 Found 84 hits with Last Name = 'standeven' and Initial = 'am'
Found 84 hits with Last Name = 'standeven' and Initial = 'am' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50075879
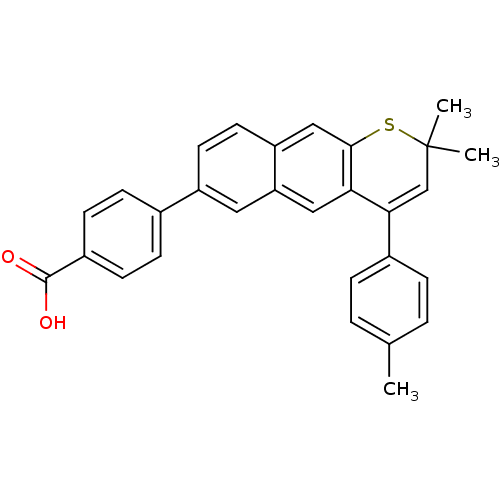
(4-(2,2-Dimethyl-4-p-tolyl-2H-1-thia-anthracen-6-yl...)Show SMILES Cc1ccc(cc1)C1=CC(C)(C)Sc2cc3ccc(cc3cc12)-c1ccc(cc1)C(O)=O |t:8| Show InChI InChI=1S/C29H24O2S/c1-18-4-6-20(7-5-18)26-17-29(2,3)32-27-16-23-13-12-22(14-24(23)15-25(26)27)19-8-10-21(11-9-19)28(30)31/h4-17H,1-3H3,(H,30,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Pharmaceuticals
Curated by ChEMBL
| Assay Description
Antagonistic activity against RAR gamma in transcriptional activation assay with 3.2 nM TTNPB |
Bioorg Med Chem Lett 9: 743-8 (1999)
BindingDB Entry DOI: 10.7270/Q26Q1WFP |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor alpha
(Homo sapiens (Human)) | BDBM50075879

(4-(2,2-Dimethyl-4-p-tolyl-2H-1-thia-anthracen-6-yl...)Show SMILES Cc1ccc(cc1)C1=CC(C)(C)Sc2cc3ccc(cc3cc12)-c1ccc(cc1)C(O)=O |t:8| Show InChI InChI=1S/C29H24O2S/c1-18-4-6-20(7-5-18)26-17-29(2,3)32-27-16-23-13-12-22(14-24(23)15-25(26)27)19-8-10-21(11-9-19)28(30)31/h4-17H,1-3H3,(H,30,31) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Pharmaceuticals
Curated by ChEMBL
| Assay Description
Antagonistic activity against RAR gamma in transcriptional activation assay with 3.2 nM TTNPB |
Bioorg Med Chem Lett 9: 743-8 (1999)
BindingDB Entry DOI: 10.7270/Q26Q1WFP |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Homo sapiens (Human)) | BDBM50075879

(4-(2,2-Dimethyl-4-p-tolyl-2H-1-thia-anthracen-6-yl...)Show SMILES Cc1ccc(cc1)C1=CC(C)(C)Sc2cc3ccc(cc3cc12)-c1ccc(cc1)C(O)=O |t:8| Show InChI InChI=1S/C29H24O2S/c1-18-4-6-20(7-5-18)26-17-29(2,3)32-27-16-23-13-12-22(14-24(23)15-25(26)27)19-8-10-21(11-9-19)28(30)31/h4-17H,1-3H3,(H,30,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Pharmaceuticals
Curated by ChEMBL
| Assay Description
Antagonistic activity against RAR beta in transcriptional activation assay with 10 nM TTNPB |
Bioorg Med Chem Lett 9: 743-8 (1999)
BindingDB Entry DOI: 10.7270/Q26Q1WFP |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50075877
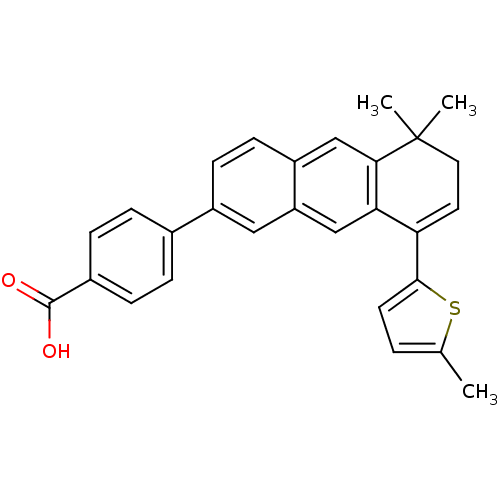
(4-[5,5-Dimethyl-8-(5-methyl-thiophen-2-yl)-5,6-dih...)Show SMILES Cc1ccc(s1)C1=CCC(C)(C)c2cc3ccc(cc3cc12)-c1ccc(cc1)C(O)=O |t:7| Show InChI InChI=1S/C28H24O2S/c1-17-4-11-26(31-17)23-12-13-28(2,3)25-16-21-10-9-20(14-22(21)15-24(23)25)18-5-7-19(8-6-18)27(29)30/h4-12,14-16H,13H2,1-3H3,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Pharmaceuticals
Curated by ChEMBL
| Assay Description
Antagonistic activity against RAR alpha in transcriptional activation assay with 32 nM TTNPB |
Bioorg Med Chem Lett 9: 743-8 (1999)
BindingDB Entry DOI: 10.7270/Q26Q1WFP |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50075880

(4-(5,5-Dimethyl-8-p-tolyl-5,6-dihydro-anthracen-2-...)Show SMILES Cc1ccc(cc1)C1=CCC(C)(C)c2cc3ccc(cc3cc12)-c1ccc(cc1)C(O)=O |t:8| Show InChI InChI=1S/C30H26O2/c1-19-4-6-21(7-5-19)26-14-15-30(2,3)28-18-24-13-12-23(16-25(24)17-27(26)28)20-8-10-22(11-9-20)29(31)32/h4-14,16-18H,15H2,1-3H3,(H,31,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Pharmaceuticals
Curated by ChEMBL
| Assay Description
Binding affinity of [3H]- RA to baculovirus expressed human Retinoic acid receptor beta |
Bioorg Med Chem Lett 9: 743-8 (1999)
BindingDB Entry DOI: 10.7270/Q26Q1WFP |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50075878
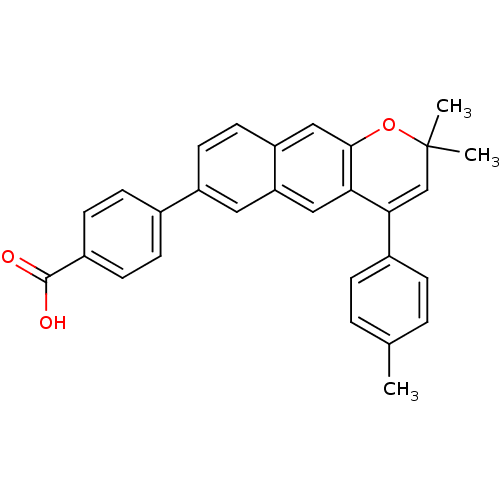
(4-(2,2-Dimethyl-4-p-tolyl-2H-benzo[g]chromen-7-yl)...)Show SMILES Cc1ccc(cc1)C1=CC(C)(C)Oc2cc3ccc(cc3cc12)-c1ccc(cc1)C(O)=O |t:8| Show InChI InChI=1S/C29H24O3/c1-18-4-6-20(7-5-18)26-17-29(2,3)32-27-16-23-13-12-22(14-24(23)15-25(26)27)19-8-10-21(11-9-19)28(30)31/h4-17H,1-3H3,(H,30,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Pharmaceuticals
Curated by ChEMBL
| Assay Description
Binding affinity of [3H]- RA to baculovirus expressed human Retinoic acid receptor beta |
Bioorg Med Chem Lett 9: 743-8 (1999)
BindingDB Entry DOI: 10.7270/Q26Q1WFP |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50097821
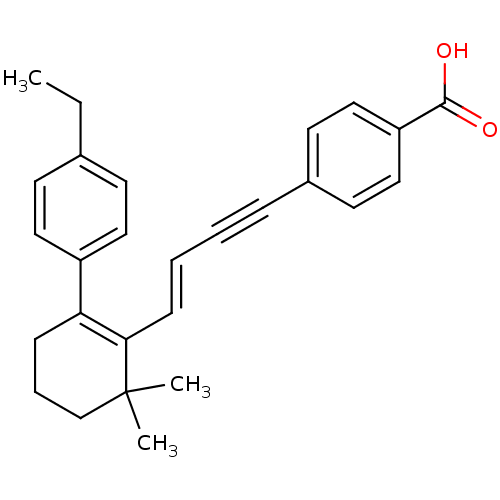
(4-{(E)-4-[2-(4-Ethyl-phenyl)-6,6-dimethyl-cyclohex...)Show SMILES CCc1ccc(cc1)C1=C(\C=C\C#Cc2ccc(cc2)C(O)=O)C(C)(C)CCC1 |c:9| Show InChI InChI=1S/C27H28O2/c1-4-20-11-15-22(16-12-20)24-9-7-19-27(2,3)25(24)10-6-5-8-21-13-17-23(18-14-21)26(28)29/h6,10-18H,4,7,9,19H2,1-3H3,(H,28,29)/b10-6+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Antagonist activity of TTNPB (10 nM) function at retinoic acid receptor beta |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50029774
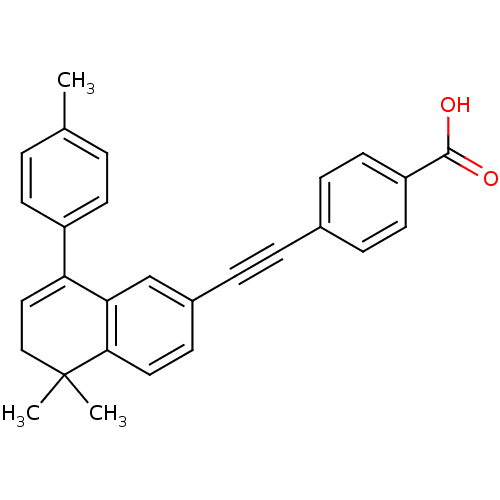
(4-(5,5-Dimethyl-8-p-tolyl-5,6-dihydro-naphthalen-2...)Show SMILES Cc1ccc(cc1)C1=CCC(C)(C)c2ccc(cc12)C#Cc1ccc(cc1)C(O)=O |t:8| Show InChI InChI=1S/C28H24O2/c1-19-4-11-22(12-5-19)24-16-17-28(2,3)26-15-10-21(18-25(24)26)7-6-20-8-13-23(14-9-20)27(29)30/h4-5,8-16,18H,17H2,1-3H3,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Binding affinity towards retinoic acid receptor alpha was determined using [3H]-ATRA (5 nM) as radioligand |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50075876
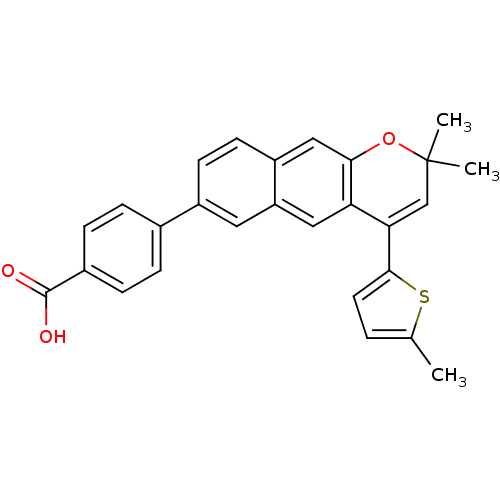
(4-[2,2-Dimethyl-4-(5-methyl-thiophen-2-yl)-2H-benz...)Show SMILES Cc1ccc(s1)C1=CC(C)(C)Oc2cc3ccc(cc3cc12)-c1ccc(cc1)C(O)=O |t:7| Show InChI InChI=1S/C27H22O3S/c1-16-4-11-25(31-16)23-15-27(2,3)30-24-14-20-10-9-19(12-21(20)13-22(23)24)17-5-7-18(8-6-17)26(28)29/h4-15H,1-3H3,(H,28,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Pharmaceuticals
Curated by ChEMBL
| Assay Description
Antagonistic activity against RAR gamma in transcriptional activation assay with 3.2 nM TTNPB |
Bioorg Med Chem Lett 9: 743-8 (1999)
BindingDB Entry DOI: 10.7270/Q26Q1WFP |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50097823
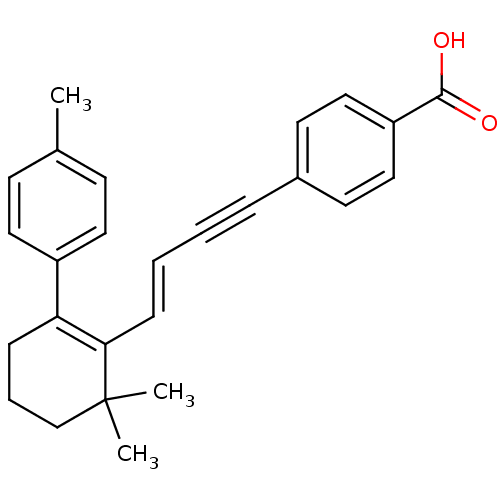
(4-[(E)-4-(6,6-Dimethyl-2-p-tolyl-cyclohex-1-enyl)-...)Show SMILES Cc1ccc(cc1)C1=C(\C=C\C#Cc2ccc(cc2)C(O)=O)C(C)(C)CCC1 |c:8| Show InChI InChI=1S/C26H26O2/c1-19-10-14-21(15-11-19)23-8-6-18-26(2,3)24(23)9-5-4-7-20-12-16-22(17-13-20)25(27)28/h5,9-17H,6,8,18H2,1-3H3,(H,27,28)/b9-5+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Antagonist activity of TTNPB (10 nM) function at retinoic acid receptor gamma |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor alpha
(Homo sapiens (Human)) | BDBM50075878

(4-(2,2-Dimethyl-4-p-tolyl-2H-benzo[g]chromen-7-yl)...)Show SMILES Cc1ccc(cc1)C1=CC(C)(C)Oc2cc3ccc(cc3cc12)-c1ccc(cc1)C(O)=O |t:8| Show InChI InChI=1S/C29H24O3/c1-18-4-6-20(7-5-18)26-17-29(2,3)32-27-16-23-13-12-22(14-24(23)15-25(26)27)19-8-10-21(11-9-19)28(30)31/h4-17H,1-3H3,(H,30,31) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| n/a | n/a | 5.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Pharmaceuticals
Curated by ChEMBL
| Assay Description
Binding affinity of [3H]- RA to baculovirus expressed human RAR alpha |
Bioorg Med Chem Lett 9: 743-8 (1999)
BindingDB Entry DOI: 10.7270/Q26Q1WFP |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Homo sapiens (Human)) | BDBM50075876

(4-[2,2-Dimethyl-4-(5-methyl-thiophen-2-yl)-2H-benz...)Show SMILES Cc1ccc(s1)C1=CC(C)(C)Oc2cc3ccc(cc3cc12)-c1ccc(cc1)C(O)=O |t:7| Show InChI InChI=1S/C27H22O3S/c1-16-4-11-25(31-16)23-15-27(2,3)30-24-14-20-10-9-19(12-21(20)13-22(23)24)17-5-7-18(8-6-17)26(28)29/h4-15H,1-3H3,(H,28,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 6.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Pharmaceuticals
Curated by ChEMBL
| Assay Description
Antagonistic activity against RAR beta in transcriptional activation assay with 10 nM TTNPB |
Bioorg Med Chem Lett 9: 743-8 (1999)
BindingDB Entry DOI: 10.7270/Q26Q1WFP |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor alpha
(Homo sapiens (Human)) | BDBM50075876

(4-[2,2-Dimethyl-4-(5-methyl-thiophen-2-yl)-2H-benz...)Show SMILES Cc1ccc(s1)C1=CC(C)(C)Oc2cc3ccc(cc3cc12)-c1ccc(cc1)C(O)=O |t:7| Show InChI InChI=1S/C27H22O3S/c1-16-4-11-25(31-16)23-15-27(2,3)30-24-14-20-10-9-19(12-21(20)13-22(23)24)17-5-7-18(8-6-17)26(28)29/h4-15H,1-3H3,(H,28,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 6.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Pharmaceuticals
Curated by ChEMBL
| Assay Description
Antagonistic activity against RAR alpha in transcriptional activation assay with 32 nM TTNPB |
Bioorg Med Chem Lett 9: 743-8 (1999)
BindingDB Entry DOI: 10.7270/Q26Q1WFP |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Homo sapiens (Human)) | BDBM50075878

(4-(2,2-Dimethyl-4-p-tolyl-2H-benzo[g]chromen-7-yl)...)Show SMILES Cc1ccc(cc1)C1=CC(C)(C)Oc2cc3ccc(cc3cc12)-c1ccc(cc1)C(O)=O |t:8| Show InChI InChI=1S/C29H24O3/c1-18-4-6-20(7-5-18)26-17-29(2,3)32-27-16-23-13-12-22(14-24(23)15-25(26)27)19-8-10-21(11-9-19)28(30)31/h4-17H,1-3H3,(H,30,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| n/a | n/a | 7.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Pharmaceuticals
Curated by ChEMBL
| Assay Description
Antagonistic activity against RAR beta in transcriptional activation assay with 10 nM TTNPB |
Bioorg Med Chem Lett 9: 743-8 (1999)
BindingDB Entry DOI: 10.7270/Q26Q1WFP |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Homo sapiens (Human)) | BDBM50075877

(4-[5,5-Dimethyl-8-(5-methyl-thiophen-2-yl)-5,6-dih...)Show SMILES Cc1ccc(s1)C1=CCC(C)(C)c2cc3ccc(cc3cc12)-c1ccc(cc1)C(O)=O |t:7| Show InChI InChI=1S/C28H24O2S/c1-17-4-11-26(31-17)23-12-13-28(2,3)25-16-21-10-9-20(14-22(21)15-24(23)25)18-5-7-19(8-6-18)27(29)30/h4-12,14-16H,13H2,1-3H3,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Pharmaceuticals
Curated by ChEMBL
| Assay Description
Antagonistic activity against RAR gamma in transcriptional activation assay with 3.2 nM TTNPB |
Bioorg Med Chem Lett 9: 743-8 (1999)
BindingDB Entry DOI: 10.7270/Q26Q1WFP |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor alpha
(Homo sapiens (Human)) | BDBM50075877

(4-[5,5-Dimethyl-8-(5-methyl-thiophen-2-yl)-5,6-dih...)Show SMILES Cc1ccc(s1)C1=CCC(C)(C)c2cc3ccc(cc3cc12)-c1ccc(cc1)C(O)=O |t:7| Show InChI InChI=1S/C28H24O2S/c1-17-4-11-26(31-17)23-12-13-28(2,3)25-16-21-10-9-20(14-22(21)15-24(23)25)18-5-7-19(8-6-18)27(29)30/h4-12,14-16H,13H2,1-3H3,(H,29,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Pharmaceuticals
Curated by ChEMBL
| Assay Description
Antagonistic activity against RAR beta in transcriptional activation assay with 10 nM TTNPB |
Bioorg Med Chem Lett 9: 743-8 (1999)
BindingDB Entry DOI: 10.7270/Q26Q1WFP |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Homo sapiens (Human)) | BDBM50029774

(4-(5,5-Dimethyl-8-p-tolyl-5,6-dihydro-naphthalen-2...)Show SMILES Cc1ccc(cc1)C1=CCC(C)(C)c2ccc(cc12)C#Cc1ccc(cc1)C(O)=O |t:8| Show InChI InChI=1S/C28H24O2/c1-19-4-11-22(12-5-19)24-16-17-28(2,3)26-15-10-21(18-25(24)26)7-6-20-8-13-23(14-9-20)27(29)30/h4-5,8-16,18H,17H2,1-3H3,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Antagonist activity of TTNPB (10 nM) function at retinoic acid receptor beta |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor alpha
(Homo sapiens (Human)) | BDBM50075880

(4-(5,5-Dimethyl-8-p-tolyl-5,6-dihydro-anthracen-2-...)Show SMILES Cc1ccc(cc1)C1=CCC(C)(C)c2cc3ccc(cc3cc12)-c1ccc(cc1)C(O)=O |t:8| Show InChI InChI=1S/C30H26O2/c1-19-4-6-21(7-5-19)26-14-15-30(2,3)28-18-24-13-12-23(16-25(24)17-27(26)28)20-8-10-22(11-9-20)29(31)32/h4-14,16-18H,15H2,1-3H3,(H,31,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Pharmaceuticals
Curated by ChEMBL
| Assay Description
Antagonistic activity against RAR alpha in transcriptional activation assay with 32 nM TTNPB |
Bioorg Med Chem Lett 9: 743-8 (1999)
BindingDB Entry DOI: 10.7270/Q26Q1WFP |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Homo sapiens (Human)) | BDBM50075880

(4-(5,5-Dimethyl-8-p-tolyl-5,6-dihydro-anthracen-2-...)Show SMILES Cc1ccc(cc1)C1=CCC(C)(C)c2cc3ccc(cc3cc12)-c1ccc(cc1)C(O)=O |t:8| Show InChI InChI=1S/C30H26O2/c1-19-4-6-21(7-5-19)26-14-15-30(2,3)28-18-24-13-12-23(16-25(24)17-27(26)28)20-8-10-22(11-9-20)29(31)32/h4-14,16-18H,15H2,1-3H3,(H,31,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Pharmaceuticals
Curated by ChEMBL
| Assay Description
Antagonistic activity against RAR beta in transcriptional activation assay with 10 nM TTNPB |
Bioorg Med Chem Lett 9: 743-8 (1999)
BindingDB Entry DOI: 10.7270/Q26Q1WFP |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Homo sapiens (Human)) | BDBM50097823

(4-[(E)-4-(6,6-Dimethyl-2-p-tolyl-cyclohex-1-enyl)-...)Show SMILES Cc1ccc(cc1)C1=C(\C=C\C#Cc2ccc(cc2)C(O)=O)C(C)(C)CCC1 |c:8| Show InChI InChI=1S/C26H26O2/c1-19-10-14-21(15-11-19)23-8-6-18-26(2,3)24(23)9-5-4-7-20-12-16-22(17-13-20)25(27)28/h5,9-17H,6,8,18H2,1-3H3,(H,27,28)/b9-5+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Binding affinity towards retinoic acid receptor beta was determined using [3H]-ATRA (5 nM) as radioligand |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Homo sapiens (Human)) | BDBM50097822
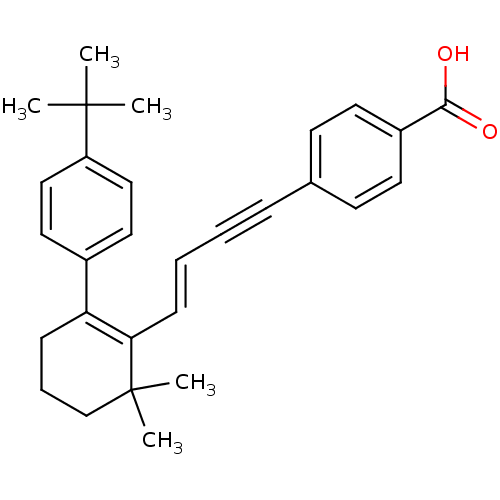
(4-{(E)-4-[2-(4-tert-Butyl-phenyl)-6,6-dimethyl-cyc...)Show SMILES CC(C)(C)c1ccc(cc1)C1=C(\C=C\C#Cc2ccc(cc2)C(O)=O)C(C)(C)CCC1 |c:11| Show InChI InChI=1S/C29H32O2/c1-28(2,3)24-18-16-22(17-19-24)25-10-8-20-29(4,5)26(25)11-7-6-9-21-12-14-23(15-13-21)27(30)31/h7,11-19H,8,10,20H2,1-5H3,(H,30,31)/b11-7+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 25 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Antagonist activity of TTNPB (10 nM) function at retinoic acid receptor gamma |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor alpha
(Homo sapiens (Human)) | BDBM50029774

(4-(5,5-Dimethyl-8-p-tolyl-5,6-dihydro-naphthalen-2...)Show SMILES Cc1ccc(cc1)C1=CCC(C)(C)c2ccc(cc12)C#Cc1ccc(cc1)C(O)=O |t:8| Show InChI InChI=1S/C28H24O2/c1-19-4-11-22(12-5-19)24-16-17-28(2,3)26-15-10-21(18-25(24)26)7-6-20-8-13-23(14-9-20)27(29)30/h4-5,8-16,18H,17H2,1-3H3,(H,29,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 31 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Antagonist activity of TTNPB (10 nM) function at retinoic acid receptor alpha |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50097820
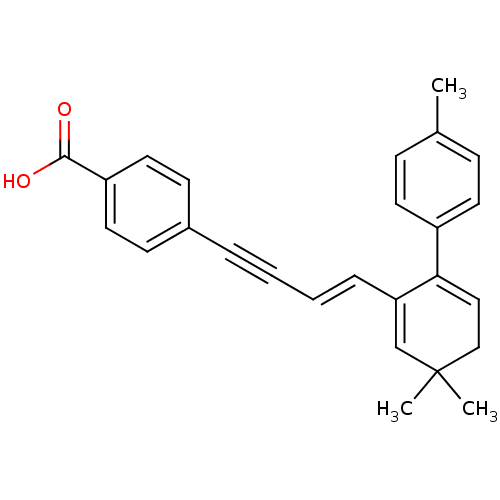
(4-[(E)-4-(3,3-Dimethyl-6-p-tolyl-cyclohexa-1,5-die...)Show SMILES Cc1ccc(cc1)C1=CCC(C)(C)C=C1\C=C\C#Cc1ccc(cc1)C(O)=O |c:14,t:8| Show InChI InChI=1S/C26H24O2/c1-19-8-12-21(13-9-19)24-16-17-26(2,3)18-23(24)7-5-4-6-20-10-14-22(15-11-20)25(27)28/h5,7-16,18H,17H2,1-3H3,(H,27,28)/b7-5+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 31 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Antagonist activity of TTNPB (10 nM) function at retinoic acid receptor alpha |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Homo sapiens (Human)) | BDBM50097821

(4-{(E)-4-[2-(4-Ethyl-phenyl)-6,6-dimethyl-cyclohex...)Show SMILES CCc1ccc(cc1)C1=C(\C=C\C#Cc2ccc(cc2)C(O)=O)C(C)(C)CCC1 |c:9| Show InChI InChI=1S/C27H28O2/c1-4-20-11-15-22(16-12-20)24-9-7-19-27(2,3)25(24)10-6-5-8-21-13-17-23(18-14-21)26(28)29/h6,10-18H,4,7,9,19H2,1-3H3,(H,28,29)/b10-6+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Antagonist activity of TTNPB (10 nM) function at retinoic acid receptor beta |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Homo sapiens (Human)) | BDBM50097820

(4-[(E)-4-(3,3-Dimethyl-6-p-tolyl-cyclohexa-1,5-die...)Show SMILES Cc1ccc(cc1)C1=CCC(C)(C)C=C1\C=C\C#Cc1ccc(cc1)C(O)=O |c:14,t:8| Show InChI InChI=1S/C26H24O2/c1-19-8-12-21(13-9-19)24-16-17-26(2,3)18-23(24)7-5-4-6-20-10-14-22(15-11-20)25(27)28/h5,7-16,18H,17H2,1-3H3,(H,27,28)/b7-5+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 53 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Antagonist activity of TTNPB (10 nM) function at retinoic acid receptor beta |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor alpha
(Homo sapiens (Human)) | BDBM50097823

(4-[(E)-4-(6,6-Dimethyl-2-p-tolyl-cyclohex-1-enyl)-...)Show SMILES Cc1ccc(cc1)C1=C(\C=C\C#Cc2ccc(cc2)C(O)=O)C(C)(C)CCC1 |c:8| Show InChI InChI=1S/C26H26O2/c1-19-10-14-21(15-11-19)23-8-6-18-26(2,3)24(23)9-5-4-7-20-12-16-22(17-13-20)25(27)28/h5,9-17H,6,8,18H2,1-3H3,(H,27,28)/b9-5+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 210 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Antagonist activity of TTNPB (10 nM) function at retinoic acid receptor gamma |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor alpha
(Homo sapiens (Human)) | BDBM50097821

(4-{(E)-4-[2-(4-Ethyl-phenyl)-6,6-dimethyl-cyclohex...)Show SMILES CCc1ccc(cc1)C1=C(\C=C\C#Cc2ccc(cc2)C(O)=O)C(C)(C)CCC1 |c:9| Show InChI InChI=1S/C27H28O2/c1-4-20-11-15-22(16-12-20)24-9-7-19-27(2,3)25(24)10-6-5-8-21-13-17-23(18-14-21)26(28)29/h6,10-18H,4,7,9,19H2,1-3H3,(H,28,29)/b10-6+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 220 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Antagonist activity of TTNPB (10 nM) function at retinoic acid receptor alpha |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor alpha
(Homo sapiens (Human)) | BDBM50097820

(4-[(E)-4-(3,3-Dimethyl-6-p-tolyl-cyclohexa-1,5-die...)Show SMILES Cc1ccc(cc1)C1=CCC(C)(C)C=C1\C=C\C#Cc1ccc(cc1)C(O)=O |c:14,t:8| Show InChI InChI=1S/C26H24O2/c1-19-8-12-21(13-9-19)24-16-17-26(2,3)18-23(24)7-5-4-6-20-10-14-22(15-11-20)25(27)28/h5,7-16,18H,17H2,1-3H3,(H,27,28)/b7-5+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 225 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Antagonist activity of TTNPB (10 nM) function at retinoic acid receptor alpha |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50097822

(4-{(E)-4-[2-(4-tert-Butyl-phenyl)-6,6-dimethyl-cyc...)Show SMILES CC(C)(C)c1ccc(cc1)C1=C(\C=C\C#Cc2ccc(cc2)C(O)=O)C(C)(C)CCC1 |c:11| Show InChI InChI=1S/C29H32O2/c1-28(2,3)24-18-16-22(17-19-24)25-10-8-20-29(4,5)26(25)11-7-6-9-21-12-14-23(15-13-21)27(30)31/h7,11-19H,8,10,20H2,1-5H3,(H,30,31)/b11-7+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 331 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Antagonist activity of TTNPB (10 nM) function at retinoic acid receptor gamma |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor alpha
(Homo sapiens (Human)) | BDBM50097822

(4-{(E)-4-[2-(4-tert-Butyl-phenyl)-6,6-dimethyl-cyc...)Show SMILES CC(C)(C)c1ccc(cc1)C1=C(\C=C\C#Cc2ccc(cc2)C(O)=O)C(C)(C)CCC1 |c:11| Show InChI InChI=1S/C29H32O2/c1-28(2,3)24-18-16-22(17-19-24)25-10-8-20-29(4,5)26(25)11-7-6-9-21-12-14-23(15-13-21)27(30)31/h7,11-19H,8,10,20H2,1-5H3,(H,30,31)/b11-7+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 875 | n/a | n/a | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit TTNPB-induced transactivation at retinoic acid receptor alpha |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor alpha
(Homo sapiens (Human)) | BDBM50097821

(4-{(E)-4-[2-(4-Ethyl-phenyl)-6,6-dimethyl-cyclohex...)Show SMILES CCc1ccc(cc1)C1=C(\C=C\C#Cc2ccc(cc2)C(O)=O)C(C)(C)CCC1 |c:9| Show InChI InChI=1S/C27H28O2/c1-4-20-11-15-22(16-12-20)24-9-7-19-27(2,3)25(24)10-6-5-8-21-13-17-23(18-14-21)26(28)29/h6,10-18H,4,7,9,19H2,1-3H3,(H,28,29)/b10-6+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 136 | n/a | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Antagonist activity of TTNPB (10 nM) function at retinoic acid receptor beta |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM31883

(9-cis-retinoic acid (9cRA) | ALL-TRANS-RETINOIC AC...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C/C=C/C(/C)=C/C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8+,16-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| MMDB
PubMed
| n/a | n/a | n/a | n/a | 6 | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Binding affinity towards retinoic acid receptor gamma was determined using [3H]-ATRA (5 nM) as radioligand |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor alpha
(Homo sapiens (Human)) | BDBM50029774

(4-(5,5-Dimethyl-8-p-tolyl-5,6-dihydro-naphthalen-2...)Show SMILES Cc1ccc(cc1)C1=CCC(C)(C)c2ccc(cc12)C#Cc1ccc(cc1)C(O)=O |t:8| Show InChI InChI=1S/C28H24O2/c1-19-4-11-22(12-5-19)24-16-17-28(2,3)26-15-10-21(18-25(24)26)7-6-20-8-13-23(14-9-20)27(29)30/h4-5,8-16,18H,17H2,1-3H3,(H,29,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Binding affinity towards retinoic acid receptor alpha was determined using [3H]-ATRA (5 nM) as radioligand |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Homo sapiens (Human)) | BDBM50097819
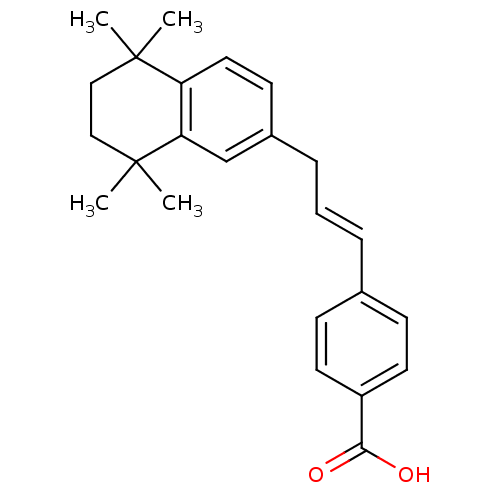
(4-[(E)-3-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-n...)Show SMILES CC1(C)CCC(C)(C)c2cc(C\C=C\c3ccc(cc3)C(O)=O)ccc12 Show InChI InChI=1S/C24H28O2/c1-23(2)14-15-24(3,4)21-16-18(10-13-20(21)23)7-5-6-17-8-11-19(12-9-17)22(25)26/h5-6,8-13,16H,7,14-15H2,1-4H3,(H,25,26)/b6-5+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 8 | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit TTNPB-induced transactivation at retinoic acid receptor beta |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50097823

(4-[(E)-4-(6,6-Dimethyl-2-p-tolyl-cyclohex-1-enyl)-...)Show SMILES Cc1ccc(cc1)C1=C(\C=C\C#Cc2ccc(cc2)C(O)=O)C(C)(C)CCC1 |c:8| Show InChI InChI=1S/C26H26O2/c1-19-10-14-21(15-11-19)23-8-6-18-26(2,3)24(23)9-5-4-7-20-12-16-22(17-13-20)25(27)28/h5,9-17H,6,8,18H2,1-3H3,(H,27,28)/b9-5+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Binding affinity towards retinoic acid receptor beta was determined using [3H]-ATRA (5 nM) as radioligand |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50097824
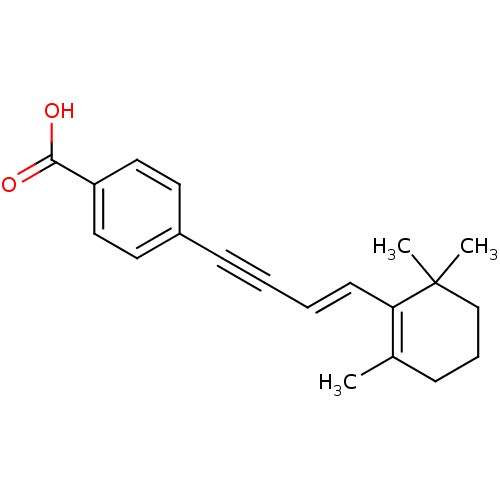
(4-[(E)-4-(2,6,6-Trimethyl-cyclohex-1-enyl)-but-3-e...)Show SMILES CC1=C(\C=C\C#Cc2ccc(cc2)C(O)=O)C(C)(C)CCC1 |c:1| Show InChI InChI=1S/C20H22O2/c1-15-7-6-14-20(2,3)18(15)9-5-4-8-16-10-12-17(13-11-16)19(21)22/h5,9-13H,6-7,14H2,1-3H3,(H,21,22)/b9-5+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | 190 | n/a | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit TTNPB-induced transactivation at retinoic acid receptor beta |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Homo sapiens (Human)) | BDBM50029774

(4-(5,5-Dimethyl-8-p-tolyl-5,6-dihydro-naphthalen-2...)Show SMILES Cc1ccc(cc1)C1=CCC(C)(C)c2ccc(cc12)C#Cc1ccc(cc1)C(O)=O |t:8| Show InChI InChI=1S/C28H24O2/c1-19-4-11-22(12-5-19)24-16-17-28(2,3)26-15-10-21(18-25(24)26)7-6-20-8-13-23(14-9-20)27(29)30/h4-5,8-16,18H,17H2,1-3H3,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit TTNPB-induced transactivation at retinoic acid receptor gamma |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor alpha
(Homo sapiens (Human)) | BDBM50097824

(4-[(E)-4-(2,6,6-Trimethyl-cyclohex-1-enyl)-but-3-e...)Show SMILES CC1=C(\C=C\C#Cc2ccc(cc2)C(O)=O)C(C)(C)CCC1 |c:1| Show InChI InChI=1S/C20H22O2/c1-15-7-6-14-20(2,3)18(15)9-5-4-8-16-10-12-17(13-11-16)19(21)22/h5,9-13H,6-7,14H2,1-3H3,(H,21,22)/b9-5+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | n/a | 435 | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Antagonist activity of TTNPB (10 nM) function at retinoic acid receptor gamma |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Homo sapiens (Human)) | BDBM50097821

(4-{(E)-4-[2-(4-Ethyl-phenyl)-6,6-dimethyl-cyclohex...)Show SMILES CCc1ccc(cc1)C1=C(\C=C\C#Cc2ccc(cc2)C(O)=O)C(C)(C)CCC1 |c:9| Show InChI InChI=1S/C27H28O2/c1-4-20-11-15-22(16-12-20)24-9-7-19-27(2,3)25(24)10-6-5-8-21-13-17-23(18-14-21)26(28)29/h6,10-18H,4,7,9,19H2,1-3H3,(H,28,29)/b10-6+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit TTNPB-induced transactivation at retinoic acid receptor gamma |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50097819

(4-[(E)-3-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-n...)Show SMILES CC1(C)CCC(C)(C)c2cc(C\C=C\c3ccc(cc3)C(O)=O)ccc12 Show InChI InChI=1S/C24H28O2/c1-23(2)14-15-24(3,4)21-16-18(10-13-20(21)23)7-5-6-17-8-11-19(12-9-17)22(25)26/h5-6,8-13,16H,7,14-15H2,1-4H3,(H,25,26)/b6-5+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 4 | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Binding affinity towards retinoic acid receptor beta was determined using [3H]-ATRA (5 nM) as radioligand |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Homo sapiens (Human)) | BDBM50097824

(4-[(E)-4-(2,6,6-Trimethyl-cyclohex-1-enyl)-but-3-e...)Show SMILES CC1=C(\C=C\C#Cc2ccc(cc2)C(O)=O)C(C)(C)CCC1 |c:1| Show InChI InChI=1S/C20H22O2/c1-15-7-6-14-20(2,3)18(15)9-5-4-8-16-10-12-17(13-11-16)19(21)22/h5,9-13H,6-7,14H2,1-3H3,(H,21,22)/b9-5+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | n/a | 7 | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Binding affinity towards retinoic acid receptor alpha was determined using [3H]-ATRA (5 nM) as radioligand |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor alpha
(Homo sapiens (Human)) | BDBM31883

(9-cis-retinoic acid (9cRA) | ALL-TRANS-RETINOIC AC...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C/C=C/C(/C)=C/C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8+,16-14+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit TTNPB-induced transactivation at retinoic acid receptor beta |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50097822

(4-{(E)-4-[2-(4-tert-Butyl-phenyl)-6,6-dimethyl-cyc...)Show SMILES CC(C)(C)c1ccc(cc1)C1=C(\C=C\C#Cc2ccc(cc2)C(O)=O)C(C)(C)CCC1 |c:11| Show InChI InChI=1S/C29H32O2/c1-28(2,3)24-18-16-22(17-19-24)25-10-8-20-29(4,5)26(25)11-7-6-9-21-12-14-23(15-13-21)27(30)31/h7,11-19H,8,10,20H2,1-5H3,(H,30,31)/b11-7+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Binding affinity towards retinoic acid receptor gamma was determined using [3H]-ATRA (5 nM) as radioligand |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor alpha
(Homo sapiens (Human)) | BDBM50097819

(4-[(E)-3-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-n...)Show SMILES CC1(C)CCC(C)(C)c2cc(C\C=C\c3ccc(cc3)C(O)=O)ccc12 Show InChI InChI=1S/C24H28O2/c1-23(2)14-15-24(3,4)21-16-18(10-13-20(21)23)7-5-6-17-8-11-19(12-9-17)22(25)26/h5-6,8-13,16H,7,14-15H2,1-4H3,(H,25,26)/b6-5+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 30 | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Antagonist activity of TTNPB (10 nM) function at retinoic acid receptor alpha |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor alpha
(Homo sapiens (Human)) | BDBM50097820

(4-[(E)-4-(3,3-Dimethyl-6-p-tolyl-cyclohexa-1,5-die...)Show SMILES Cc1ccc(cc1)C1=CCC(C)(C)C=C1\C=C\C#Cc1ccc(cc1)C(O)=O |c:14,t:8| Show InChI InChI=1S/C26H24O2/c1-19-8-12-21(13-9-19)24-16-17-26(2,3)18-23(24)7-5-4-6-20-10-14-22(15-11-20)25(27)28/h5,7-16,18H,17H2,1-3H3,(H,27,28)/b7-5+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 129 | n/a | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Binding affinity towards retinoic acid receptor gamma was determined using [3H]-ATRA (5 nM) as radioligand |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Homo sapiens (Human)) | BDBM31883

(9-cis-retinoic acid (9cRA) | ALL-TRANS-RETINOIC AC...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C/C=C/C(/C)=C/C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8+,16-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| MMDB
PubMed
| n/a | n/a | n/a | n/a | 38 | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit TTNPB-induced transactivation at retinoic acid receptor alpha |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Homo sapiens (Human)) | BDBM50097820

(4-[(E)-4-(3,3-Dimethyl-6-p-tolyl-cyclohexa-1,5-die...)Show SMILES Cc1ccc(cc1)C1=CCC(C)(C)C=C1\C=C\C#Cc1ccc(cc1)C(O)=O |c:14,t:8| Show InChI InChI=1S/C26H24O2/c1-19-8-12-21(13-9-19)24-16-17-26(2,3)18-23(24)7-5-4-6-20-10-14-22(15-11-20)25(27)28/h5,7-16,18H,17H2,1-3H3,(H,27,28)/b7-5+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | 35 | n/a | n/a | n/a | n/a | n/a |
Allergan Inc
Curated by ChEMBL
| Assay Description
Binding affinity towards retinoic acid receptor alpha was determined using [3H]-ATRA (5 nM) as radioligand |
Bioorg Med Chem Lett 11: 765-8 (2001)
BindingDB Entry DOI: 10.7270/Q2348JN3 |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Homo sapiens (Human)) | BDBM50075879

(4-(2,2-Dimethyl-4-p-tolyl-2H-1-thia-anthracen-6-yl...)Show SMILES Cc1ccc(cc1)C1=CC(C)(C)Sc2cc3ccc(cc3cc12)-c1ccc(cc1)C(O)=O |t:8| Show InChI InChI=1S/C29H24O2S/c1-18-4-6-20(7-5-18)26-17-29(2,3)32-27-16-23-13-12-22(14-24(23)15-25(26)27)19-8-10-21(11-9-19)28(30)31/h4-17H,1-3H3,(H,30,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a |
Allergan Pharmaceuticals
Curated by ChEMBL
| Assay Description
Binding affinity of [3H]- RA to baculovirus expressed human Retinoic acid receptor beta |
Bioorg Med Chem Lett 9: 743-8 (1999)
BindingDB Entry DOI: 10.7270/Q26Q1WFP |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50075877

(4-[5,5-Dimethyl-8-(5-methyl-thiophen-2-yl)-5,6-dih...)Show SMILES Cc1ccc(s1)C1=CCC(C)(C)c2cc3ccc(cc3cc12)-c1ccc(cc1)C(O)=O |t:7| Show InChI InChI=1S/C28H24O2S/c1-17-4-11-26(31-17)23-12-13-28(2,3)25-16-21-10-9-20(14-22(21)15-24(23)25)18-5-7-19(8-6-18)27(29)30/h4-12,14-16H,13H2,1-3H3,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a |
Allergan Pharmaceuticals
Curated by ChEMBL
| Assay Description
Binding affinity of [3H]- RA to baculovirus expressed human Retinoic acid receptor gamma |
Bioorg Med Chem Lett 9: 743-8 (1999)
BindingDB Entry DOI: 10.7270/Q26Q1WFP |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor alpha
(Homo sapiens (Human)) | BDBM50075877

(4-[5,5-Dimethyl-8-(5-methyl-thiophen-2-yl)-5,6-dih...)Show SMILES Cc1ccc(s1)C1=CCC(C)(C)c2cc3ccc(cc3cc12)-c1ccc(cc1)C(O)=O |t:7| Show InChI InChI=1S/C28H24O2S/c1-17-4-11-26(31-17)23-12-13-28(2,3)25-16-21-10-9-20(14-22(21)15-24(23)25)18-5-7-19(8-6-18)27(29)30/h4-12,14-16H,13H2,1-3H3,(H,29,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a |
Allergan Pharmaceuticals
Curated by ChEMBL
| Assay Description
Binding affinity of [3H]- RA to baculovirus expressed human RAR alpha |
Bioorg Med Chem Lett 9: 743-8 (1999)
BindingDB Entry DOI: 10.7270/Q26Q1WFP |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data