 Found 889 hits with Last Name = 'tada' and Initial = 'h'
Found 889 hits with Last Name = 'tada' and Initial = 'h' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM519
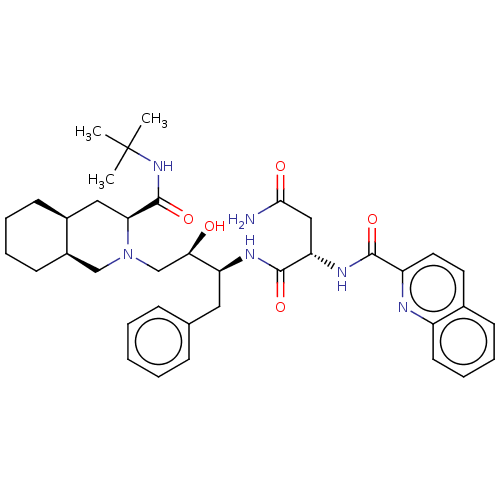
((2S)-N-[(2S,3R)-4-[(3S,4aS,8aS)-3-(tert-butylcarba...)Show SMILES [H][C@@]12CCCC[C@]1([H])CN(C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(N)=O)NC(=O)c1ccc3ccccc3n1)[C@@H](C2)C(=O)NC(C)(C)C |r| Show InChI InChI=1S/C38H50N6O5/c1-38(2,3)43-37(49)32-20-26-14-7-8-15-27(26)22-44(32)23-33(45)30(19-24-11-5-4-6-12-24)41-36(48)31(21-34(39)46)42-35(47)29-18-17-25-13-9-10-16-28(25)40-29/h4-6,9-13,16-18,26-27,30-33,45H,7-8,14-15,19-23H2,1-3H3,(H2,39,46)(H,41,48)(H,42,47)(H,43,49)/t26-,27+,30-,31-,32-,33+/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Products Ltd.
Curated by ChEMBL
| Assay Description
Binding activity against HIV-1 Protease |
J Med Chem 34: 3340-2 (1991)
BindingDB Entry DOI: 10.7270/Q2B858Q3 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM519

((2S)-N-[(2S,3R)-4-[(3S,4aS,8aS)-3-(tert-butylcarba...)Show SMILES [H][C@@]12CCCC[C@]1([H])CN(C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(N)=O)NC(=O)c1ccc3ccccc3n1)[C@@H](C2)C(=O)NC(C)(C)C |r| Show InChI InChI=1S/C38H50N6O5/c1-38(2,3)43-37(49)32-20-26-14-7-8-15-27(26)22-44(32)23-33(45)30(19-24-11-5-4-6-12-24)41-36(48)31(21-34(39)46)42-35(47)29-18-17-25-13-9-10-16-28(25)40-29/h4-6,9-13,16-18,26-27,30-33,45H,7-8,14-15,19-23H2,1-3H3,(H2,39,46)(H,41,48)(H,42,47)(H,43,49)/t26-,27+,30-,31-,32-,33+/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Products Ltd.
Curated by ChEMBL
| Assay Description
The compound was tested for binding activity against HIV-1 protease |
J Med Chem 34: 3340-2 (1991)
BindingDB Entry DOI: 10.7270/Q2B858Q3 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50010497
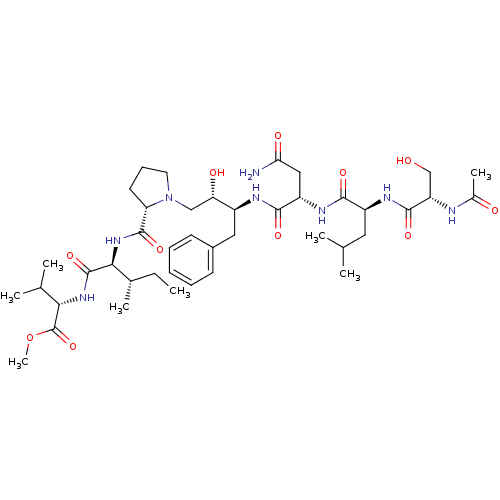
(Acetyl-Ser-Leu-Asn-Phe-[S]-[CH(OH)CH2N]Pro-Ile-Val...)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@@H]1CCCN1C[C@H](O)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(C)=O)C(=O)N[C@@H](C(C)C)C(=O)OC Show InChI InChI=1S/C42H68N8O11/c1-9-25(6)36(41(59)48-35(24(4)5)42(60)61-8)49-40(58)32-16-13-17-50(32)21-33(53)28(19-27-14-11-10-12-15-27)45-38(56)30(20-34(43)54)47-37(55)29(18-23(2)3)46-39(57)31(22-51)44-26(7)52/h10-12,14-15,23-25,28-33,35-36,51,53H,9,13,16-22H2,1-8H3,(H2,43,54)(H,44,52)(H,45,56)(H,46,57)(H,47,55)(H,48,59)(H,49,58)/t25-,28-,29-,30-,31-,32-,33-,35-,36-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Products Ltd.
Curated by ChEMBL
| Assay Description
Binding activity against HIV-1 Protease |
J Med Chem 34: 3340-2 (1991)
BindingDB Entry DOI: 10.7270/Q2B858Q3 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50010497

(Acetyl-Ser-Leu-Asn-Phe-[S]-[CH(OH)CH2N]Pro-Ile-Val...)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@@H]1CCCN1C[C@H](O)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(C)=O)C(=O)N[C@@H](C(C)C)C(=O)OC Show InChI InChI=1S/C42H68N8O11/c1-9-25(6)36(41(59)48-35(24(4)5)42(60)61-8)49-40(58)32-16-13-17-50(32)21-33(53)28(19-27-14-11-10-12-15-27)45-38(56)30(20-34(43)54)47-37(55)29(18-23(2)3)46-39(57)31(22-51)44-26(7)52/h10-12,14-15,23-25,28-33,35-36,51,53H,9,13,16-22H2,1-8H3,(H2,43,54)(H,44,52)(H,45,56)(H,46,57)(H,47,55)(H,48,59)(H,49,58)/t25-,28-,29-,30-,31-,32-,33-,35-,36-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Products Ltd.
Curated by ChEMBL
| Assay Description
The compound was tested for binding activity against HIV-1 Protease |
J Med Chem 34: 3340-2 (1991)
BindingDB Entry DOI: 10.7270/Q2B858Q3 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50010495

(Acetyl-Ser-Leu-Asn-Phe-[CH(OH)CH2N]Pro-Ile-Val-OMe...)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@@H]1CCCN1CC(O)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(C)=O)C(=O)N[C@@H](C(C)C)C(=O)OC Show InChI InChI=1S/C42H68N8O11/c1-9-25(6)36(41(59)48-35(24(4)5)42(60)61-8)49-40(58)32-16-13-17-50(32)21-33(53)28(19-27-14-11-10-12-15-27)45-38(56)30(20-34(43)54)47-37(55)29(18-23(2)3)46-39(57)31(22-51)44-26(7)52/h10-12,14-15,23-25,28-33,35-36,51,53H,9,13,16-22H2,1-8H3,(H2,43,54)(H,44,52)(H,45,56)(H,46,57)(H,47,55)(H,48,59)(H,49,58)/t25-,28-,29-,30-,31-,32-,33?,35-,36-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Products Ltd.
Curated by ChEMBL
| Assay Description
The compound was tested for binding activity against HIV-1 Protease |
J Med Chem 34: 3340-2 (1991)
BindingDB Entry DOI: 10.7270/Q2B858Q3 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50010495

(Acetyl-Ser-Leu-Asn-Phe-[CH(OH)CH2N]Pro-Ile-Val-OMe...)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@@H]1CCCN1CC(O)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(C)=O)C(=O)N[C@@H](C(C)C)C(=O)OC Show InChI InChI=1S/C42H68N8O11/c1-9-25(6)36(41(59)48-35(24(4)5)42(60)61-8)49-40(58)32-16-13-17-50(32)21-33(53)28(19-27-14-11-10-12-15-27)45-38(56)30(20-34(43)54)47-37(55)29(18-23(2)3)46-39(57)31(22-51)44-26(7)52/h10-12,14-15,23-25,28-33,35-36,51,53H,9,13,16-22H2,1-8H3,(H2,43,54)(H,44,52)(H,45,56)(H,46,57)(H,47,55)(H,48,59)(H,49,58)/t25-,28-,29-,30-,31-,32-,33?,35-,36-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Products Ltd.
Curated by ChEMBL
| Assay Description
Binding activity against HIV-1 Protease |
J Med Chem 34: 3340-2 (1991)
BindingDB Entry DOI: 10.7270/Q2B858Q3 |
More data for this
Ligand-Target Pair | |
Solute carrier family 15 member 1
(Homo sapiens (Human)) | BDBM50162073

((S)-2-((2-amino-6-oxo-3H-purin-9(6H)-yl)methoxy)et...)Show SMILES CC(C)[C@H](N)C(=O)OCCOCn1cnc2c1nc(N)[nH]c2=O |r| Show InChI InChI=1S/C13H20N6O4/c1-7(2)8(14)12(21)23-4-3-22-6-19-5-16-9-10(19)17-13(15)18-11(9)20/h5,7-8H,3-4,6,14H2,1-2H3,(H3,15,17,18,20)/t8-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 4.08E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
State University of New Jersey
Curated by ChEMBL
| Assay Description
TP_TRANSPORTER: inhibition of Gly-Sar uptake in Xenopus laevis oocytes |
Biochem Biophys Res Commun 250: 246-51 (1998)
Article DOI: 10.1006/bbrc.1998.9298
BindingDB Entry DOI: 10.7270/Q2X92CCB |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM519

((2S)-N-[(2S,3R)-4-[(3S,4aS,8aS)-3-(tert-butylcarba...)Show SMILES [H][C@@]12CCCC[C@]1([H])CN(C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(N)=O)NC(=O)c1ccc3ccccc3n1)[C@@H](C2)C(=O)NC(C)(C)C |r| Show InChI InChI=1S/C38H50N6O5/c1-38(2,3)43-37(49)32-20-26-14-7-8-15-27(26)22-44(32)23-33(45)30(19-24-11-5-4-6-12-24)41-36(48)31(21-34(39)46)42-35(47)29-18-17-25-13-9-10-16-28(25)40-29/h4-6,9-13,16-18,26-27,30-33,45H,7-8,14-15,19-23H2,1-3H3,(H2,39,46)(H,41,48)(H,42,47)(H,43,49)/t26-,27+,30-,31-,32-,33+/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| n/a | n/a | <0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Products Ltd.
Curated by ChEMBL
| Assay Description
Binding activity against HIV-1 Protease |
J Med Chem 34: 3340-2 (1991)
BindingDB Entry DOI: 10.7270/Q2B858Q3 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM519

((2S)-N-[(2S,3R)-4-[(3S,4aS,8aS)-3-(tert-butylcarba...)Show SMILES [H][C@@]12CCCC[C@]1([H])CN(C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(N)=O)NC(=O)c1ccc3ccccc3n1)[C@@H](C2)C(=O)NC(C)(C)C |r| Show InChI InChI=1S/C38H50N6O5/c1-38(2,3)43-37(49)32-20-26-14-7-8-15-27(26)22-44(32)23-33(45)30(19-24-11-5-4-6-12-24)41-36(48)31(21-34(39)46)42-35(47)29-18-17-25-13-9-10-16-28(25)40-29/h4-6,9-13,16-18,26-27,30-33,45H,7-8,14-15,19-23H2,1-3H3,(H2,39,46)(H,41,48)(H,42,47)(H,43,49)/t26-,27+,30-,31-,32-,33+/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| n/a | n/a | <0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Products Ltd.
Curated by ChEMBL
| Assay Description
Binding activity against HIV-1 Protease |
J Med Chem 34: 3340-2 (1991)
BindingDB Entry DOI: 10.7270/Q2B858Q3 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
C-C chemokine receptor type 5
(Homo sapiens (Human)) | BDBM50198936
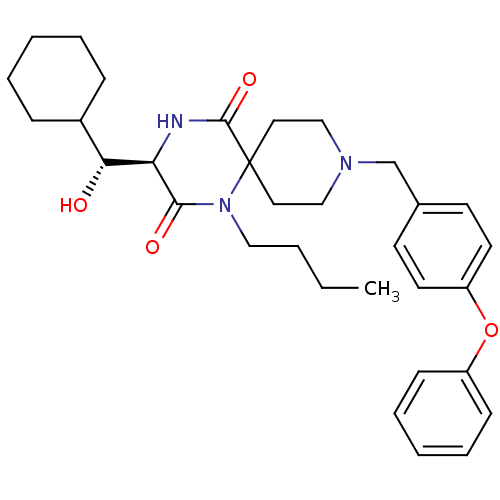
((R)-1-butyl-3-((R)-cyclohexyl-hydroxy-methyl)-9-(4...)Show SMILES CCCCN1C(=O)[C@H](NC(=O)C11CCN(Cc2ccc(Oc3ccccc3)cc2)CC1)[C@H](O)C1CCCCC1 Show InChI InChI=1S/C32H43N3O4/c1-2-3-20-35-30(37)28(29(36)25-10-6-4-7-11-25)33-31(38)32(35)18-21-34(22-19-32)23-24-14-16-27(17-15-24)39-26-12-8-5-9-13-26/h5,8-9,12-17,25,28-29,36H,2-4,6-7,10-11,18-23H2,1H3,(H,33,38)/t28-,29-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Ono Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Displacement of [125I]MIP1-alpha from human CCR5 expressed in CHO cells |
Bioorg Med Chem Lett 17: 727-31 (2007)
Article DOI: 10.1016/j.bmcl.2006.10.084
BindingDB Entry DOI: 10.7270/Q2XK8GCM |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 5
(Homo sapiens (Human)) | BDBM50190513
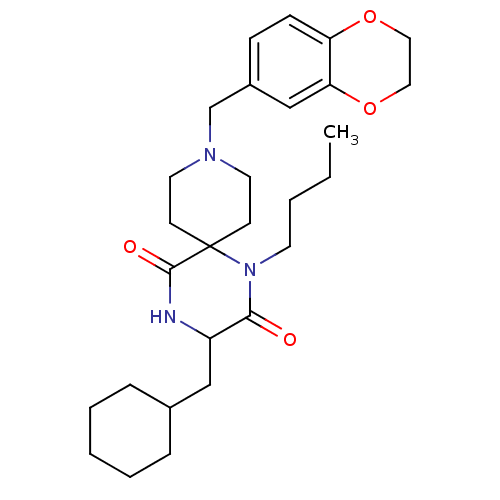
((RS)-1-butyl-3-(cyclohexylmethyl)-9-((2,3-dihydrob...)Show SMILES CCCCN1C(=O)C(CC2CCCCC2)NC(=O)C11CCN(Cc2ccc3OCCOc3c2)CC1 Show InChI InChI=1S/C28H41N3O4/c1-2-3-13-31-26(32)23(18-21-7-5-4-6-8-21)29-27(33)28(31)11-14-30(15-12-28)20-22-9-10-24-25(19-22)35-17-16-34-24/h9-10,19,21,23H,2-8,11-18,20H2,1H3,(H,29,33) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Ono Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of radio-isotope labeled MIP1-alpha binding to human recombinant CCR5 |
J Med Chem 49: 4140-52 (2006)
Article DOI: 10.1021/jm060051s
BindingDB Entry DOI: 10.7270/Q2ZG6T1S |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50229036
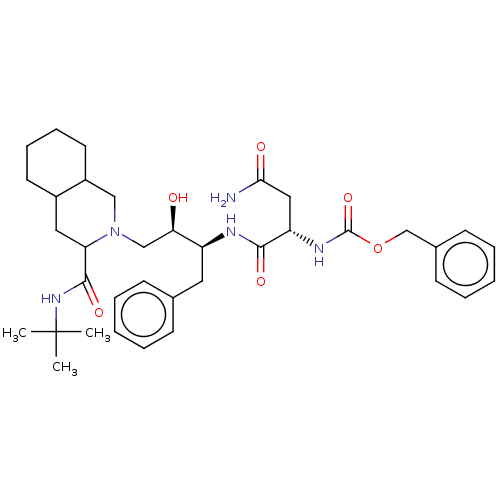
(CHEMBL323014)Show SMILES CC(C)(C)NC(=O)C1CC2CCCCC2CN1C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(N)=O)NC(=O)OCc1ccccc1 Show InChI InChI=1S/C36H51N5O6/c1-36(2,3)40-34(45)30-19-26-16-10-11-17-27(26)21-41(30)22-31(42)28(18-24-12-6-4-7-13-24)38-33(44)29(20-32(37)43)39-35(46)47-23-25-14-8-5-9-15-25/h4-9,12-15,26-31,42H,10-11,16-23H2,1-3H3,(H2,37,43)(H,38,44)(H,39,46)(H,40,45)/t26?,27?,28-,29-,30?,31+/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | <2.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Products Ltd.
Curated by ChEMBL
| Assay Description
Binding activity against HIV-1 Protease |
J Med Chem 34: 3340-2 (1991)
BindingDB Entry DOI: 10.7270/Q2B858Q3 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50229036

(CHEMBL323014)Show SMILES CC(C)(C)NC(=O)C1CC2CCCCC2CN1C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(N)=O)NC(=O)OCc1ccccc1 Show InChI InChI=1S/C36H51N5O6/c1-36(2,3)40-34(45)30-19-26-16-10-11-17-27(26)21-41(30)22-31(42)28(18-24-12-6-4-7-13-24)38-33(44)29(20-32(37)43)39-35(46)47-23-25-14-8-5-9-15-25/h4-9,12-15,26-31,42H,10-11,16-23H2,1-3H3,(H2,37,43)(H,38,44)(H,39,46)(H,40,45)/t26?,27?,28-,29-,30?,31+/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | <2.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Products Ltd.
Curated by ChEMBL
| Assay Description
Binding activity against HIV-1 Protease |
J Med Chem 34: 3340-2 (1991)
BindingDB Entry DOI: 10.7270/Q2B858Q3 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 5
(Homo sapiens (Human)) | BDBM50198939
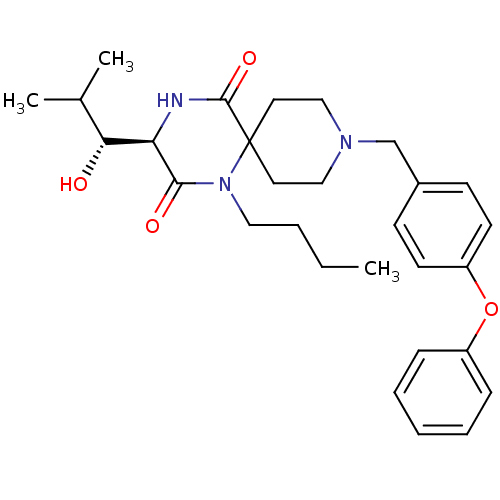
((R)-1-butyl-3-((R)-1-hydroxy-2-methyl-propyl)-9-(4...)Show SMILES CCCCN1C(=O)[C@H](NC(=O)C11CCN(Cc2ccc(Oc3ccccc3)cc2)CC1)[C@H](O)C(C)C Show InChI InChI=1S/C29H39N3O4/c1-4-5-17-32-27(34)25(26(33)21(2)3)30-28(35)29(32)15-18-31(19-16-29)20-22-11-13-24(14-12-22)36-23-9-7-6-8-10-23/h6-14,21,25-26,33H,4-5,15-20H2,1-3H3,(H,30,35)/t25-,26-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Ono Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Displacement of [125I]MIP1-alpha from human CCR5 expressed in CHO cells |
Bioorg Med Chem Lett 17: 727-31 (2007)
Article DOI: 10.1016/j.bmcl.2006.10.084
BindingDB Entry DOI: 10.7270/Q2XK8GCM |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 5
(Homo sapiens (Human)) | BDBM50190525

((RS)-1-butyl-3-(cyclohexylmethyl)-9-(4-methoxybenz...)Show SMILES CCCCN1C(=O)C(CC2CCCCC2)NC(=O)C11CCN(Cc2ccc(OC)cc2)CC1 Show InChI InChI=1S/C27H41N3O3/c1-3-4-16-30-25(31)24(19-21-8-6-5-7-9-21)28-26(32)27(30)14-17-29(18-15-27)20-22-10-12-23(33-2)13-11-22/h10-13,21,24H,3-9,14-20H2,1-2H3,(H,28,32) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Ono Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of radio-isotope labeled MIP1-alpha binding to human recombinant CCR5 |
J Med Chem 49: 4140-52 (2006)
Article DOI: 10.1021/jm060051s
BindingDB Entry DOI: 10.7270/Q2ZG6T1S |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50530873

(CHEMBL4456215)Show SMILES CN(C1CCCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cnc(N)nc2)C1)S(C)(=O)=O Show InChI InChI=1S/C22H30N8O3S2/c1-28(35(2,31)32)16-4-3-5-29(13-16)14-17-10-18-19(34-17)21(30-6-8-33-9-7-30)27-20(26-18)15-11-24-22(23)25-12-15/h10-12,16H,3-9,13-14H2,1-2H3,(H2,23,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50530873

(CHEMBL4456215)Show SMILES CN(C1CCCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cnc(N)nc2)C1)S(C)(=O)=O Show InChI InChI=1S/C22H30N8O3S2/c1-28(35(2,31)32)16-4-3-5-29(13-16)14-17-10-18-19(34-17)21(30-6-8-33-9-7-30)27-20(26-18)15-11-24-22(23)25-12-15/h10-12,16H,3-9,13-14H2,1-2H3,(H2,23,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 5
(Homo sapiens (Human)) | BDBM50336345
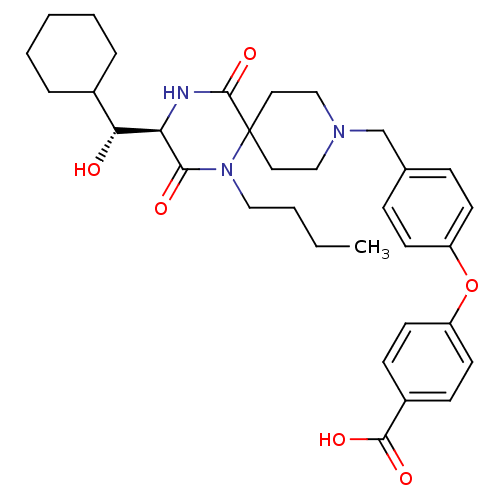
(4-(4-(((R)-1-butyl-3-((R)-cyclohexyl(hydroxy)methy...)Show SMILES CCCCN1C(=O)[C@H](NC(=O)C11CCN(Cc2ccc(Oc3ccc(cc3)C(O)=O)cc2)CC1)[C@H](O)C1CCCCC1 |r| Show InChI InChI=1S/C33H43N3O6/c1-2-3-19-36-30(38)28(29(37)24-7-5-4-6-8-24)34-32(41)33(36)17-20-35(21-18-33)22-23-9-13-26(14-10-23)42-27-15-11-25(12-16-27)31(39)40/h9-16,24,28-29,37H,2-8,17-22H2,1H3,(H,34,41)(H,39,40)/t28-,29-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.62 | n/a | n/a | n/a | n/a | n/a | n/a |
Ono Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Antagonist activity at human CCR5 expressed in HOS cells assessed as inhibition of cell fusion with HIV gp120 expressing HEK293 cells by LTR lucifera... |
Bioorg Med Chem Lett 21: 1141-5 (2011)
Article DOI: 10.1016/j.bmcl.2010.12.109
BindingDB Entry DOI: 10.7270/Q24T6JP0 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 5
(Homo sapiens (Human)) | BDBM50336345

(4-(4-(((R)-1-butyl-3-((R)-cyclohexyl(hydroxy)methy...)Show SMILES CCCCN1C(=O)[C@H](NC(=O)C11CCN(Cc2ccc(Oc3ccc(cc3)C(O)=O)cc2)CC1)[C@H](O)C1CCCCC1 |r| Show InChI InChI=1S/C33H43N3O6/c1-2-3-19-36-30(38)28(29(37)24-7-5-4-6-8-24)34-32(41)33(36)17-20-35(21-18-33)22-23-9-13-26(14-10-23)42-27-15-11-25(12-16-27)31(39)40/h9-16,24,28-29,37H,2-8,17-22H2,1H3,(H,34,41)(H,39,40)/t28-,29-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Ono Pharmaceutical Co. Ltd
Curated by ChEMBL
| Assay Description
Antagonist activity at human CCR5 expressed in CHO cells assessed as inhibition of MIP-1alpha-induced chemotaxis |
Bioorg Med Chem 19: 4028-42 (2011)
Article DOI: 10.1016/j.bmc.2011.05.022
BindingDB Entry DOI: 10.7270/Q2H70G5Q |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 5
(Homo sapiens (Human)) | BDBM50336345

(4-(4-(((R)-1-butyl-3-((R)-cyclohexyl(hydroxy)methy...)Show SMILES CCCCN1C(=O)[C@H](NC(=O)C11CCN(Cc2ccc(Oc3ccc(cc3)C(O)=O)cc2)CC1)[C@H](O)C1CCCCC1 |r| Show InChI InChI=1S/C33H43N3O6/c1-2-3-19-36-30(38)28(29(37)24-7-5-4-6-8-24)34-32(41)33(36)17-20-35(21-18-33)22-23-9-13-26(14-10-23)42-27-15-11-25(12-16-27)31(39)40/h9-16,24,28-29,37H,2-8,17-22H2,1H3,(H,34,41)(H,39,40)/t28-,29-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Ono Pharmaceutical Co. Ltd
Curated by ChEMBL
| Assay Description
Displacement of MIP-1alpha from human CCR5 expressed in CHO cells |
Bioorg Med Chem 19: 4028-42 (2011)
Article DOI: 10.1016/j.bmc.2011.05.022
BindingDB Entry DOI: 10.7270/Q2H70G5Q |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 5
(Homo sapiens (Human)) | BDBM50190516
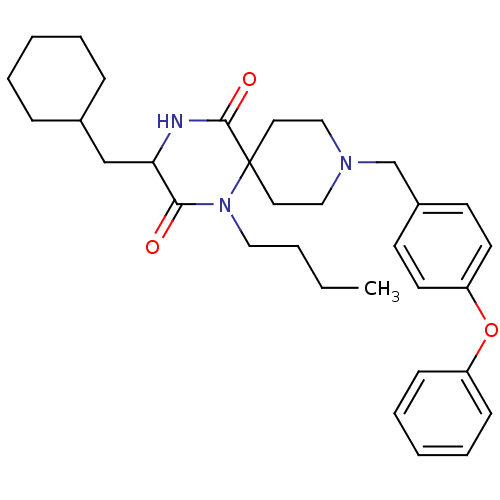
((RS)-1-butyl-3-(cyclohexylmethyl)-9-(4-phenoxybenz...)Show SMILES CCCCN1C(=O)C(CC2CCCCC2)NC(=O)C11CCN(Cc2ccc(Oc3ccccc3)cc2)CC1 Show InChI InChI=1S/C32H43N3O3/c1-2-3-20-35-30(36)29(23-25-10-6-4-7-11-25)33-31(37)32(35)18-21-34(22-19-32)24-26-14-16-28(17-15-26)38-27-12-8-5-9-13-27/h5,8-9,12-17,25,29H,2-4,6-7,10-11,18-24H2,1H3,(H,33,37) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Ono Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Displacement of [125I]MIP1-alpha from human CCR5 expressed in CHO cells |
Bioorg Med Chem Lett 17: 727-31 (2007)
Article DOI: 10.1016/j.bmcl.2006.10.084
BindingDB Entry DOI: 10.7270/Q2XK8GCM |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 5
(Homo sapiens (Human)) | BDBM50190516

((RS)-1-butyl-3-(cyclohexylmethyl)-9-(4-phenoxybenz...)Show SMILES CCCCN1C(=O)C(CC2CCCCC2)NC(=O)C11CCN(Cc2ccc(Oc3ccccc3)cc2)CC1 Show InChI InChI=1S/C32H43N3O3/c1-2-3-20-35-30(36)29(23-25-10-6-4-7-11-25)33-31(37)32(35)18-21-34(22-19-32)24-26-14-16-28(17-15-26)38-27-12-8-5-9-13-27/h5,8-9,12-17,25,29H,2-4,6-7,10-11,18-24H2,1H3,(H,33,37) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Ono Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of radio-isotope labeled MIP1-alpha binding to human recombinant CCR5 |
J Med Chem 49: 4140-52 (2006)
Article DOI: 10.1021/jm060051s
BindingDB Entry DOI: 10.7270/Q2ZG6T1S |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50530894
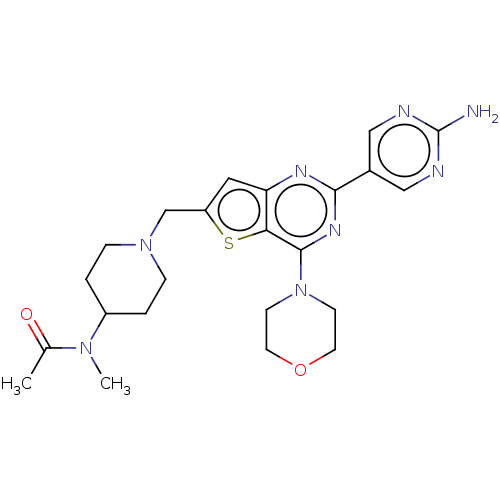
(CHEMBL4437468)Show SMILES CN(C1CCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cnc(N)nc2)CC1)C(C)=O Show InChI InChI=1S/C23H30N8O2S/c1-15(32)29(2)17-3-5-30(6-4-17)14-18-11-19-20(34-18)22(31-7-9-33-10-8-31)28-21(27-19)16-12-25-23(24)26-13-16/h11-13,17H,3-10,14H2,1-2H3,(H2,24,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6.30 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50530895
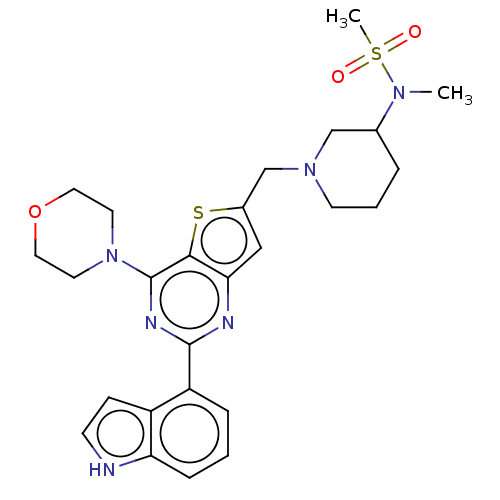
(CHEMBL4590897)Show SMILES CN(C1CCCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cccc3[nH]ccc23)C1)S(C)(=O)=O Show InChI InChI=1S/C26H32N6O3S2/c1-30(37(2,33)34)18-5-4-10-31(16-18)17-19-15-23-24(36-19)26(32-11-13-35-14-12-32)29-25(28-23)21-6-3-7-22-20(21)8-9-27-22/h3,6-9,15,18,27H,4-5,10-14,16-17H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6.30 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50530887
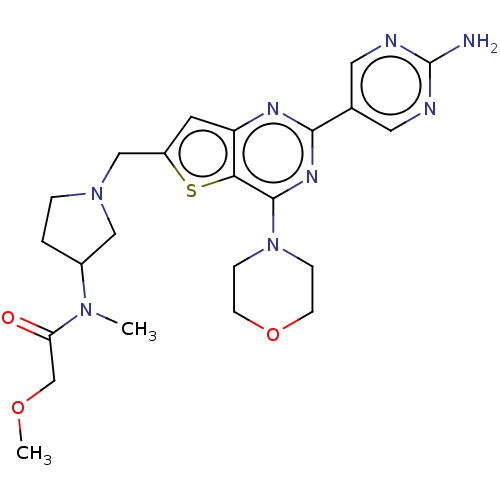
(CHEMBL4538734)Show SMILES COCC(=O)N(C)C1CCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cnc(N)nc2)C1 Show InChI InChI=1S/C23H30N8O3S/c1-29(19(32)14-33-2)16-3-4-30(12-16)13-17-9-18-20(35-17)22(31-5-7-34-8-6-31)28-21(27-18)15-10-25-23(24)26-11-15/h9-11,16H,3-8,12-14H2,1-2H3,(H2,24,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6.30 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50530877
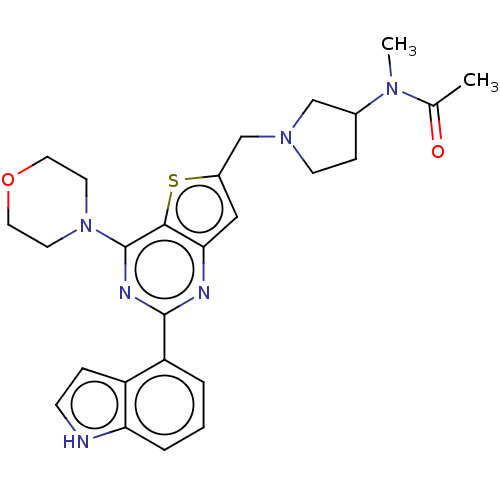
(CHEMBL4580111)Show SMILES CN(C1CCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cccc3[nH]ccc23)C1)C(C)=O Show InChI InChI=1S/C26H30N6O2S/c1-17(33)30(2)18-7-9-31(15-18)16-19-14-23-24(35-19)26(32-10-12-34-13-11-32)29-25(28-23)21-4-3-5-22-20(21)6-8-27-22/h3-6,8,14,18,27H,7,9-13,15-16H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6.30 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50530895

(CHEMBL4590897)Show SMILES CN(C1CCCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cccc3[nH]ccc23)C1)S(C)(=O)=O Show InChI InChI=1S/C26H32N6O3S2/c1-30(37(2,33)34)18-5-4-10-31(16-18)17-19-15-23-24(36-19)26(32-11-13-35-14-12-32)29-25(28-23)21-6-3-7-22-20(21)8-9-27-22/h3,6-9,15,18,27H,4-5,10-14,16-17H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6.30 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50530887

(CHEMBL4538734)Show SMILES COCC(=O)N(C)C1CCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cnc(N)nc2)C1 Show InChI InChI=1S/C23H30N8O3S/c1-29(19(32)14-33-2)16-3-4-30(12-16)13-17-9-18-20(35-17)22(31-5-7-34-8-6-31)28-21(27-18)15-10-25-23(24)26-11-15/h9-11,16H,3-8,12-14H2,1-2H3,(H2,24,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6.30 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50530877

(CHEMBL4580111)Show SMILES CN(C1CCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cccc3[nH]ccc23)C1)C(C)=O Show InChI InChI=1S/C26H30N6O2S/c1-17(33)30(2)18-7-9-31(15-18)16-19-14-23-24(35-19)26(32-10-12-34-13-11-32)29-25(28-23)21-4-3-5-22-20(21)6-8-27-22/h3-6,8,14,18,27H,7,9-13,15-16H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6.30 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50530928

(CHEMBL4534127)Show SMILES CN(C1CCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cnc(N)nc2)C1)S(C)(=O)=O Show InChI InChI=1S/C21H28N8O3S2/c1-27(34(2,30)31)15-3-4-28(12-15)13-16-9-17-18(33-16)20(29-5-7-32-8-6-29)26-19(25-17)14-10-23-21(22)24-11-14/h9-11,15H,3-8,12-13H2,1-2H3,(H2,22,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6.30 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50530894

(CHEMBL4437468)Show SMILES CN(C1CCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cnc(N)nc2)CC1)C(C)=O Show InChI InChI=1S/C23H30N8O2S/c1-15(32)29(2)17-3-5-30(6-4-17)14-18-11-19-20(34-18)22(31-7-9-33-10-8-31)28-21(27-19)16-12-25-23(24)26-13-16/h11-13,17H,3-10,14H2,1-2H3,(H2,24,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6.30 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50530928

(CHEMBL4534127)Show SMILES CN(C1CCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cnc(N)nc2)C1)S(C)(=O)=O Show InChI InChI=1S/C21H28N8O3S2/c1-27(34(2,30)31)15-3-4-28(12-15)13-16-9-17-18(33-16)20(29-5-7-32-8-6-29)26-19(25-17)14-10-23-21(22)24-11-14/h9-11,15H,3-8,12-13H2,1-2H3,(H2,22,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6.30 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 5
(Homo sapiens (Human)) | BDBM50198936

((R)-1-butyl-3-((R)-cyclohexyl-hydroxy-methyl)-9-(4...)Show SMILES CCCCN1C(=O)[C@H](NC(=O)C11CCN(Cc2ccc(Oc3ccccc3)cc2)CC1)[C@H](O)C1CCCCC1 Show InChI InChI=1S/C32H43N3O4/c1-2-3-20-35-30(37)28(29(36)25-10-6-4-7-11-25)33-31(38)32(35)18-21-34(22-19-32)23-24-14-16-27(17-15-24)39-26-12-8-5-9-13-26/h5,8-9,12-17,25,28-29,36H,2-4,6-7,10-11,18-23H2,1H3,(H,33,38)/t28-,29-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.76 | n/a | n/a | n/a | n/a | n/a | n/a |
Ono Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Antagonist activity at human CCR5 expressed in HOS cells assessed as inhibition of cell fusion with HIV gp120 expressing HEK293 cells by LTR lucifera... |
Bioorg Med Chem Lett 21: 1141-5 (2011)
Article DOI: 10.1016/j.bmcl.2010.12.109
BindingDB Entry DOI: 10.7270/Q24T6JP0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50530905

(CHEMBL4453879)Show SMILES CN(C1CCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cnc(N)nc2)CC1)S(C)(=O)=O Show InChI InChI=1S/C22H30N8O3S2/c1-28(35(2,31)32)16-3-5-29(6-4-16)14-17-11-18-19(34-17)21(30-7-9-33-10-8-30)27-20(26-18)15-12-24-22(23)25-13-15/h11-13,16H,3-10,14H2,1-2H3,(H2,23,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7.90 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50530887

(CHEMBL4538734)Show SMILES COCC(=O)N(C)C1CCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cnc(N)nc2)C1 Show InChI InChI=1S/C23H30N8O3S/c1-29(19(32)14-33-2)16-3-4-30(12-16)13-17-9-18-20(35-17)22(31-5-7-34-8-6-31)28-21(27-18)15-10-25-23(24)26-11-15/h9-11,16H,3-8,12-14H2,1-2H3,(H2,24,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7.90 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50530887

(CHEMBL4538734)Show SMILES COCC(=O)N(C)C1CCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cnc(N)nc2)C1 Show InChI InChI=1S/C23H30N8O3S/c1-29(19(32)14-33-2)16-3-4-30(12-16)13-17-9-18-20(35-17)22(31-5-7-34-8-6-31)28-21(27-18)15-10-25-23(24)26-11-15/h9-11,16H,3-8,12-14H2,1-2H3,(H2,24,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7.90 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50530872
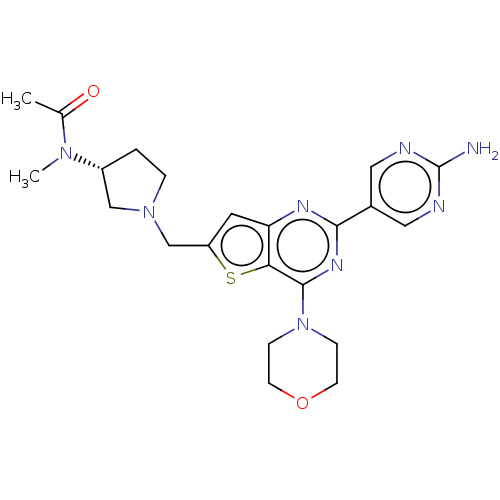
(CHEMBL4462986)Show SMILES CN([C@@H]1CCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cnc(N)nc2)C1)C(C)=O |r| Show InChI InChI=1S/C22H28N8O2S/c1-14(31)28(2)16-3-4-29(12-16)13-17-9-18-19(33-17)21(30-5-7-32-8-6-30)27-20(26-18)15-10-24-22(23)25-11-15/h9-11,16H,3-8,12-13H2,1-2H3,(H2,23,24,25)/t16-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7.90 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50530928

(CHEMBL4534127)Show SMILES CN(C1CCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cnc(N)nc2)C1)S(C)(=O)=O Show InChI InChI=1S/C21H28N8O3S2/c1-27(34(2,30)31)15-3-4-28(12-15)13-16-9-17-18(33-16)20(29-5-7-32-8-6-29)26-19(25-17)14-10-23-21(22)24-11-14/h9-11,15H,3-8,12-13H2,1-2H3,(H2,22,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7.90 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kalpha (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50530905

(CHEMBL4453879)Show SMILES CN(C1CCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cnc(N)nc2)CC1)S(C)(=O)=O Show InChI InChI=1S/C22H30N8O3S2/c1-28(35(2,31)32)16-3-5-29(6-4-16)14-17-11-18-19(34-17)21(30-7-9-33-10-8-30)27-20(26-18)15-12-24-22(23)25-13-15/h11-13,16H,3-10,14H2,1-2H3,(H2,23,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7.90 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50530928

(CHEMBL4534127)Show SMILES CN(C1CCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cnc(N)nc2)C1)S(C)(=O)=O Show InChI InChI=1S/C21H28N8O3S2/c1-27(34(2,30)31)15-3-4-28(12-15)13-16-9-17-18(33-16)20(29-5-7-32-8-6-29)26-19(25-17)14-10-23-21(22)24-11-14/h9-11,15H,3-8,12-13H2,1-2H3,(H2,22,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7.90 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kalpha (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50530872

(CHEMBL4462986)Show SMILES CN([C@@H]1CCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cnc(N)nc2)C1)C(C)=O |r| Show InChI InChI=1S/C22H28N8O2S/c1-14(31)28(2)16-3-4-29(12-16)13-17-9-18-19(33-17)21(30-5-7-32-8-6-30)27-20(26-18)15-10-24-22(23)25-11-15/h9-11,16H,3-8,12-13H2,1-2H3,(H2,23,24,25)/t16-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7.90 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 5
(Homo sapiens (Human)) | BDBM50190520
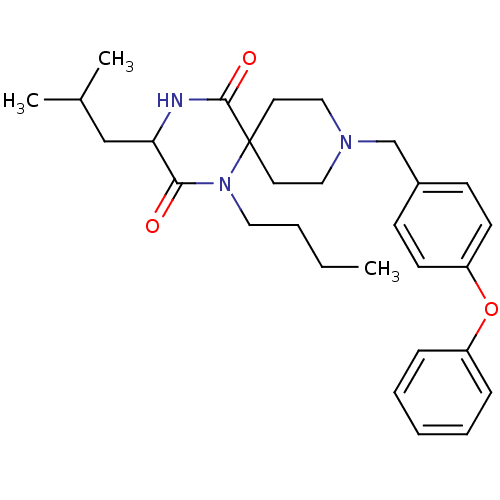
((RS)-1-butyl-3-isobutyl-9-(4-phenoxybenzyl)-1,4,9-...)Show SMILES CCCCN1C(=O)C(CC(C)C)NC(=O)C11CCN(Cc2ccc(Oc3ccccc3)cc2)CC1 Show InChI InChI=1S/C29H39N3O3/c1-4-5-17-32-27(33)26(20-22(2)3)30-28(34)29(32)15-18-31(19-16-29)21-23-11-13-25(14-12-23)35-24-9-7-6-8-10-24/h6-14,22,26H,4-5,15-21H2,1-3H3,(H,30,34) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Ono Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Displacement of [125I]MIP1-alpha from human CCR5 expressed in CHO cells |
Bioorg Med Chem Lett 17: 727-31 (2007)
Article DOI: 10.1016/j.bmcl.2006.10.084
BindingDB Entry DOI: 10.7270/Q2XK8GCM |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50403068
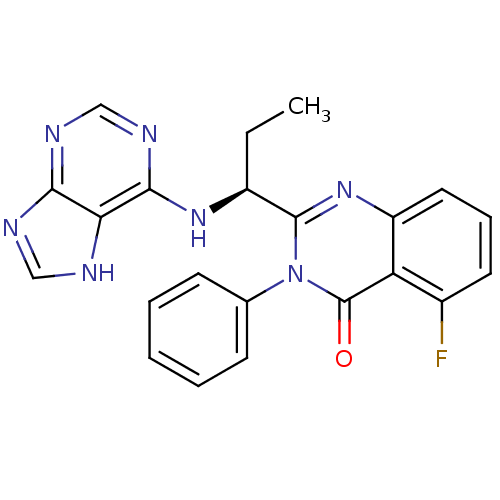
(CHEMBL2216870 | IDELALISIB | US9745321, CAL-101)Show SMILES CC[C@H](Nc1ncnc2nc[nH]c12)c1nc2cccc(F)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C22H18FN7O/c1-2-15(28-20-18-19(25-11-24-18)26-12-27-20)21-29-16-10-6-9-14(23)17(16)22(31)30(21)13-7-4-3-5-8-13/h3-12,15H,2H2,1H3,(H2,24,25,26,27,28)/t15-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50530893

(CHEMBL4532070)Show SMILES CN(C1CCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cccc3[nH]ccc23)C1)S(C)(=O)=O Show InChI InChI=1S/C25H30N6O3S2/c1-29(36(2,32)33)17-7-9-30(15-17)16-18-14-22-23(35-18)25(31-10-12-34-13-11-31)28-24(27-22)20-4-3-5-21-19(20)6-8-26-21/h3-6,8,14,17,26H,7,9-13,15-16H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50530893

(CHEMBL4532070)Show SMILES CN(C1CCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cccc3[nH]ccc23)C1)S(C)(=O)=O Show InChI InChI=1S/C25H30N6O3S2/c1-29(36(2,32)33)17-7-9-30(15-17)16-18-14-22-23(35-18)25(31-10-12-34-13-11-31)28-24(27-22)20-4-3-5-21-19(20)6-8-26-21/h3-6,8,14,17,26H,7,9-13,15-16H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
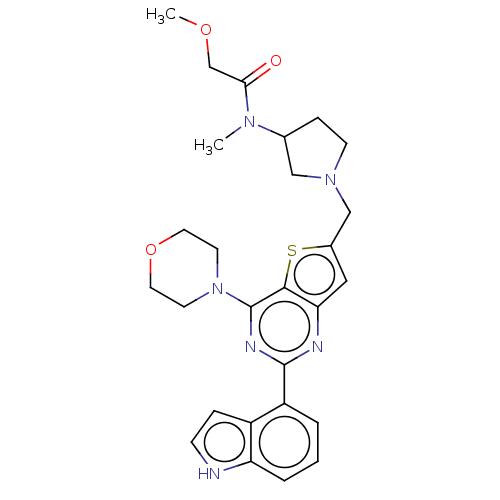
(Homo sapiens (Human)) | BDBM50530896
(CHEMBL4589808)Show SMILES COCC(=O)N(C)C1CCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cccc3[nH]ccc23)C1 Show InChI InChI=1S/C27H32N6O3S/c1-31(24(34)17-35-2)18-7-9-32(15-18)16-19-14-23-25(37-19)27(33-10-12-36-13-11-33)30-26(29-23)21-4-3-5-22-20(21)6-8-28-22/h3-6,8,14,18,28H,7,9-13,15-17H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50530878
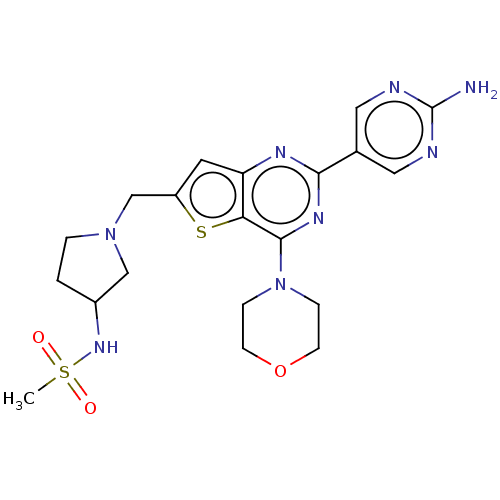
(CHEMBL4467891)Show SMILES CS(=O)(=O)NC1CCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cnc(N)nc2)C1 Show InChI InChI=1S/C20H26N8O3S2/c1-33(29,30)26-14-2-3-27(11-14)12-15-8-16-17(32-15)19(28-4-6-31-7-5-28)25-18(24-16)13-9-22-20(21)23-10-13/h8-10,14,26H,2-7,11-12H2,1H3,(H2,21,22,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kalpha (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50403068

(CHEMBL2216870 | IDELALISIB | US9745321, CAL-101)Show SMILES CC[C@H](Nc1ncnc2nc[nH]c12)c1nc2cccc(F)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C22H18FN7O/c1-2-15(28-20-18-19(25-11-24-18)26-12-27-20)21-29-16-10-6-9-14(23)17(16)22(31)30(21)13-7-4-3-5-8-13/h3-12,15H,2H2,1H3,(H2,24,25,26,27,28)/t15-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50530878

(CHEMBL4467891)Show SMILES CS(=O)(=O)NC1CCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cnc(N)nc2)C1 Show InChI InChI=1S/C20H26N8O3S2/c1-33(29,30)26-14-2-3-27(11-14)12-15-8-16-17(32-15)19(28-4-6-31-7-5-28)25-18(24-16)13-9-22-20(21)23-10-13/h8-10,14,26H,2-7,11-12H2,1H3,(H2,21,22,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kalpha (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50530896

(CHEMBL4589808)Show SMILES COCC(=O)N(C)C1CCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cccc3[nH]ccc23)C1 Show InChI InChI=1S/C27H32N6O3S/c1-31(24(34)17-35-2)18-7-9-32(15-18)16-19-14-23-25(37-19)27(33-10-12-36-13-11-33)30-26(29-23)21-4-3-5-22-20(21)6-8-28-22/h3-6,8,14,18,28H,7,9-13,15-17H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data