 Found 13 hits in this display
Found 13 hits in this display Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Proto-oncogene tyrosine-protein kinase receptor Ret
(Homo sapiens (Human)) | BDBM50170222
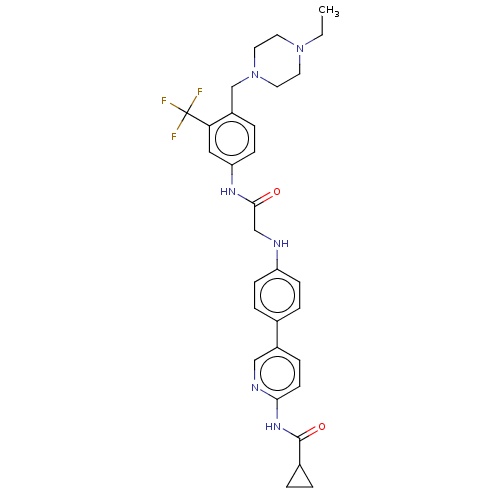
(CHEMBL3805771)Show SMILES CCN1CCN(Cc2ccc(NC(=O)CNc3ccc(cc3)-c3ccc(NC(=O)C4CC4)nc3)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C31H35F3N6O2/c1-2-39-13-15-40(16-14-39)20-24-7-11-26(17-27(24)31(32,33)34)37-29(41)19-35-25-9-5-21(6-10-25)23-8-12-28(36-18-23)38-30(42)22-3-4-22/h5-12,17-18,22,35H,2-4,13-16,19-20H2,1H3,(H,37,41)(H,36,38,42) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Experimental Therapeutics Centre
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant RET expressed in sf21 cells using Poly(Glu:Tyr) as substrate in presence of [gamma32P]ATP |
J Med Chem 59: 3063-78 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01712
BindingDB Entry DOI: 10.7270/Q2028TF3 |
More data for this
Ligand-Target Pair | |
MAP kinase-interacting serine/threonine-protein kinase 2
(Homo sapiens (Human)) | BDBM50170222

(CHEMBL3805771)Show SMILES CCN1CCN(Cc2ccc(NC(=O)CNc3ccc(cc3)-c3ccc(NC(=O)C4CC4)nc3)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C31H35F3N6O2/c1-2-39-13-15-40(16-14-39)20-24-7-11-26(17-27(24)31(32,33)34)37-29(41)19-35-25-9-5-21(6-10-25)23-8-12-28(36-18-23)38-30(42)22-3-4-22/h5-12,17-18,22,35H,2-4,13-16,19-20H2,1H3,(H,37,41)(H,36,38,42) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
Experimental Therapeutics Centre
Curated by ChEMBL
| Assay Description
Inhibition of N-terminal GST-tagged MNK2 kinase domain (72 to 385 residues) (unknown origin) expressed in Escherichia coli BL21(DE3) cells using JH3 ... |
J Med Chem 59: 3063-78 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01712
BindingDB Entry DOI: 10.7270/Q2028TF3 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein kinase FLT3
(Homo sapiens (Human)) | BDBM50170222

(CHEMBL3805771)Show SMILES CCN1CCN(Cc2ccc(NC(=O)CNc3ccc(cc3)-c3ccc(NC(=O)C4CC4)nc3)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C31H35F3N6O2/c1-2-39-13-15-40(16-14-39)20-24-7-11-26(17-27(24)31(32,33)34)37-29(41)19-35-25-9-5-21(6-10-25)23-8-12-28(36-18-23)38-30(42)22-3-4-22/h5-12,17-18,22,35H,2-4,13-16,19-20H2,1H3,(H,37,41)(H,36,38,42) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Experimental Therapeutics Centre
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant FLT3 expressed in sf21 cells using Poly(Glu:Tyr) as substrate in presence of [gamma32P]ATP |
J Med Chem 59: 3063-78 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01712
BindingDB Entry DOI: 10.7270/Q2028TF3 |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM50170222

(CHEMBL3805771)Show SMILES CCN1CCN(Cc2ccc(NC(=O)CNc3ccc(cc3)-c3ccc(NC(=O)C4CC4)nc3)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C31H35F3N6O2/c1-2-39-13-15-40(16-14-39)20-24-7-11-26(17-27(24)31(32,33)34)37-29(41)19-35-25-9-5-21(6-10-25)23-8-12-28(36-18-23)38-30(42)22-3-4-22/h5-12,17-18,22,35H,2-4,13-16,19-20H2,1H3,(H,37,41)(H,36,38,42) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 24 | n/a | n/a | n/a | n/a | n/a | n/a |
Experimental Therapeutics Centre
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant VEGFR2 expressed in sf21 cells using Poly(Glu:Tyr) as substrate in presence of [gamma32P]ATP |
J Med Chem 59: 3063-78 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01712
BindingDB Entry DOI: 10.7270/Q2028TF3 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL1
(Homo sapiens (Human)) | BDBM50170222

(CHEMBL3805771)Show SMILES CCN1CCN(Cc2ccc(NC(=O)CNc3ccc(cc3)-c3ccc(NC(=O)C4CC4)nc3)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C31H35F3N6O2/c1-2-39-13-15-40(16-14-39)20-24-7-11-26(17-27(24)31(32,33)34)37-29(41)19-35-25-9-5-21(6-10-25)23-8-12-28(36-18-23)38-30(42)22-3-4-22/h5-12,17-18,22,35H,2-4,13-16,19-20H2,1H3,(H,37,41)(H,36,38,42) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
Experimental Therapeutics Centre
Curated by ChEMBL
| Assay Description
Inhibition of full length human recombinant N-terminal His-tagged ABL (2 to 1130 residues) T315I mutant expressed in baculovirus expression system us... |
J Med Chem 59: 3063-78 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01712
BindingDB Entry DOI: 10.7270/Q2028TF3 |
More data for this
Ligand-Target Pair | |
Breakpoint cluster region protein/Tyrosine-protein kinase ABL1
(Homo sapiens (Human)) | BDBM50170222

(CHEMBL3805771)Show SMILES CCN1CCN(Cc2ccc(NC(=O)CNc3ccc(cc3)-c3ccc(NC(=O)C4CC4)nc3)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C31H35F3N6O2/c1-2-39-13-15-40(16-14-39)20-24-7-11-26(17-27(24)31(32,33)34)37-29(41)19-35-25-9-5-21(6-10-25)23-8-12-28(36-18-23)38-30(42)22-3-4-22/h5-12,17-18,22,35H,2-4,13-16,19-20H2,1H3,(H,37,41)(H,36,38,42) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 89 | n/a | n/a | n/a | n/a | n/a | n/a |
Experimental Therapeutics Centre
Curated by ChEMBL
| Assay Description
Inhibition of BCR fused full length human recombinant N-terminal His-tagged ABL (2 to 1130 residues) expressed in baculovirus expression system using... |
J Med Chem 59: 3063-78 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01712
BindingDB Entry DOI: 10.7270/Q2028TF3 |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha
(Homo sapiens (Human)) | BDBM50170222

(CHEMBL3805771)Show SMILES CCN1CCN(Cc2ccc(NC(=O)CNc3ccc(cc3)-c3ccc(NC(=O)C4CC4)nc3)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C31H35F3N6O2/c1-2-39-13-15-40(16-14-39)20-24-7-11-26(17-27(24)31(32,33)34)37-29(41)19-35-25-9-5-21(6-10-25)23-8-12-28(36-18-23)38-30(42)22-3-4-22/h5-12,17-18,22,35H,2-4,13-16,19-20H2,1H3,(H,37,41)(H,36,38,42) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 672 | n/a | n/a | n/a | n/a | n/a | n/a |
Experimental Therapeutics Centre
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PDGFRalpha expressed in sf21 cells using Poly(Glu:Tyr) as substrate in presence of [gamma32P]ATP |
J Med Chem 59: 3063-78 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01712
BindingDB Entry DOI: 10.7270/Q2028TF3 |
More data for this
Ligand-Target Pair | |
Mast/stem cell growth factor receptor Kit
(Homo sapiens (Human)) | BDBM50170222

(CHEMBL3805771)Show SMILES CCN1CCN(Cc2ccc(NC(=O)CNc3ccc(cc3)-c3ccc(NC(=O)C4CC4)nc3)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C31H35F3N6O2/c1-2-39-13-15-40(16-14-39)20-24-7-11-26(17-27(24)31(32,33)34)37-29(41)19-35-25-9-5-21(6-10-25)23-8-12-28(36-18-23)38-30(42)22-3-4-22/h5-12,17-18,22,35H,2-4,13-16,19-20H2,1H3,(H,37,41)(H,36,38,42) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 888 | n/a | n/a | n/a | n/a | n/a | n/a |
Experimental Therapeutics Centre
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant c-KIT expressed in insect cells using Poly(Glu:Tyr) as substrate in presence of [gamma32P]ATP |
J Med Chem 59: 3063-78 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01712
BindingDB Entry DOI: 10.7270/Q2028TF3 |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 2
(Homo sapiens (Human)) | BDBM50170222

(CHEMBL3805771)Show SMILES CCN1CCN(Cc2ccc(NC(=O)CNc3ccc(cc3)-c3ccc(NC(=O)C4CC4)nc3)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C31H35F3N6O2/c1-2-39-13-15-40(16-14-39)20-24-7-11-26(17-27(24)31(32,33)34)37-29(41)19-35-25-9-5-21(6-10-25)23-8-12-28(36-18-23)38-30(42)22-3-4-22/h5-12,17-18,22,35H,2-4,13-16,19-20H2,1H3,(H,37,41)(H,36,38,42) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 930 | n/a | n/a | n/a | n/a | n/a | n/a |
Experimental Therapeutics Centre
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant FGFR2 expressed in sf21 cells using Poly(Glu:Tyr) as substrate in presence of [gamma32P]ATP |
J Med Chem 59: 3063-78 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01712
BindingDB Entry DOI: 10.7270/Q2028TF3 |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50170222

(CHEMBL3805771)Show SMILES CCN1CCN(Cc2ccc(NC(=O)CNc3ccc(cc3)-c3ccc(NC(=O)C4CC4)nc3)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C31H35F3N6O2/c1-2-39-13-15-40(16-14-39)20-24-7-11-26(17-27(24)31(32,33)34)37-29(41)19-35-25-9-5-21(6-10-25)23-8-12-28(36-18-23)38-30(42)22-3-4-22/h5-12,17-18,22,35H,2-4,13-16,19-20H2,1H3,(H,37,41)(H,36,38,42) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.08E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Experimental Therapeutics Centre
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PDGFRbeta expressed in sf21 cells using Poly(Glu:Tyr) as substrate in presence of [gamma32P]ATP |
J Med Chem 59: 3063-78 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01712
BindingDB Entry DOI: 10.7270/Q2028TF3 |
More data for this
Ligand-Target Pair | |
MAP kinase-interacting serine/threonine-protein kinase 1
(Homo sapiens (Human)) | BDBM50170222

(CHEMBL3805771)Show SMILES CCN1CCN(Cc2ccc(NC(=O)CNc3ccc(cc3)-c3ccc(NC(=O)C4CC4)nc3)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C31H35F3N6O2/c1-2-39-13-15-40(16-14-39)20-24-7-11-26(17-27(24)31(32,33)34)37-29(41)19-35-25-9-5-21(6-10-25)23-8-12-28(36-18-23)38-30(42)22-3-4-22/h5-12,17-18,22,35H,2-4,13-16,19-20H2,1H3,(H,37,41)(H,36,38,42) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.52E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Experimental Therapeutics Centre
Curated by ChEMBL
| Assay Description
Inhibition of N-terminal GST-tagged MNK1 kinase domain (37 to 341 residues) (unknown origin) expressed in Escherichia coli BL21(DE3) cells using JH3 ... |
J Med Chem 59: 3063-78 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01712
BindingDB Entry DOI: 10.7270/Q2028TF3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50170222

(CHEMBL3805771)Show SMILES CCN1CCN(Cc2ccc(NC(=O)CNc3ccc(cc3)-c3ccc(NC(=O)C4CC4)nc3)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C31H35F3N6O2/c1-2-39-13-15-40(16-14-39)20-24-7-11-26(17-27(24)31(32,33)34)37-29(41)19-35-25-9-5-21(6-10-25)23-8-12-28(36-18-23)38-30(42)22-3-4-22/h5-12,17-18,22,35H,2-4,13-16,19-20H2,1H3,(H,37,41)(H,36,38,42) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Experimental Therapeutics Centre
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
J Med Chem 59: 3063-78 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01712
BindingDB Entry DOI: 10.7270/Q2028TF3 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50170222

(CHEMBL3805771)Show SMILES CCN1CCN(Cc2ccc(NC(=O)CNc3ccc(cc3)-c3ccc(NC(=O)C4CC4)nc3)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C31H35F3N6O2/c1-2-39-13-15-40(16-14-39)20-24-7-11-26(17-27(24)31(32,33)34)37-29(41)19-35-25-9-5-21(6-10-25)23-8-12-28(36-18-23)38-30(42)22-3-4-22/h5-12,17-18,22,35H,2-4,13-16,19-20H2,1H3,(H,37,41)(H,36,38,42) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Experimental Therapeutics Centre
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
J Med Chem 59: 3063-78 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01712
BindingDB Entry DOI: 10.7270/Q2028TF3 |
More data for this
Ligand-Target Pair | |