31-17-11-9-15(10-12-17)27(36)30-19-6-2-1-3-8-23(19)40-29(39)16-13-21(33)25(22(34)14-16)26(35)24-18(28(37)38)5-4-7-20(24)32/h4-5,7,9-14,19,23,31-34H,1-3,6,8H2,(H,30,36)(H,37,38)/t19-,23+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 270 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human Protein kinase C alpha |
Bioorg Med Chem Lett 5: 2147-2150 (1995)
Article DOI: 10.1016/0960-894X(95)00364-Y
BindingDB Entry DOI: 10.7270/Q2RF5TZ9 |
More data for this
Ligand-Target Pair | |
Protein kinase C epsilon type
(Homo sapiens (Human)) | BDBM50285236

(4-(2-Carboxy-6-hydroxy-benzoyl)-3,5-dihydroxy-benz...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCCC[C@@H]1NC(=O)c1ccc(O)cc1 Show InChI InChI=1S/C29H27NO10/c31-17-11-9-15(10-12-17)27(36)30-19-6-2-1-3-8-23(19)40-29(39)16-13-21(33)25(22(34)14-16)26(35)24-18(28(37)38)5-4-7-20(24)32/h4-5,7,9-14,19,23,31-34H,1-3,6,8H2,(H,30,36)(H,37,38)/t19-,23+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 280 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human Protein kinase C epsilon |
Bioorg Med Chem Lett 5: 2147-2150 (1995)
Article DOI: 10.1016/0960-894X(95)00364-Y
BindingDB Entry DOI: 10.7270/Q2RF5TZ9 |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM3239

((1R,2R)-2-[(4-hydroxybenzene)amido]cyclopentyl 3,5...)Show SMILES Oc1ccc(cc1)C(=O)N[C@@H]1CCC[C@H]1OC(=O)c1cc(O)c(C(=O)c2c(O)cccc2-c2nnn[nH]2)c(O)c1 |r| Show InChI InChI=1S/C27H23N5O8/c33-15-9-7-13(8-10-15)26(38)28-17-4-2-6-21(17)40-27(39)14-11-19(35)23(20(36)12-14)24(37)22-16(3-1-5-18(22)34)25-29-31-32-30-25/h1,3,5,7-12,17,21,33-36H,2,4,6H2,(H,28,38)(H,29,30,31,32)/t17-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 290 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Human Protein kinase C beta 2 |
Bioorg Med Chem Lett 5: 1839-1842 (1995)
Article DOI: 10.1016/0960-894X(95)00303-B
BindingDB Entry DOI: 10.7270/Q2QN66R9 |
More data for this
Ligand-Target Pair | |
cAMP-dependent protein kinase catalytic subunit alpha
(Bos taurus (bovine)) | BDBM3228

((3R,4R)-4-[(4-hydroxybenzene)amido]pyrrolidin-3-yl...)Show SMILES Oc1ccc(cc1)C(=O)N[C@@H]1CNC[C@H]1OC(=O)c1cc(O)c(C(=O)c2c(O)ccc3ccccc23)c(O)c1 |r| Show InChI InChI=1S/C29H24N2O8/c32-18-8-5-16(6-9-18)28(37)31-20-13-30-14-24(20)39-29(38)17-11-22(34)26(23(35)12-17)27(36)25-19-4-2-1-3-15(19)7-10-21(25)33/h1-12,20,24,30,32-35H,13-14H2,(H,31,37)/t20-,24-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 290 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
The activity of PKA, activated by cAMP, is measured by its ability to transfer phosphate from [gamma-32P]ATP to histone. |
J Med Chem 45: 2624-43 (2002)
Article DOI: 10.1021/jm020018f
BindingDB Entry DOI: 10.7270/Q2BG2M50 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data