

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Neuraminidase (Influenza A virus (A/Puerto Rico/8/34/Mount Sinai(...) | BDBM50040516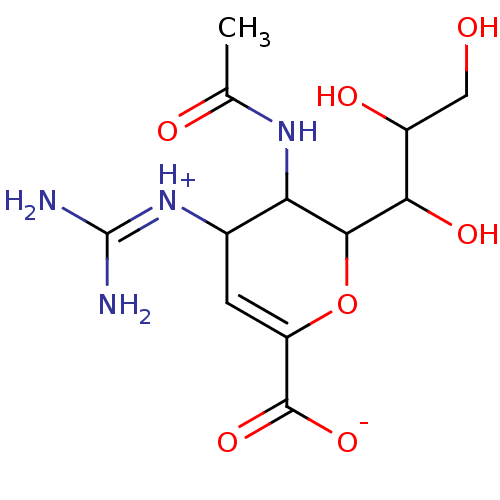 (3-(acetylamino)-4-{[amino(iminio)methyl]amino}-2-[...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | PubMed | 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Monash University Curated by ChEMBL | Assay Description inhibition of Influenza A Sialidase | J Med Chem 37: 616-24 (1994) Checked by Author BindingDB Entry DOI: 10.7270/Q27P901J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470598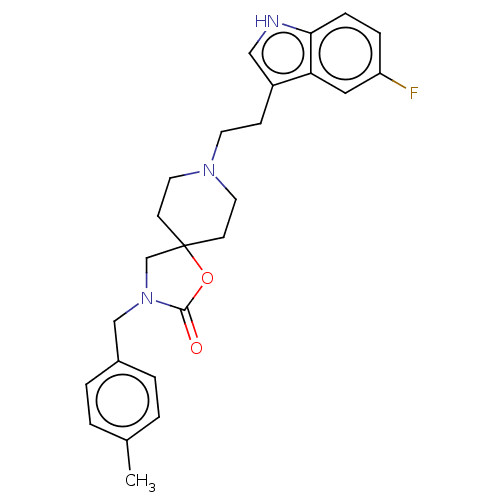 (CHEMBL126050) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470591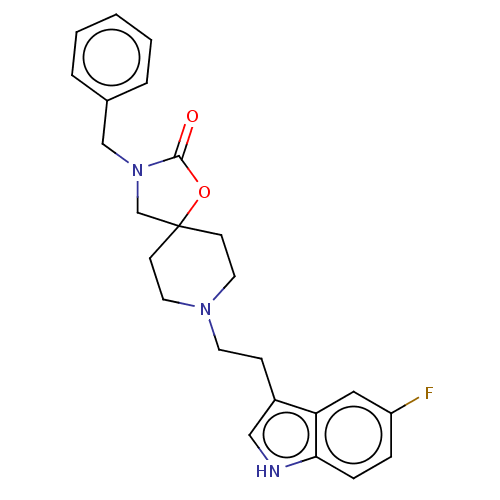 (CHEMBL124208) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470604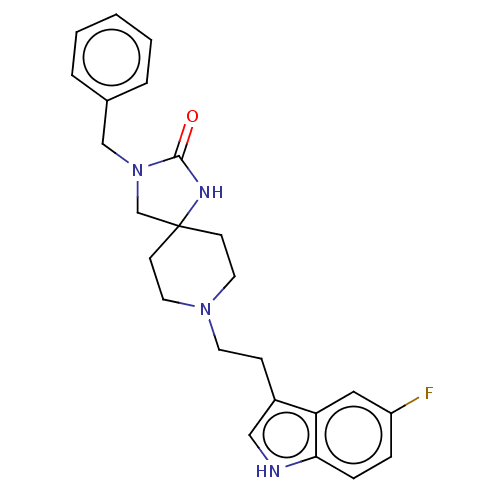 (CHEMBL338825) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470590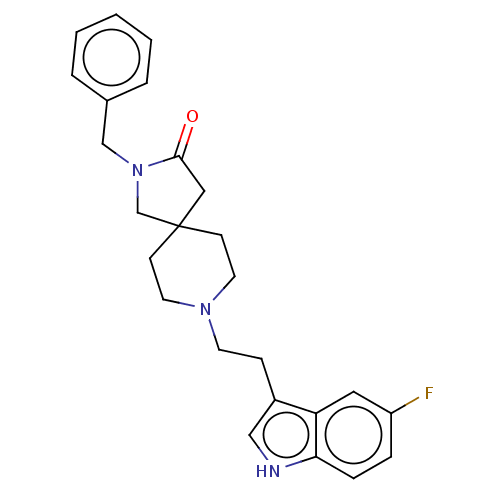 (CHEMBL341357) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470583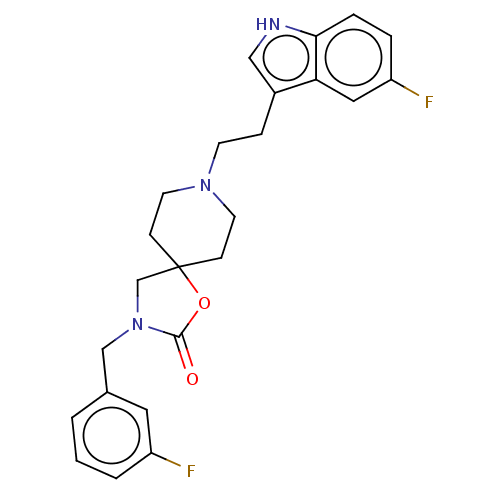 (CHEMBL125696) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470596 (CHEMBL125310) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470587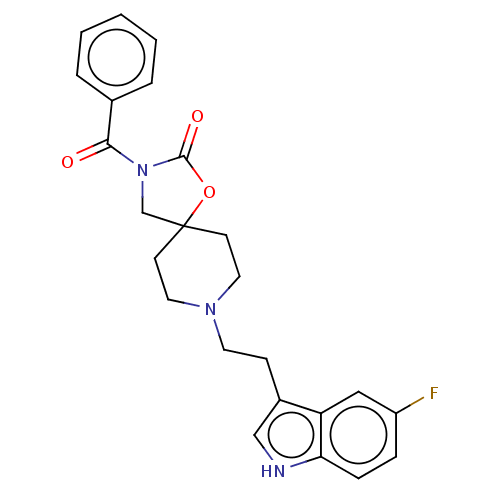 (CHEMBL124648) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (GUINEA PIG) | BDBM50470591 (CHEMBL124208) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description The compound was tested for binding affinity against Tachykinin receptor 2 from guinea pig trachea | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470603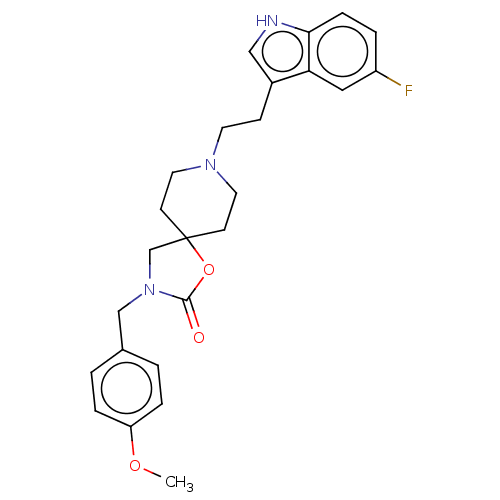 (CHEMBL124013) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470605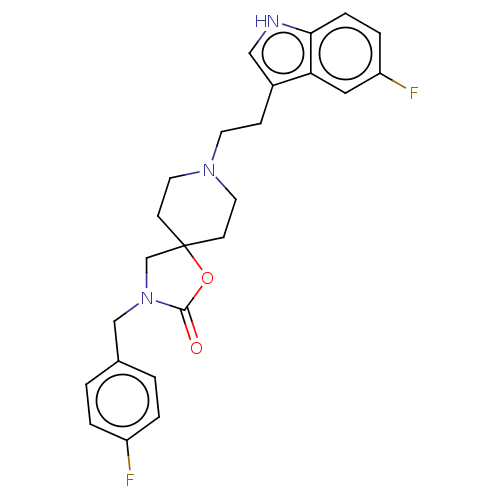 (CHEMBL122169) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470582 (CHEMBL126043) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470595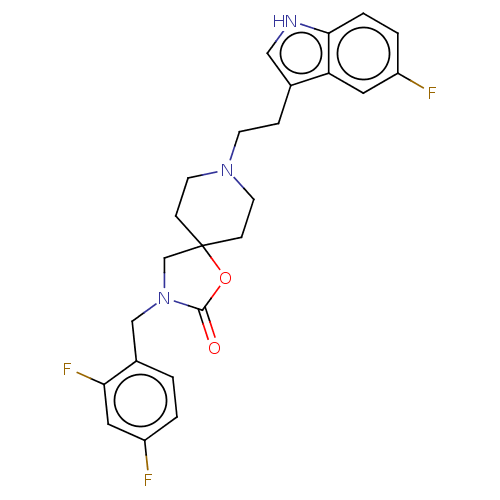 (CHEMBL122019) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 6.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470599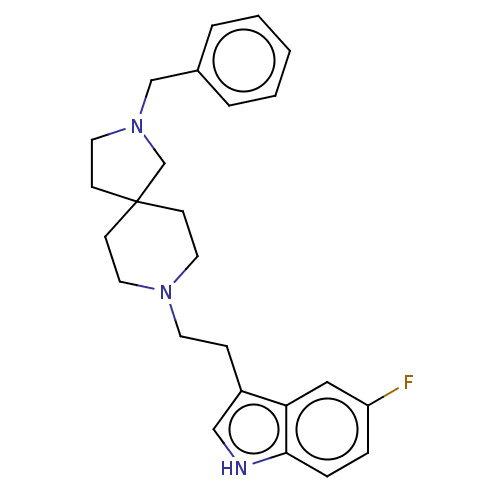 (CHEMBL330766) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 6.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Homo sapiens (Human)) | BDBM50470591 (CHEMBL124208) | PDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 7.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description The compound was tested for binding affinity against Tachykinin receptor 2 from human expressed in CHO cells | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470592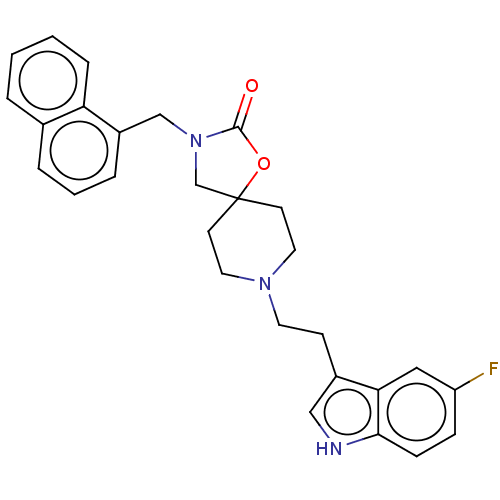 (CHEMBL339311) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 7.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470597 (CHEMBL330826) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470606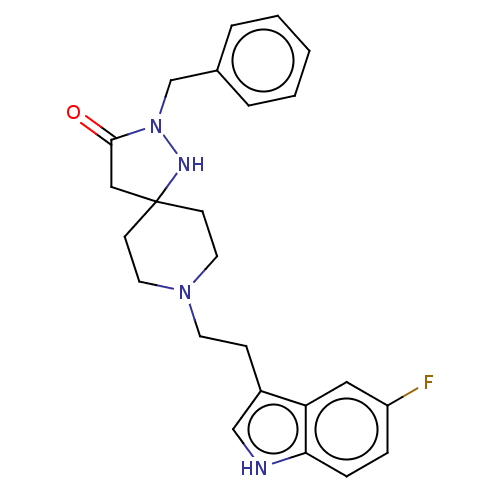 (CHEMBL446099) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470581 (CHEMBL341008) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470584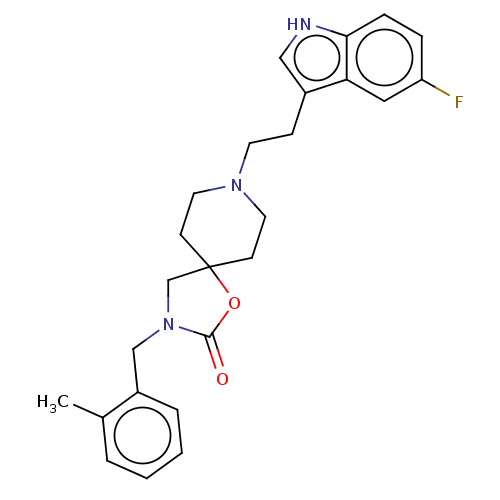 (CHEMBL125001) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470585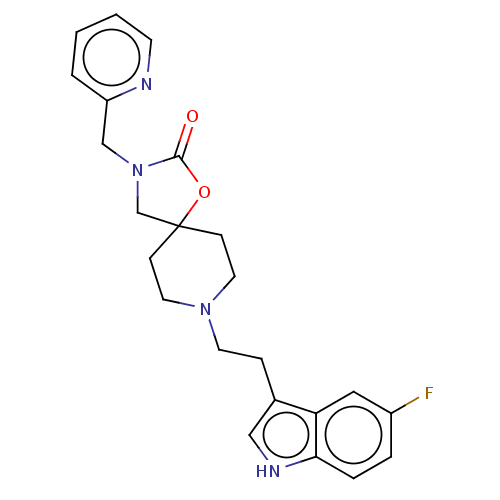 (CHEMBL415518) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470608 (CHEMBL338030) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470607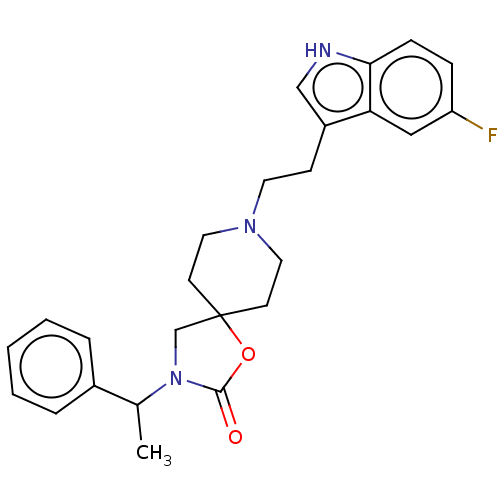 (CHEMBL124624) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470601 (CHEMBL123731) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuraminidase (Influenza A virus (A/Puerto Rico/8/34/Mount Sinai(...) | BDBM50040515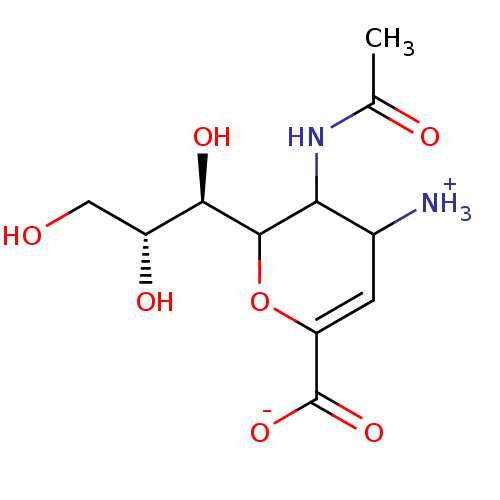 (3-(acetylamino)-4-ammonio-2-[(1R,2R)-1,2,3-trihydr...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | PubMed | 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Monash University Curated by ChEMBL | Assay Description inhibition of Influenza A Sialidase | J Med Chem 37: 616-24 (1994) Checked by Author BindingDB Entry DOI: 10.7270/Q27P901J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470589 (CHEMBL331011) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 79 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470602 (CHEMBL340747) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470594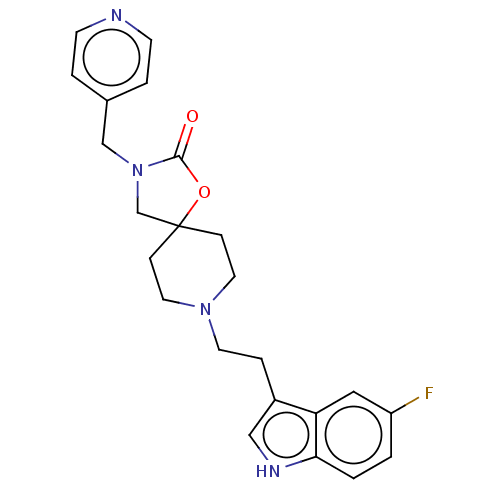 (CHEMBL340405) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470600 (CHEMBL435619) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470586 (CHEMBL435610) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 126 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470593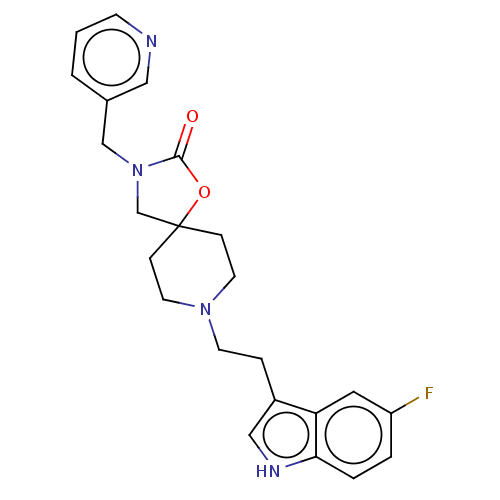 (CHEMBL126158) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-K receptor (Rattus norvegicus (Rat)) | BDBM50470588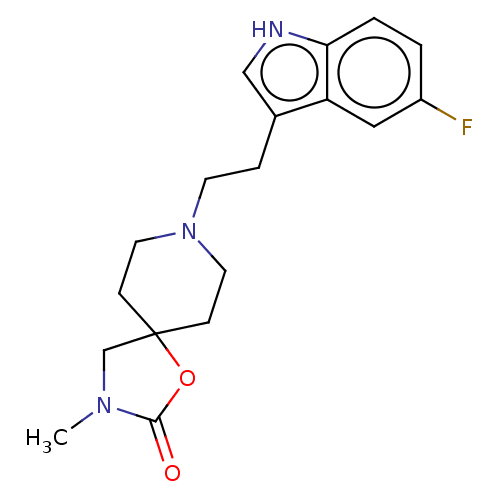 (CHEMBL340548) | PDB Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 1.59E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description Binding affinity against tachykinin receptor 2 from rat colon. | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuraminidase (Influenza A virus (A/Puerto Rico/8/34/Mount Sinai(...) | BDBM50032861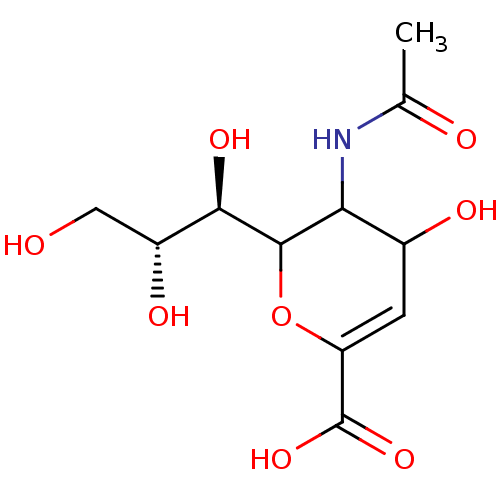 (5-((R)-Acetylamino)-4-hydroxy-6-((1R,2R)-1,2,3-tri...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | PubMed | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Monash University Curated by ChEMBL | Assay Description inhibition of Influenza A Sialidase | J Med Chem 37: 616-24 (1994) Checked by Author BindingDB Entry DOI: 10.7270/Q27P901J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-P receptor (Homo sapiens (Human)) | BDBM50470591 (CHEMBL124208) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 7.94E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description The compound was tested for binding affinity against Tachykinin receptor 1 from rabbit cortex | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Substance-P receptor (GUINEA PIG) | BDBM50470591 (CHEMBL124208) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | <1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description The compound was tested for binding affinity against Tachykinin receptor 1 from guinea pig trachea | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuromedin-K receptor (GUINEA PIG) | BDBM50470591 (CHEMBL124208) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 7.94E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Medicines Research Centre Curated by ChEMBL | Assay Description The compound was tested for binding affinity against Tachykinin receptor 3 from guinea pig cerebral cortex | J Med Chem 38: 3772-9 (1995) Article DOI: 10.1021/jm00019a006 BindingDB Entry DOI: 10.7270/Q2JM2DCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuraminidase (Influenza A virus (A/Puerto Rico/8/34/Mount Sinai(...) | BDBM4945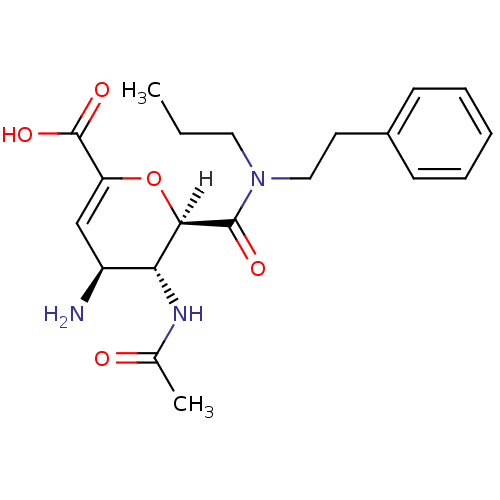 ((2R,3R,4S)-4-amino-3-acetamido-2-[(2-phenylethyl)(...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Limited Curated by ChEMBL | Assay Description Compound was tested for inhibitory concentration against Influenza sialidase type A | J Med Chem 41: 798-807 (1998) Article DOI: 10.1021/jm9703754 BindingDB Entry DOI: 10.7270/Q2M32WFK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuraminidase (Influenza A virus (A/Puerto Rico/8/34/Mount Sinai(...) | BDBM4945 ((2R,3R,4S)-4-amino-3-acetamido-2-[(2-phenylethyl)(...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article | n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory activity against influenza A virus sialidase | Bioorg Med Chem Lett 7: 2239-2242 (1997) Article DOI: 10.1016/S0960-894X(97)00399-5 BindingDB Entry DOI: 10.7270/Q2RX9CKB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuraminidase (Influenza A virus (A/Puerto Rico/8/34/Mount Sinai(...) | BDBM50330326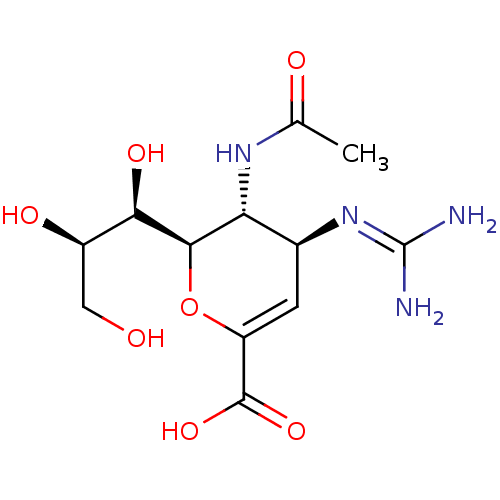 ((4S,5R,6R)-5-Acetylamino-4-guanidino-6-((1R,3R)-1,...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article | n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory activity against influenza A virus sialidase | Bioorg Med Chem Lett 7: 2239-2242 (1997) Article DOI: 10.1016/S0960-894X(97)00399-5 BindingDB Entry DOI: 10.7270/Q2RX9CKB | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Neuraminidase (Influenza B virus (B/Lee/40)) | BDBM50330326 ((4S,5R,6R)-5-Acetylamino-4-guanidino-6-((1R,3R)-1,...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Limited Curated by ChEMBL | Assay Description Compound was tested for inhibitory concentration against Influenza sialidase type B | J Med Chem 41: 798-807 (1998) Article DOI: 10.1021/jm9703754 BindingDB Entry DOI: 10.7270/Q2M32WFK | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Neuraminidase (Influenza A virus (A/Puerto Rico/8/34/Mount Sinai(...) | BDBM50330326 ((4S,5R,6R)-5-Acetylamino-4-guanidino-6-((1R,3R)-1,...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Limited Curated by ChEMBL | Assay Description Compound was tested for inhibitory concentration against Influenza sialidase type A | J Med Chem 41: 798-807 (1998) Article DOI: 10.1021/jm9703754 BindingDB Entry DOI: 10.7270/Q2M32WFK | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Neuraminidase (Influenza A virus (A/Puerto Rico/8/34/Mount Sinai(...) | BDBM4952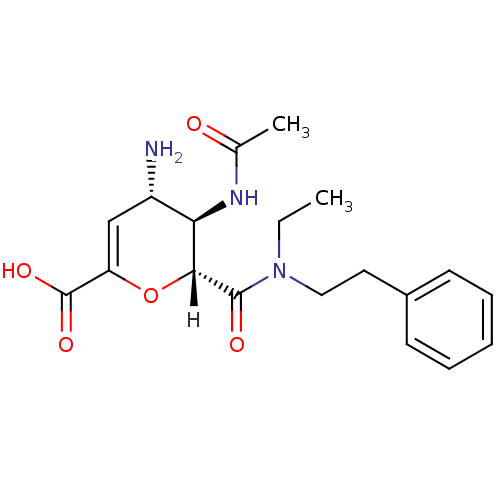 ((2R,3R,4S)-4-amino-3-acetamido-2-[ethyl(2-phenylet...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Limited Curated by ChEMBL | Assay Description Compound was tested for inhibitory concentration against Influenza sialidase type A | J Med Chem 41: 798-807 (1998) Article DOI: 10.1021/jm9703754 BindingDB Entry DOI: 10.7270/Q2M32WFK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuraminidase (Influenza A virus (A/Puerto Rico/8/34/Mount Sinai(...) | BDBM4958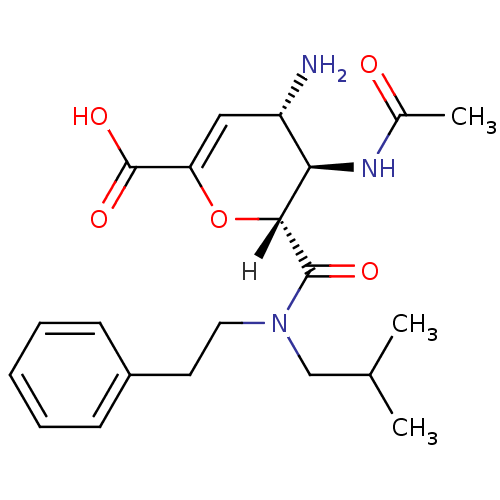 ((2R,3R,4S)-4-amino-3-acetamido-2-[(2-methylpropyl)...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Limited Curated by ChEMBL | Assay Description Compound was tested for inhibitory concentration against Influenza sialidase type A | J Med Chem 41: 798-807 (1998) Article DOI: 10.1021/jm9703754 BindingDB Entry DOI: 10.7270/Q2M32WFK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuraminidase (Influenza B virus (B/Lee/40)) | BDBM50330326 ((4S,5R,6R)-5-Acetylamino-4-guanidino-6-((1R,3R)-1,...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article | n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory activity against influenza B virus sialidase | Bioorg Med Chem Lett 7: 2239-2242 (1997) Article DOI: 10.1016/S0960-894X(97)00399-5 BindingDB Entry DOI: 10.7270/Q2RX9CKB | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Neuraminidase (Influenza A virus (A/Puerto Rico/8/34/Mount Sinai(...) | BDBM50290063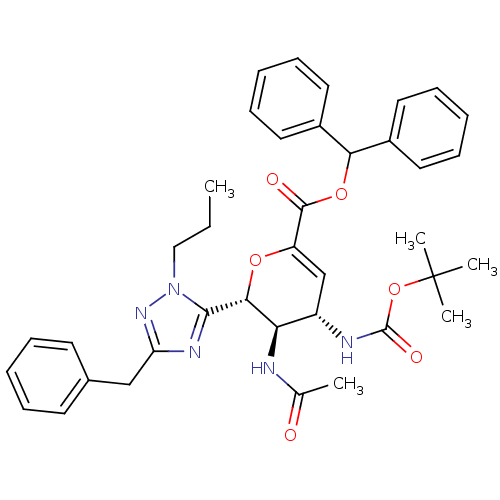 ((4S,5R,6R)-5-Acetylamino-6-(5-benzyl-2-propyl-2H-[...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article | n/a | n/a | 70 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory activity against influenza A virus sialidase | Bioorg Med Chem Lett 7: 2239-2242 (1997) Article DOI: 10.1016/S0960-894X(97)00399-5 BindingDB Entry DOI: 10.7270/Q2RX9CKB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuraminidase (Influenza A virus (A/Puerto Rico/8/34/Mount Sinai(...) | BDBM4951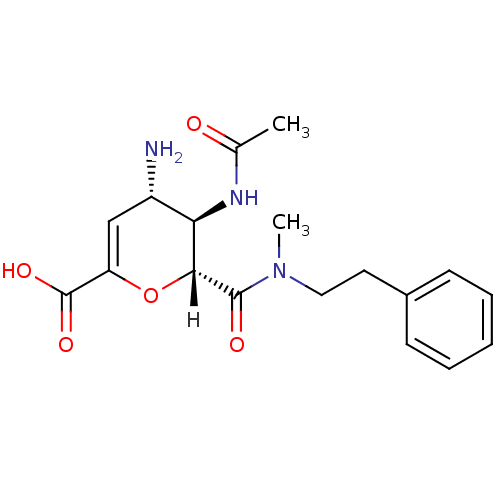 ((2R,3R,4S)-4-amino-3-acetamido-2-[methyl(2-phenyle...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 320 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Limited Curated by ChEMBL | Assay Description Compound was tested for inhibitory concentration against Influenza sialidase type A | J Med Chem 41: 798-807 (1998) Article DOI: 10.1021/jm9703754 BindingDB Entry DOI: 10.7270/Q2M32WFK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuraminidase (Influenza A virus (A/Puerto Rico/8/34/Mount Sinai(...) | BDBM50063303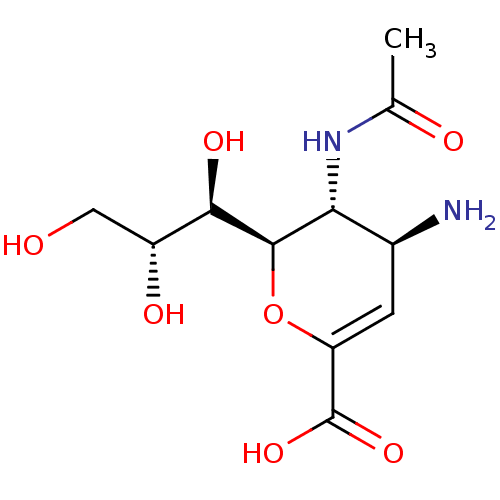 ((2R,3R,4S)-3-acetamido-4-amino-2-((1R,2R)-1,2,3-tr...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 320 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Limited Curated by ChEMBL | Assay Description Compound was tested for inhibitory concentration against Influenza sialidase type A | J Med Chem 41: 798-807 (1998) Article DOI: 10.1021/jm9703754 BindingDB Entry DOI: 10.7270/Q2M32WFK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuraminidase (Influenza A virus (A/Puerto Rico/8/34/Mount Sinai(...) | BDBM50290066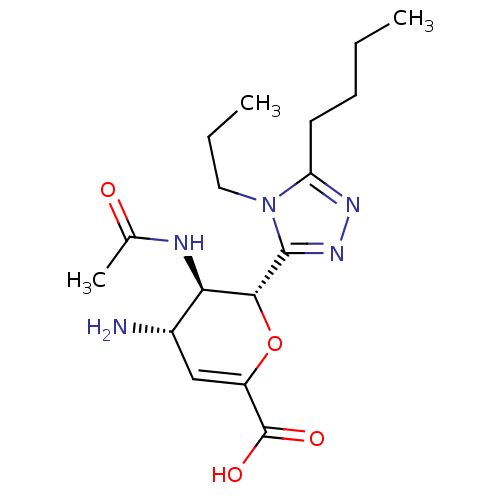 ((4S,5R,6R)-5-Acetylamino-4-amino-6-(5-butyl-4-prop...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article | n/a | n/a | 330 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory activity against influenza A virus sialidase | Bioorg Med Chem Lett 7: 2239-2242 (1997) Article DOI: 10.1016/S0960-894X(97)00399-5 BindingDB Entry DOI: 10.7270/Q2RX9CKB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuraminidase (Influenza B virus (B/Lee/40)) | BDBM50063303 ((2R,3R,4S)-3-acetamido-4-amino-2-((1R,2R)-1,2,3-tr...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 410 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Limited Curated by ChEMBL | Assay Description Compound was tested for inhibitory concentration against Influenza sialidase type B | J Med Chem 41: 798-807 (1998) Article DOI: 10.1021/jm9703754 BindingDB Entry DOI: 10.7270/Q2M32WFK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuraminidase (Influenza B virus (B/Lee/40)) | BDBM4945 ((2R,3R,4S)-4-amino-3-acetamido-2-[(2-phenylethyl)(...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB Article PubMed | n/a | n/a | 3.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Limited Curated by ChEMBL | Assay Description Compound was tested for inhibitory concentration against Influenza sialidase type B | J Med Chem 41: 798-807 (1998) Article DOI: 10.1021/jm9703754 BindingDB Entry DOI: 10.7270/Q2M32WFK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 76 total ) | Next | Last >> |