| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Neuraminidase |
|---|
| Ligand | BDBM50063303 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_200809 (CHEMBL809158) |
|---|
| IC50 | 410±n/a nM |
|---|
| Citation |  Taylor, NR; Cleasby, A; Singh, O; Skarzynski, T; Wonacott, AJ; Smith, PW; Sollis, SL; Howes, PD; Cherry, PC; Bethell, R; Colman, P; Varghese, J Dihydropyrancarboxamides related to zanamivir: a new series of inhibitors of influenza virus sialidases. 2. Crystallographic and molecular modeling study of complexes of 4-amino-4H-pyran-6-carboxamides and sialidase from influenza virus types A and B. J Med Chem41:798-807 (1998) [PubMed] Article Taylor, NR; Cleasby, A; Singh, O; Skarzynski, T; Wonacott, AJ; Smith, PW; Sollis, SL; Howes, PD; Cherry, PC; Bethell, R; Colman, P; Varghese, J Dihydropyrancarboxamides related to zanamivir: a new series of inhibitors of influenza virus sialidases. 2. Crystallographic and molecular modeling study of complexes of 4-amino-4H-pyran-6-carboxamides and sialidase from influenza virus types A and B. J Med Chem41:798-807 (1998) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Neuraminidase |
|---|
| Name: | Neuraminidase |
|---|
| Synonyms: | Influenza B Virus Neuraminidase | NA | NRAM_INBLE | Neuraminidase | Neuraminidase B |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 51446.67 |
|---|
| Organism: | Influenza B virus (B/Lee/40) |
|---|
| Description: | n/a |
|---|
| Residue: | 466 |
|---|
| Sequence: | MLPSTVQTLTLLLTSGGVLLSLYVSASLSYLLYSDVLLKFSSTKTTAPTMSLECTNASNA
QTVNHSATKEMTFPPPEPEWTYPRLSCQGSTFQKALLISPHRFGEIKGNSAPLIIREPFV
ACGPKECRHFALTHYAAQPGGYYNGTRKDRNKLRHLVSVKLGKIPTVENSIFHMAAWSGS
ACHDGREWTYIGVDGPDNDALVKIKYGEAYTDTYHSYAHNILRTQESACNCIGGDCYLMI
TDGSASGISKCRFLKIREGRIIKEILPTGRVEHTEECTCGFASNKTIECACRDNSYTAKR
PFVKLNVETDTAEIRLMCTKTYLDTPRPDDGSIAGPCESNGDKWLGGIKGGFVHQRMASK
IGRWYSRTMSKTNRMGMELYVKYDGDPWTDSDALTLSGVMVSIEEPGWYSFGFEIKDKKC
DVPCIGIEMVHDGGKDTWHSAATAIYCLMGSGQLLWDTVTGVDMAL
|
|
|
|---|
| BDBM50063303 |
|---|
| n/a |
|---|
| Name | BDBM50063303 |
|---|
| Synonyms: | (2R,3R,4S)-3-acetamido-4-amino-2-((1R,2R)-1,2,3-trihydroxypropyl)-3,4-dihydro-2H-pyran-6-carboxylic acid | (4S,5R,6R)-5-Acetylamino-4-amino-6-((1R,2R)-1,2,3-trihydroxy-propyl)-5,6-dihydro-4H-pyran-2-carboxylic acid | (4S,5R,6R)-5-acetamido-4-amino-6-((1R,2R)-1,2,3-trihydroxypropyl)-5,6-dihydro-4H-pyran-2-carboxylic acid | 4-AMINO-2-DEOXY-2,3-DEHYDRO-N-NEURAMINIC ACID | CHEMBL52270 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C11H18N2O7 |
|---|
| Mol. Mass. | 290.2698 |
|---|
| SMILES | CC(=O)N[C@@H]1[C@@H](N)C=C(O[C@H]1[C@H](O)[C@H](O)CO)C(O)=O |r,c:7| |
|---|
| Structure | 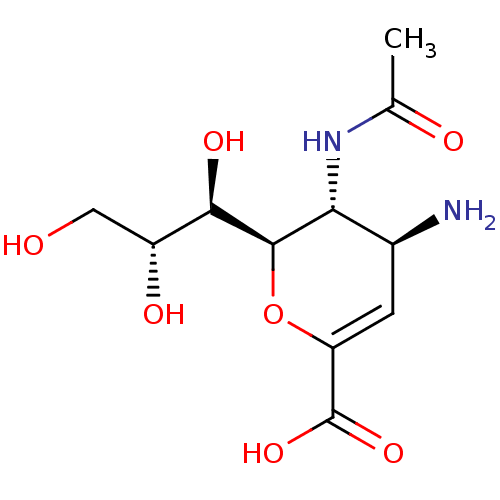
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Taylor, NR; Cleasby, A; Singh, O; Skarzynski, T; Wonacott, AJ; Smith, PW; Sollis, SL; Howes, PD; Cherry, PC; Bethell, R; Colman, P; Varghese, J Dihydropyrancarboxamides related to zanamivir: a new series of inhibitors of influenza virus sialidases. 2. Crystallographic and molecular modeling study of complexes of 4-amino-4H-pyran-6-carboxamides and sialidase from influenza virus types A and B. J Med Chem41:798-807 (1998) [PubMed] Article
Taylor, NR; Cleasby, A; Singh, O; Skarzynski, T; Wonacott, AJ; Smith, PW; Sollis, SL; Howes, PD; Cherry, PC; Bethell, R; Colman, P; Varghese, J Dihydropyrancarboxamides related to zanamivir: a new series of inhibitors of influenza virus sialidases. 2. Crystallographic and molecular modeling study of complexes of 4-amino-4H-pyran-6-carboxamides and sialidase from influenza virus types A and B. J Med Chem41:798-807 (1998) [PubMed] Article