 Found 9670 hits with Last Name = 'zhou' and Initial = 'j'
Found 9670 hits with Last Name = 'zhou' and Initial = 'j' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
5-hydroxytryptamine receptor 1B
(GUINEA PIG) | BDBM50590649
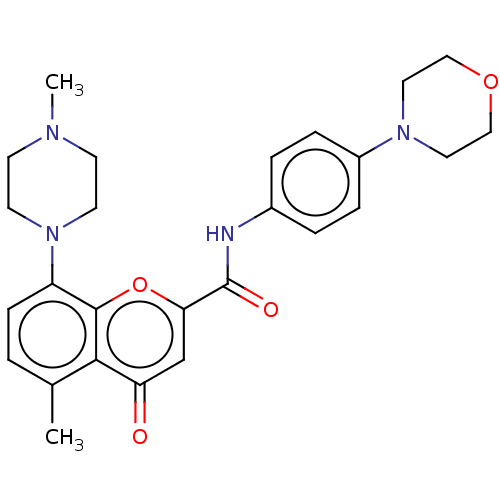
(CHEMBL5209320)Show SMILES Cc1ccc(N2CCN([11CH3])CC2)c2oc(cc(=O)c12)C(=O)Nc1ccc(cc1)N1CCOCC1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00633
BindingDB Entry DOI: 10.7270/Q26M3BS8 |
More data for this
Ligand-Target Pair | |
Non-receptor tyrosine-protein kinase TYK2
(Homo sapiens (Human)) | BDBM50507816
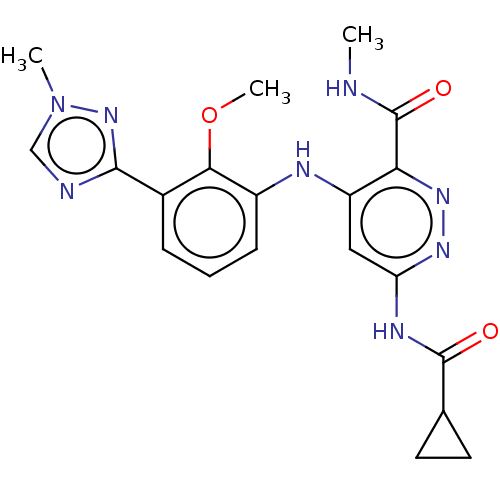
(Bms-986165 | Deucravacitinib)Show SMILES [2H]C([2H])([2H])NC(=O)c1nnc(NC(=O)C2CC2)cc1Nc1cccc(-c2ncn(C)n2)c1OC Show InChI InChI=1S/C20H22N8O3/c1-21-20(30)16-14(9-15(25-26-16)24-19(29)11-7-8-11)23-13-6-4-5-12(17(13)31-3)18-22-10-28(2)27-18/h4-6,9-11H,7-8H2,1-3H3,(H,21,30)(H2,23,24,25,29) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of TYK2 in human Jurkat cells assessed as reduction in IFN-alpha stimulated TYK2 phosphorylation by caspase3/7 reagent-based Western blot ... |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50576898
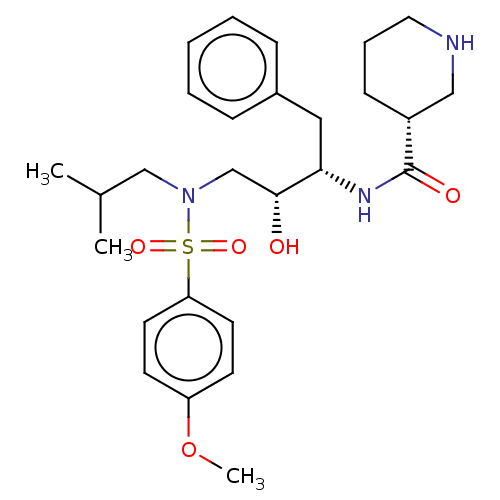
(CHEMBL4877646)Show SMILES COc1ccc(cc1)S(=O)(=O)N(CC(C)C)C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)[C@@H]1CCCNC1 |r| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of wild type HIV1 protease using Arg-Glu (EDANS)-Ser-Gln-Asn-Tyr-Pro-Ile-Val-Gln-Lys(DABCYL)-Arg as substrate preincubated for 20 to 30 mi... |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113450
BindingDB Entry DOI: 10.7270/Q2571GTH |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50590640

(CHEMBL5177580)Show SMILES COc1ccccc1N1CCN(CCCCc2ccc3n(C)c(=O)oc3c2)CC1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00633
BindingDB Entry DOI: 10.7270/Q26M3BS8 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50472582

(CHEMBL45422)Show SMILES CNc1nc(CNCC2(F)CCN(CC2)C(=O)c2ccc(F)c(Cl)c2)ccc1C Show InChI InChI=1S/C21H25ClF2N4O/c1-14-3-5-16(27-19(14)25-2)12-26-13-21(24)7-9-28(10-8-21)20(29)15-4-6-18(23)17(22)11-15/h3-6,11,26H,7-10,12-13H2,1-2H3,(H,25,27) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00633
BindingDB Entry DOI: 10.7270/Q26M3BS8 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50576910

(CHEMBL4862758)Show SMILES COc1ccc(cc1)S(=O)(=O)N(CC(C)C)C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)[C@@H]1CCCN(C)C1 |r| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of wild type HIV1 protease using Arg-Glu (EDANS)-Ser-Gln-Asn-Tyr-Pro-Ile-Val-Gln-Lys(DABCYL)-Arg as substrate preincubated for 20 to 30 mi... |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113450
BindingDB Entry DOI: 10.7270/Q2571GTH |
More data for this
Ligand-Target Pair | |
Beta-lactamase
(Escherichia coli) | BDBM50274047

(CHEMBL3613796)Show SMILES OB(O)CNS(=O)(=O)c1ccc(cc1C(F)(F)F)-c1nnn[nH]1 Show InChI InChI=1S/C9H9BF3N5O4S/c11-9(12,13)6-3-5(8-15-17-18-16-8)1-2-7(6)23(21,22)14-4-10(19)20/h1-3,14,19-20H,4H2,(H,15,16,17,18) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UCL School of Pharmacy
Curated by ChEMBL
| Assay Description
Inhibition of Escherichia coli AmpC using CENTA as substrate by spectrometry based Lineweaver-Burk plot analysis |
Bioorg Med Chem 26: 2921-2927 (2018)
Article DOI: 10.1016/j.bmc.2018.04.055
BindingDB Entry DOI: 10.7270/Q25T3P0V |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50541871
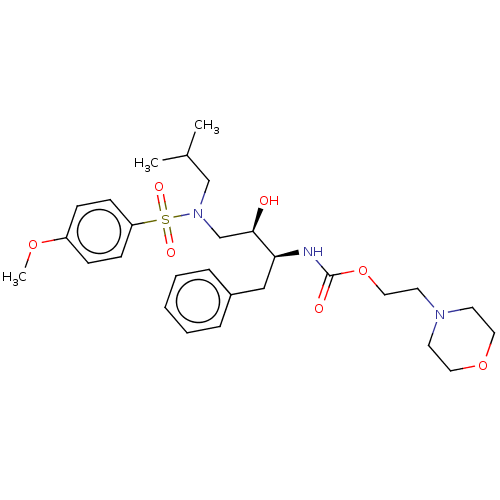
(CHEMBL4640442)Show SMILES COc1ccc(cc1)S(=O)(=O)N(CC(C)C)C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)OCCN1CCOCC1 |r| Show InChI InChI=1S/C28H41N3O7S/c1-22(2)20-31(39(34,35)25-11-9-24(36-3)10-12-25)21-27(32)26(19-23-7-5-4-6-8-23)29-28(33)38-18-15-30-13-16-37-17-14-30/h4-12,22,26-27,32H,13-21H2,1-3H3,(H,29,33)/t26-,27+/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Chinese Academy of Medical Science and Peking Union Medical College
Curated by ChEMBL
| Assay Description
Inhibition of wild type HIV1 protease expressed in Escherichia coli using Arg-Glu (EDANS)-Ser-Gln-Asn-Tyr-Pro-Ile-Val-Gln-Lys(DABCYL)-Arg as substrat... |
ACS Med Chem Lett 11: 1196-1204 (2020)
Article DOI: 10.1021/acsmedchemlett.0c00043
BindingDB Entry DOI: 10.7270/Q23200F2 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Rattus norvegicus (rat)) | BDBM50590648
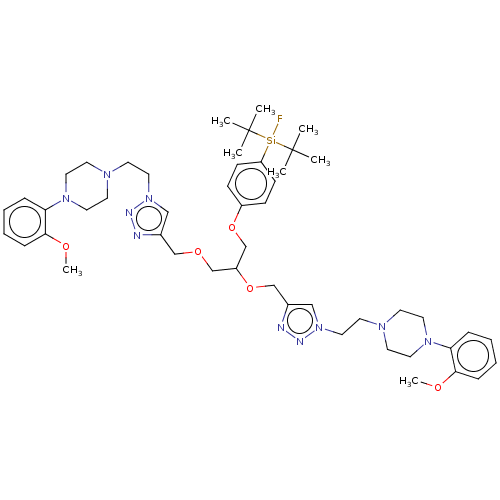
(CHEMBL5202019)Show SMILES [#6]-[#8]-c1ccccc1-[#7]-1-[#6]-[#6]-[#7](-[#6]-[#6]-n2cc(-[#6]-[#8]-[#6]-[#6](-[#6]-[#8]-c3ccc(cc3)[Si;v4]([18F])(C([#6])([#6])[#6])C([#6])([#6])[#6])-[#8]-[#6]-c3cn(-[#6]-[#6]-[#7]-4-[#6]-[#6]-[#7](-[#6]-[#6]-4)-c4ccccc4-[#8]-[#6])nn3)nn2)-[#6]-[#6]-1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0990 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00633
BindingDB Entry DOI: 10.7270/Q26M3BS8 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50576900
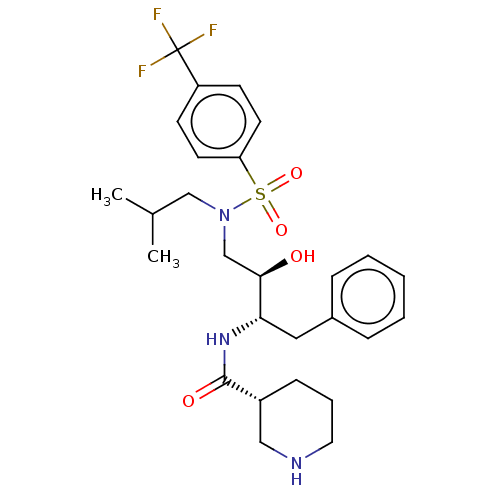
(CHEMBL4852688)Show SMILES CC(C)CN(C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)[C@@H]1CCCNC1)S(=O)(=O)c1ccc(cc1)C(F)(F)F |r| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of wild type HIV1 protease using Arg-Glu (EDANS)-Ser-Gln-Asn-Tyr-Pro-Ile-Val-Gln-Lys(DABCYL)-Arg as substrate preincubated for 20 to 30 mi... |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113450
BindingDB Entry DOI: 10.7270/Q2571GTH |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50039824
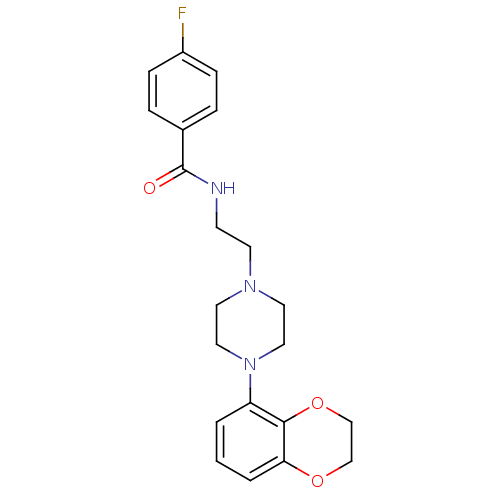
(CHEMBL81728 | N-{2-[4-(2,3-Dihydro-benzo[1,4]dioxi...)Show SMILES Fc1ccc(cc1)C(=O)NCCN1CCN(CC1)c1cccc2OCCOc12 Show InChI InChI=1S/C21H24FN3O3/c22-17-6-4-16(5-7-17)21(26)23-8-9-24-10-12-25(13-11-24)18-2-1-3-19-20(18)28-15-14-27-19/h1-7H,8-15H2,(H,23,26) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00633
BindingDB Entry DOI: 10.7270/Q26M3BS8 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50438565
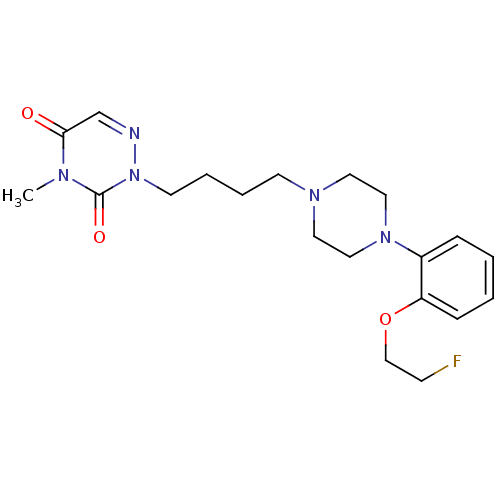
(CHEMBL2413153)Show SMILES Cn1c(=O)cnn(CCCCN2CCN(CC2)c2ccccc2OCCF)c1=O Show InChI InChI=1S/C20H28FN5O3/c1-23-19(27)16-22-26(20(23)28)10-5-4-9-24-11-13-25(14-12-24)17-6-2-3-7-18(17)29-15-8-21/h2-3,6-7,16H,4-5,8-15H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00633
BindingDB Entry DOI: 10.7270/Q26M3BS8 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50576913
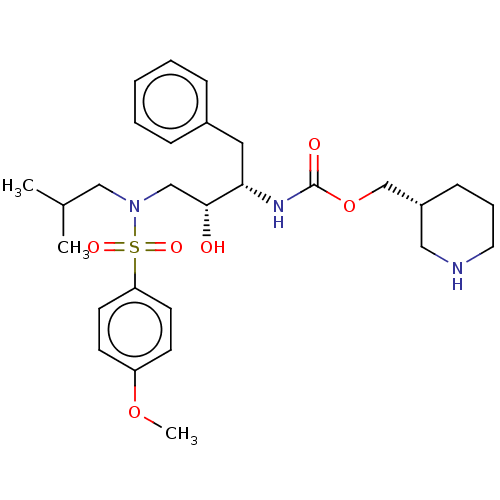
(CHEMBL4868812)Show SMILES COc1ccc(cc1)S(=O)(=O)N(CC(C)C)C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)OC[C@@H]1CCCNC1 |r| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of wild type HIV1 protease using Arg-Glu (EDANS)-Ser-Gln-Asn-Tyr-Pro-Ile-Val-Gln-Lys(DABCYL)-Arg as substrate preincubated for 20 to 30 mi... |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113450
BindingDB Entry DOI: 10.7270/Q2571GTH |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50092959
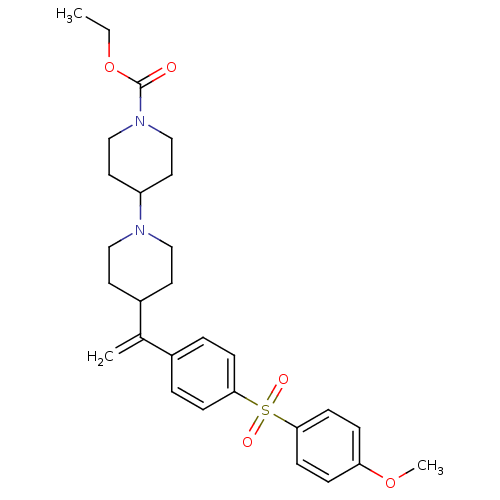
(4-{1-[4-(4-Methoxy-benzenesulfonyl)-phenyl]-vinyl}...)Show SMILES CCOC(=O)N1CCC(CC1)N1CCC(CC1)C(=C)c1ccc(cc1)S(=O)(=O)c1ccc(OC)cc1 Show InChI InChI=1S/C28H36N2O5S/c1-4-35-28(31)30-19-15-24(16-20-30)29-17-13-23(14-18-29)21(2)22-5-9-26(10-6-22)36(32,33)27-11-7-25(34-3)8-12-27/h5-12,23-24H,2,4,13-20H2,1,3H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Jiao Tong University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to muscarinic M2 receptor |
Bioorg Med Chem Lett 17: 2260-6 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.058
BindingDB Entry DOI: 10.7270/Q2668H0S |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM21398

(4-[4-(4-Chloro-phenyl)-4-hydroxy-piperidin-1-yl]-1...)Show SMILES OC1(CCN(CCCC(=O)c2ccc(F)cc2)CC1)c1ccc(Cl)cc1 Show InChI InChI=1S/C21H23ClFNO2/c22-18-7-5-17(6-8-18)21(26)11-14-24(15-12-21)13-1-2-20(25)16-3-9-19(23)10-4-16/h3-10,26H,1-2,11-15H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Antagonist activity at dopamine D2 receptor (unknown origin) |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01192
BindingDB Entry DOI: 10.7270/Q2XD15CH |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Rattus norvegicus (rat)) | BDBM50113332
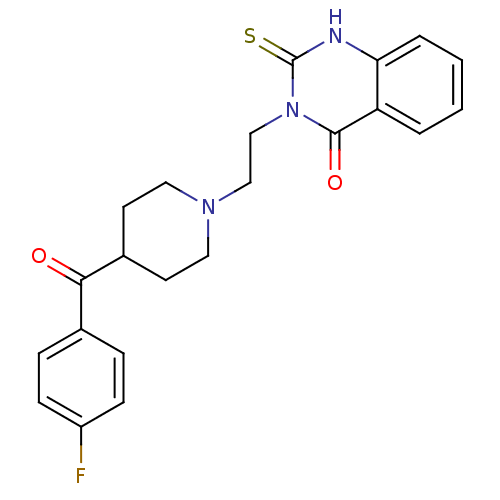
(3-(2-(4-(4-fluorobenzoyl)piperidin-1-yl)ethyl)-2-t...)Show SMILES Fc1ccc(cc1)C(=O)C1CCN(CCn2c(=S)[nH]c3ccccc3c2=O)CC1 Show InChI InChI=1S/C22H22FN3O2S/c23-17-7-5-15(6-8-17)20(27)16-9-11-25(12-10-16)13-14-26-21(28)18-3-1-2-4-19(18)24-22(26)29/h1-8,16H,9-14H2,(H,24,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00633
BindingDB Entry DOI: 10.7270/Q26M3BS8 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50377656

(CHEMBL259534)Show SMILES Clc1ccc(NC(=O)c2ccccc2NC(=O)c2ccc(cc2)-n2ccccc2=O)nc1 Show InChI InChI=1S/C24H17ClN4O3/c25-17-10-13-21(26-15-17)28-24(32)19-5-1-2-6-20(19)27-23(31)16-8-11-18(12-9-16)29-14-4-3-7-22(29)30/h1-15H,(H,27,31)(H,26,28,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a using acromogenic substrate S-2222 preincubated for 30 mins before substrate addition measured after 20 mins by spectr... |
Eur J Med Chem 95: 388-99 (2015)
Article DOI: 10.1016/j.ejmech.2015.03.052
BindingDB Entry DOI: 10.7270/Q29G5PH4 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50182020
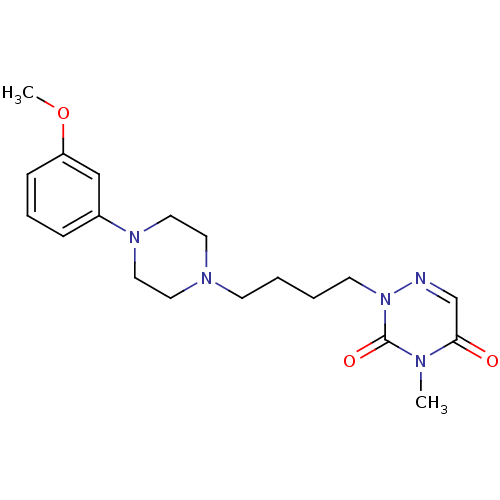
(2-(4-(4-(3-methoxyphenyl)piperazin-1-yl)butyl)-4-m...)Show SMILES COc1cccc(c1)N1CCN(CCCCn2ncc(=O)n(C)c2=O)CC1 Show InChI InChI=1S/C19H27N5O3/c1-21-18(25)15-20-24(19(21)26)9-4-3-8-22-10-12-23(13-11-22)16-6-5-7-17(14-16)27-2/h5-7,14-15H,3-4,8-13H2,1-2H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00633
BindingDB Entry DOI: 10.7270/Q26M3BS8 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50092961
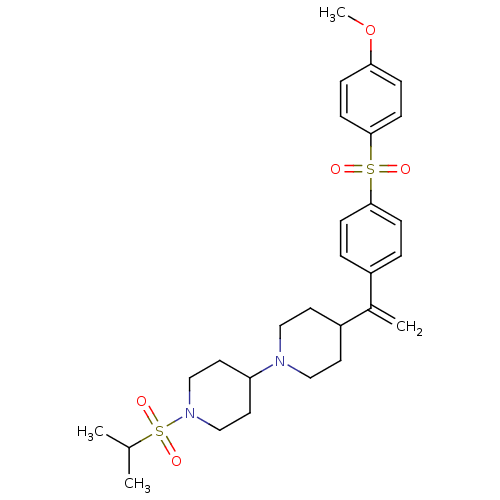
(1-(isopropylsulfonyl)-4-(4-(1-(4-(4-methoxyphenyls...)Show SMILES COc1ccc(cc1)S(=O)(=O)c1ccc(cc1)C(=C)C1CCN(CC1)C1CCN(CC1)S(=O)(=O)C(C)C Show InChI InChI=1S/C28H38N2O5S2/c1-21(2)37(33,34)30-19-15-25(16-20-30)29-17-13-24(14-18-29)22(3)23-5-9-27(10-6-23)36(31,32)28-11-7-26(35-4)8-12-28/h5-12,21,24-25H,3,13-20H2,1-2,4H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Jiao Tong University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to muscarinic M2 receptor |
Bioorg Med Chem Lett 17: 2260-6 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.058
BindingDB Entry DOI: 10.7270/Q2668H0S |
More data for this
Ligand-Target Pair | |
Sucrase-isomaltase, intestinal
(Rattus norvegicus (Rat)) | BDBM50612995

(CHEMBL5276586) | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Homo sapiens (Human)) | BDBM50011175
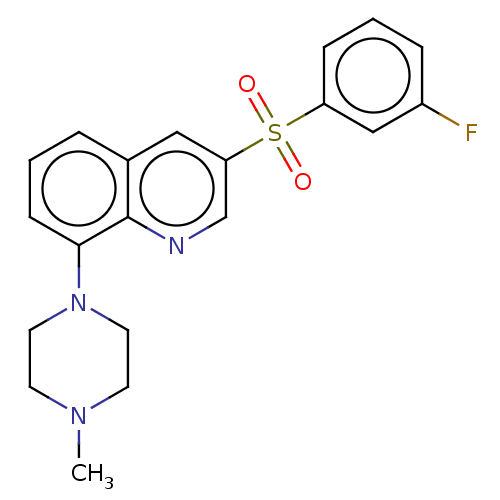
(CHEMBL3260313)Show SMILES CN1CCN(CC1)c1cccc2cc(cnc12)S(=O)(=O)c1cccc(F)c1 Show InChI InChI=1S/C20H20FN3O2S/c1-23-8-10-24(11-9-23)19-7-2-4-15-12-18(14-22-20(15)19)27(25,26)17-6-3-5-16(21)13-17/h2-7,12-14H,8-11H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00633
BindingDB Entry DOI: 10.7270/Q26M3BS8 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 7
(Homo sapiens (Human)) | BDBM50590673
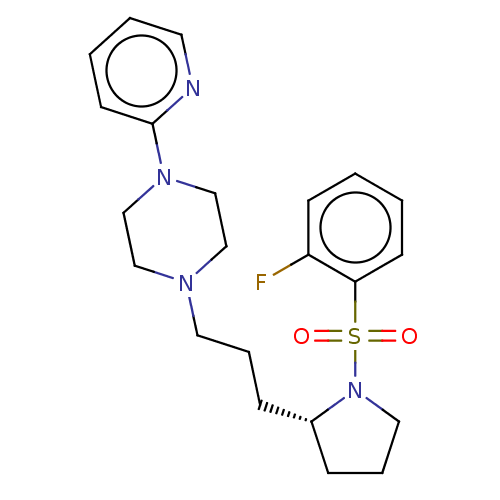
(CHEMBL5179460)Show SMILES Fc1ccccc1S(=O)(=O)N1CCC[C@@H]1CCCN1CCN(CC1)c1ccccn1 |r| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00633
BindingDB Entry DOI: 10.7270/Q26M3BS8 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein kinase FLT3
(Homo sapiens (Human)) | BDBM50355491

(CHEMBL1835870)Show SMILES Nc1nc(Nc2ccc(cc2)[C@H]2CC[C@@H](CC2)N2CCOCC2)nn1-c1ccccn1 |r,wU:11.11,wD:14.18,(-9.05,.18,;-7.58,.64,;-7.09,2.1,;-5.55,2.08,;-4.8,3.43,;-3.26,3.44,;-2.48,2.12,;-.94,2.14,;-.18,3.48,;-.98,4.81,;-2.52,4.79,;1.35,3.5,;2.1,4.85,;3.65,4.86,;4.44,3.53,;3.68,2.19,;2.14,2.18,;5.97,3.55,;6.75,2.22,;8.29,2.23,;9.05,3.57,;8.27,4.9,;6.73,4.89,;-5.09,.62,;-6.35,-.28,;-6.36,-1.81,;-5.04,-2.59,;-5.05,-4.13,;-6.39,-4.89,;-7.72,-4.11,;-7.71,-2.57,)| Show InChI InChI=1S/C23H29N7O/c24-22-27-23(28-30(22)21-3-1-2-12-25-21)26-19-8-4-17(5-9-19)18-6-10-20(11-7-18)29-13-15-31-16-14-29/h1-5,8-9,12,18,20H,6-7,10-11,13-16H2,(H3,24,26,27,28)/t18-,20- | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Kentucky
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant FLT3 by radiometric assay |
J Med Chem 55: 725-34 (2012)
Article DOI: 10.1021/jm201198w
BindingDB Entry DOI: 10.7270/Q2GQ6Z6R |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM8125

((3R,3aS,6aR)-hexahydrofuro[2,3-b]furan-3-yl N-[(2S...)Show SMILES [H][C@@]1(CO[C@@]2([H])OCC[C@@]12[H])OC(=O)N[C@@H](Cc1ccccc1)[C@H](O)CN(CC(C)C)S(=O)(=O)c1ccc(N)cc1 |r| Show InChI InChI=1S/C27H37N3O7S/c1-18(2)15-30(38(33,34)21-10-8-20(28)9-11-21)16-24(31)23(14-19-6-4-3-5-7-19)29-27(32)37-25-17-36-26-22(25)12-13-35-26/h3-11,18,22-26,31H,12-17,28H2,1-2H3,(H,29,32)/t22-,23-,24+,25-,26+/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of wild type HIV1 protease using Arg-Glu (EDANS)-Ser-Gln-Asn-Tyr-Pro-Ile-Val-Gln-Lys(DABCYL)-Arg as substrate preincubated for 20 to 30 mi... |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113450
BindingDB Entry DOI: 10.7270/Q2571GTH |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM21393

(7-(dipropylamino)-5,6,7,8-tetrahydronaphthalen-1-o...)Show InChI InChI=1S/C16H25NO/c1-3-10-17(11-4-2)14-9-8-13-6-5-7-16(18)15(13)12-14/h5-7,14,18H,3-4,8-12H2,1-2H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National University of Ireland Galway
Curated by ChEMBL
| Assay Description
Displacement of [3H]OH-DPAT from human recombinant 5-HT1A receptor measured after 60 mins by scintillation counter method |
Eur J Med Chem 176: 292-309 (2019)
Article DOI: 10.1016/j.ejmech.2019.04.064
BindingDB Entry DOI: 10.7270/Q2NP27V8 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50590637

(CHEMBL5189925) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00633
BindingDB Entry DOI: 10.7270/Q26M3BS8 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50576912
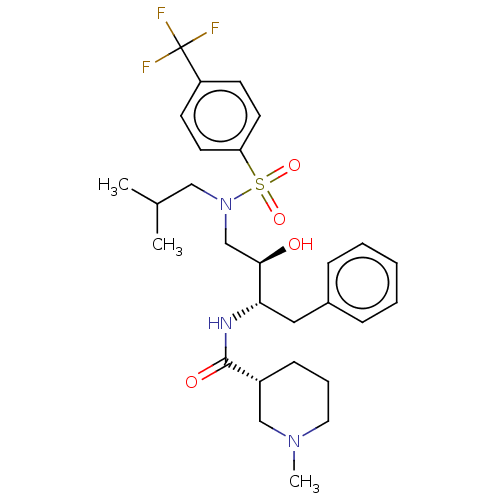
(CHEMBL4861507)Show SMILES CC(C)CN(C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)[C@@H]1CCCN(C)C1)S(=O)(=O)c1ccc(cc1)C(F)(F)F |r| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of wild type HIV1 protease using Arg-Glu (EDANS)-Ser-Gln-Asn-Tyr-Pro-Ile-Val-Gln-Lys(DABCYL)-Arg as substrate preincubated for 20 to 30 mi... |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113450
BindingDB Entry DOI: 10.7270/Q2571GTH |
More data for this
Ligand-Target Pair | |
Sucrase-isomaltase, intestinal
(Rattus norvegicus (Rat)) | BDBM50612998
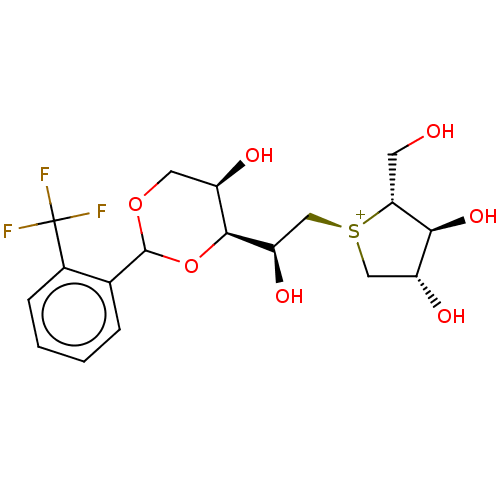
(CHEMBL5269400) | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Rattus norvegicus (rat)) | BDBM50590663
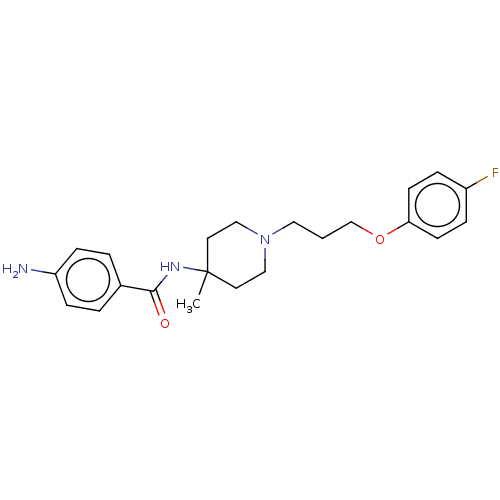
(CHEMBL5175726)Show SMILES CC1(CCN(CCCOc2ccc(F)cc2)CC1)NC(=O)c1ccc(N)cc1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00633
BindingDB Entry DOI: 10.7270/Q26M3BS8 |
More data for this
Ligand-Target Pair | |
Sigma non-opioid intracellular receptor 1
(Cavia porcellus (Guinea pig)) | BDBM50559185
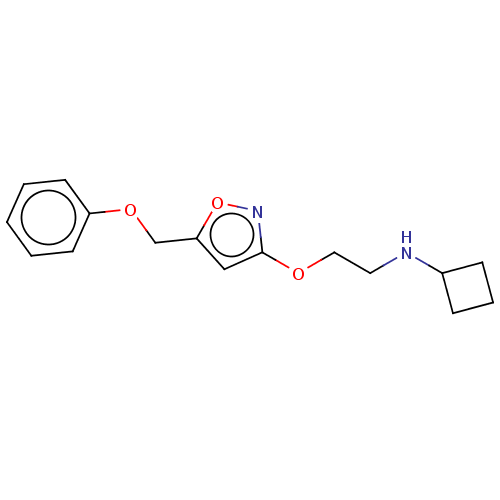
(CHEMBL4743313) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-(+)-pentazocine from sigma1 receptor in guinea pig brain homogenate incubated for 150 mins by liquid scintillation counting meth... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01192
BindingDB Entry DOI: 10.7270/Q2XD15CH |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2B
(Homo sapiens (Human)) | BDBM85862
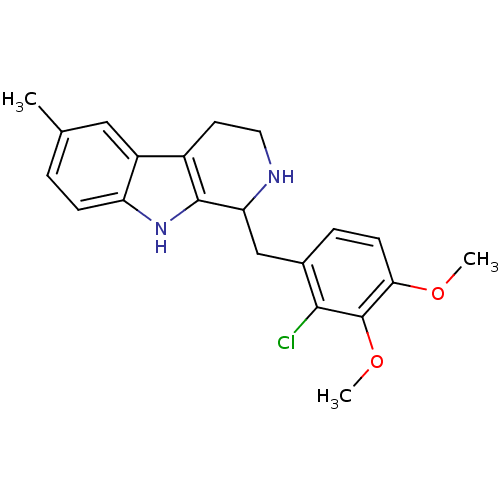
(1-(2-Chloro-3,4-dimethoxybenzyl)-6-methyl-2,3,4,9-...)Show SMILES COc1ccc(CC2NCCc3c2[nH]c2ccc(C)cc32)c(Cl)c1OC Show InChI InChI=1S/C21H23ClN2O2/c1-12-4-6-16-15(10-12)14-8-9-23-17(20(14)24-16)11-13-5-7-18(25-2)21(26-3)19(13)22/h4-7,10,17,23-24H,8-9,11H2,1-3H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00633
BindingDB Entry DOI: 10.7270/Q26M3BS8 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50541883
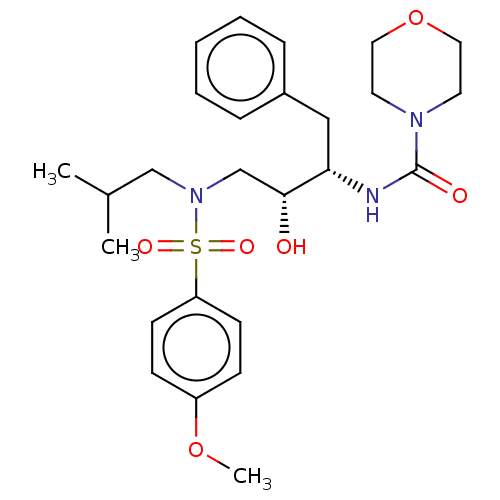
(CHEMBL4643409)Show SMILES COc1ccc(cc1)S(=O)(=O)N(CC(C)C)C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)N1CCOCC1 |r| Show InChI InChI=1S/C26H37N3O6S/c1-20(2)18-29(36(32,33)23-11-9-22(34-3)10-12-23)19-25(30)24(17-21-7-5-4-6-8-21)27-26(31)28-13-15-35-16-14-28/h4-12,20,24-25,30H,13-19H2,1-3H3,(H,27,31)/t24-,25+/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Chinese Academy of Medical Science and Peking Union Medical College
Curated by ChEMBL
| Assay Description
Inhibition of wild type HIV1 protease expressed in Escherichia coli using Arg-Glu (EDANS)-Ser-Gln-Asn-Tyr-Pro-Ile-Val-Gln-Lys(DABCYL)-Arg as substrat... |
ACS Med Chem Lett 11: 1196-1204 (2020)
Article DOI: 10.1021/acsmedchemlett.0c00043
BindingDB Entry DOI: 10.7270/Q23200F2 |
More data for this
Ligand-Target Pair | |
Sucrase-isomaltase, intestinal
(Rattus norvegicus (Rat)) | BDBM50613001

(CHEMBL5268739) | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | 0.210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 7
(Homo sapiens (Human)) | BDBM50590671
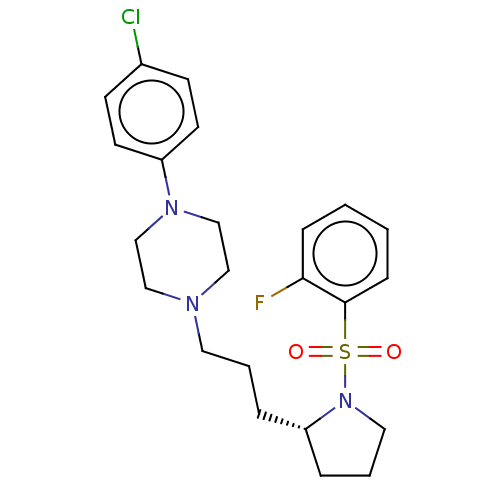
(CHEMBL5196234)Show SMILES Fc1ccccc1S(=O)(=O)N1CCC[C@@H]1CCCN1CCN(CC1)c1ccc(Cl)cc1 |r| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.220 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00633
BindingDB Entry DOI: 10.7270/Q26M3BS8 |
More data for this
Ligand-Target Pair | |
Sucrase-isomaltase, intestinal
(Rattus norvegicus (Rat)) | BDBM50613004

(CHEMBL5282029) | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Serotonin 2 (5-HT2) receptor
(RAT-Rattus norvegicus (Rat)-Rattus norvegicus (rat...) | BDBM50010044
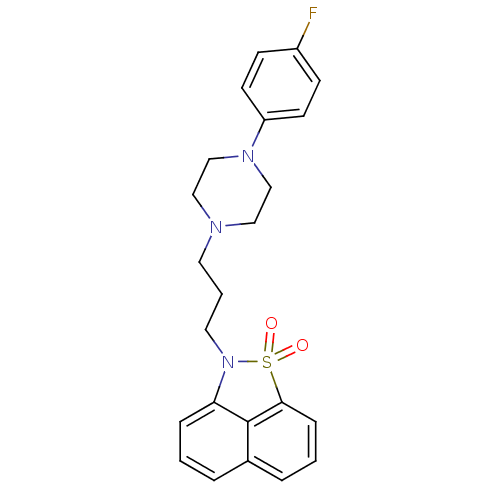
(2-{3-[4-(4-Fluoro-phenyl)-piperazin-1-yl]-propyl}-...)Show SMILES Fc1ccc(cc1)N1CCN(CCCN2c3cccc4cccc(c34)S2(=O)=O)CC1 Show InChI InChI=1S/C23H24FN3O2S/c24-19-8-10-20(11-9-19)26-16-14-25(15-17-26)12-3-13-27-21-6-1-4-18-5-2-7-22(23(18)21)30(27,28)29/h1-2,4-11H,3,12-17H2 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00633
BindingDB Entry DOI: 10.7270/Q26M3BS8 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50092969
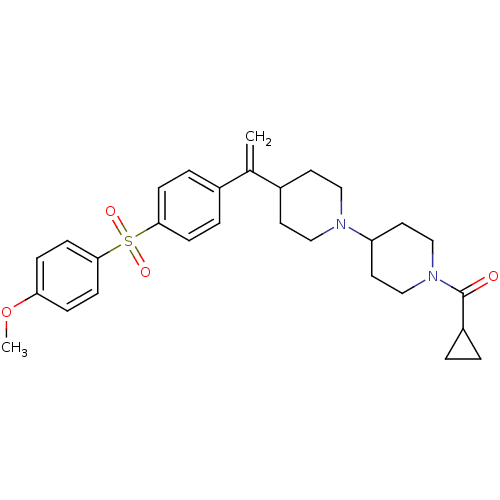
(CHEMBL74117 | Cyclopropyl-(4-{1-[4-(4-methoxy-benz...)Show SMILES COc1ccc(cc1)S(=O)(=O)c1ccc(cc1)C(=C)C1CCN(CC1)C1CCN(CC1)C(=O)C1CC1 Show InChI InChI=1S/C29H36N2O4S/c1-21(22-5-9-27(10-6-22)36(33,34)28-11-7-26(35-2)8-12-28)23-13-17-30(18-14-23)25-15-19-31(20-16-25)29(32)24-3-4-24/h5-12,23-25H,1,3-4,13-20H2,2H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Jiao Tong University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to muscarinic M2 receptor |
Bioorg Med Chem Lett 17: 2260-6 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.058
BindingDB Entry DOI: 10.7270/Q2668H0S |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50576899

(CHEMBL4852584)Show SMILES CC(C)CN(C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)[C@@H]1CCCNC1)S(=O)(=O)c1ccc(N)cc1 |r| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of wild type HIV1 protease using Arg-Glu (EDANS)-Ser-Gln-Asn-Tyr-Pro-Ile-Val-Gln-Lys(DABCYL)-Arg as substrate preincubated for 20 to 30 mi... |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113450
BindingDB Entry DOI: 10.7270/Q2571GTH |
More data for this
Ligand-Target Pair | |
Sigma non-opioid intracellular receptor 1
(Cavia porcellus (Guinea pig)) | BDBM50559179
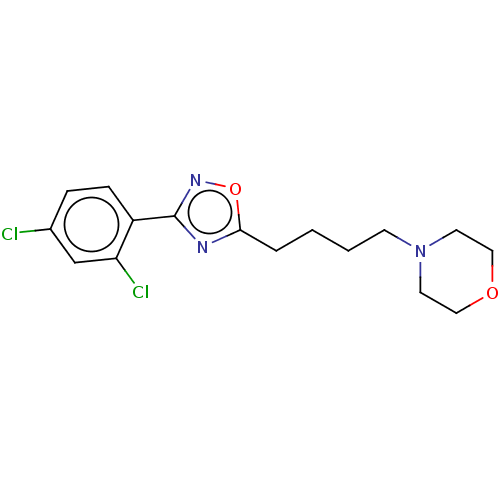
(CHEMBL4792161) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-(+)-pentazocine from sigma 1 receptor in Dunkin-Hartley guinea pig brain membranes incubated for 180 mins by liquid scintillatio... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01192
BindingDB Entry DOI: 10.7270/Q2XD15CH |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50576914

(CHEMBL4866330)Show SMILES CC(C)CN(C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)OC[C@@H]1CCCNC1)S(=O)(=O)c1ccc(N)cc1 |r| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of wild type HIV1 protease using Arg-Glu (EDANS)-Ser-Gln-Asn-Tyr-Pro-Ile-Val-Gln-Lys(DABCYL)-Arg as substrate preincubated for 20 to 30 mi... |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113450
BindingDB Entry DOI: 10.7270/Q2571GTH |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50092958
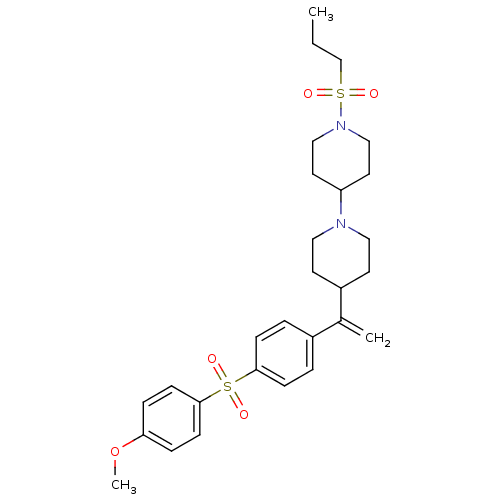
(4-(4-(1-(4-(4-methoxyphenylsulfonyl)phenyl)vinyl)p...)Show SMILES CCCS(=O)(=O)N1CCC(CC1)N1CCC(CC1)C(=C)c1ccc(cc1)S(=O)(=O)c1ccc(OC)cc1 Show InChI InChI=1S/C28H38N2O5S2/c1-4-21-36(31,32)30-19-15-25(16-20-30)29-17-13-24(14-18-29)22(2)23-5-9-27(10-6-23)37(33,34)28-11-7-26(35-3)8-12-28/h5-12,24-25H,2,4,13-21H2,1,3H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Jiao Tong University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to muscarinic M2 receptor |
Bioorg Med Chem Lett 17: 2260-6 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.058
BindingDB Entry DOI: 10.7270/Q2668H0S |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50092962
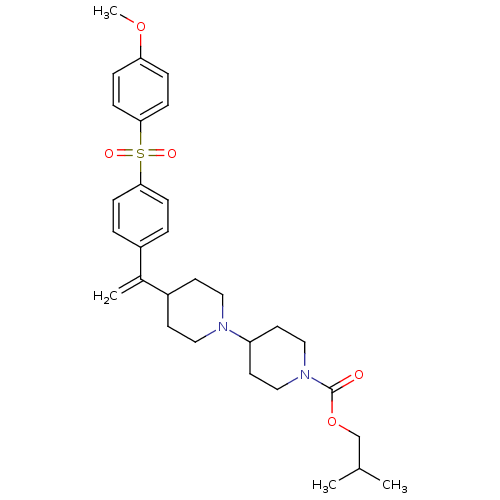
(4-{1-[4-(4-Methoxy-benzenesulfonyl)-phenyl]-vinyl}...)Show SMILES COc1ccc(cc1)S(=O)(=O)c1ccc(cc1)C(=C)C1CCN(CC1)C1CCN(CC1)C(=O)OCC(C)C Show InChI InChI=1S/C30H40N2O5S/c1-22(2)21-37-30(33)32-19-15-26(16-20-32)31-17-13-25(14-18-31)23(3)24-5-9-28(10-6-24)38(34,35)29-11-7-27(36-4)8-12-29/h5-12,22,25-26H,3,13-21H2,1-2,4H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Jiao Tong University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to muscarinic M2 receptor |
Bioorg Med Chem Lett 17: 2260-6 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.058
BindingDB Entry DOI: 10.7270/Q2668H0S |
More data for this
Ligand-Target Pair | |
Neuromedin-K receptor
(Homo sapiens (Human)) | BDBM50505284
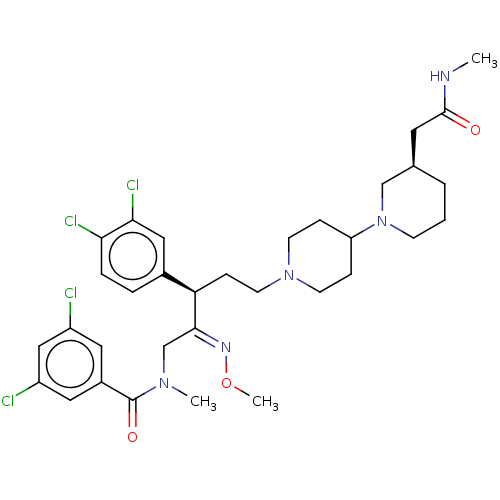
(CHEMBL4576324)Show SMILES CNC(=O)C[C@H]1CCCN(C1)C1CCN(CC[C@@H](\C(CN(C)C(=O)c2cc(Cl)cc(Cl)c2)=N\OC)c2ccc(Cl)c(Cl)c2)CC1 |r| Show InChI InChI=1S/C33H43Cl4N5O3/c1-38-32(43)15-22-5-4-11-42(20-22)27-8-12-41(13-9-27)14-10-28(23-6-7-29(36)30(37)18-23)31(39-45-3)21-40(2)33(44)24-16-25(34)19-26(35)17-24/h6-7,16-19,22,27-28H,4-5,8-15,20-21H2,1-3H3,(H,38,43)/b39-31+/t22-,28-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Displacement of [125I][MePhe]NKB from human NK3 receptor expressed in CHO cells membranes |
J Med Chem 62: 8881-8914 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00017
BindingDB Entry DOI: 10.7270/Q27084PF |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2B
(Homo sapiens (Human)) | BDBM50249134
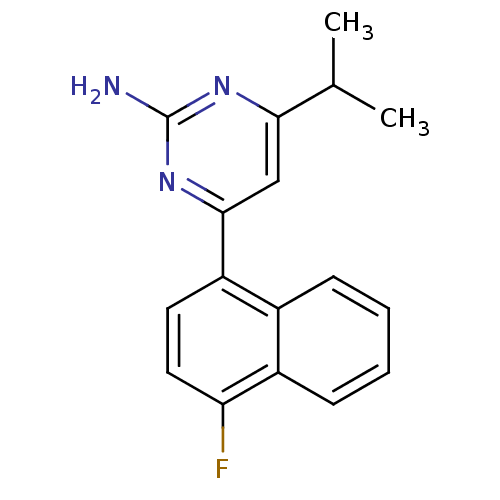
(4-(4-fluoronaphthalen-1-yl)-6-isopropylpyrimidin-2...)Show InChI InChI=1S/C17H16FN3/c1-10(2)15-9-16(21-17(19)20-15)13-7-8-14(18)12-6-4-3-5-11(12)13/h3-10H,1-2H3,(H2,19,20,21) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00633
BindingDB Entry DOI: 10.7270/Q26M3BS8 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50576916

(CHEMBL4875282)Show SMILES COc1ccc(cc1)S(=O)(=O)N(CC(C)C)C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)OC[C@H]1CCCNC1 |r| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of wild type HIV1 protease using Arg-Glu (EDANS)-Ser-Gln-Asn-Tyr-Pro-Ile-Val-Gln-Lys(DABCYL)-Arg as substrate preincubated for 20 to 30 mi... |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113450
BindingDB Entry DOI: 10.7270/Q2571GTH |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 4
(Homo sapiens (Human)) | BDBM50448450

(CHEMBL3126112)Show InChI InChI=1S/C16H21ClN2O4/c1-19-4-2-10(3-5-19)9-23-16(20)11-8-12(17)13(18)15-14(11)21-6-7-22-15/h8,10H,2-7,9,18H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00633
BindingDB Entry DOI: 10.7270/Q26M3BS8 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50576911

(CHEMBL4863180)Show SMILES CC(C)CN(C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)[C@@H]1CCCN(C)C1)S(=O)(=O)c1ccc(N)cc1 |r| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of wild type HIV1 protease using Arg-Glu (EDANS)-Ser-Gln-Asn-Tyr-Pro-Ile-Val-Gln-Lys(DABCYL)-Arg as substrate preincubated for 20 to 30 mi... |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113450
BindingDB Entry DOI: 10.7270/Q2571GTH |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2B
(Homo sapiens (Human)) | BDBM50590681
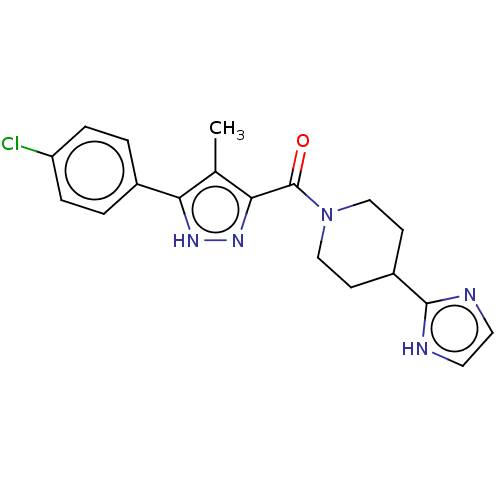
(CHEMBL5193649)Show SMILES Cc1c(n[nH]c1-c1ccc(Cl)cc1)C(=O)N1CCC(CC1)c1ncc[nH]1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00633
BindingDB Entry DOI: 10.7270/Q26M3BS8 |
More data for this
Ligand-Target Pair | |
Sucrase-isomaltase, intestinal
(Rattus norvegicus (Rat)) | BDBM50612994

(CHEMBL5283777) | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | 0.310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50590629
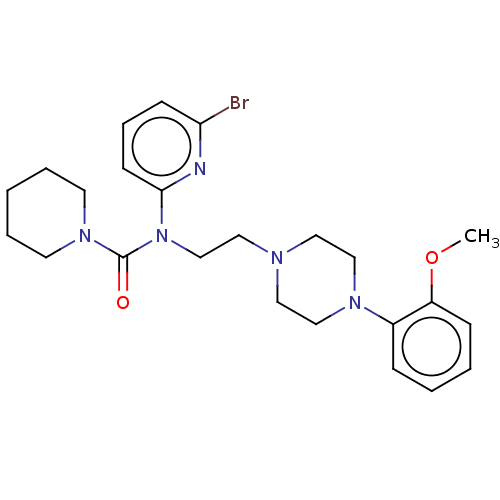
(CHEMBL5205609)Show SMILES COc1ccccc1N1CCN(CCN(C(=O)N2CCCCC2)c2cccc(Br)n2)CC1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00633
BindingDB Entry DOI: 10.7270/Q26M3BS8 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data