 Found 22 hits in this display
Found 22 hits in this display Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
P2X purinoceptor 2
(Homo sapiens (Human)) | BDBM50118232

(2-MeSATP | ATP, 2-meS | CHEMBL336208)Show SMILES CSc1nc(N)c2ncn([C@@H]3O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]3O)c2n1 Show InChI InChI=1S/C11H18N5O13P3S/c1-33-11-14-8(12)5-9(15-11)16(3-13-5)10-7(18)6(17)4(27-10)2-26-31(22,23)29-32(24,25)28-30(19,20)21/h3-4,6-7,10,17-18H,2H2,1H3,(H,22,23)(H,24,25)(H2,12,14,15)(H2,19,20,21)/t4-,6-,7-,10-/m1/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 2.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 407-16 (2004)
Article DOI: 10.1124/jpet.103.064907
BindingDB Entry DOI: 10.7270/Q2SQ8XZR |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 3
(Homo sapiens (Human)) | BDBM50118232

(2-MeSATP | ATP, 2-meS | CHEMBL336208)Show SMILES CSc1nc(N)c2ncn([C@@H]3O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]3O)c2n1 Show InChI InChI=1S/C11H18N5O13P3S/c1-33-11-14-8(12)5-9(15-11)16(3-13-5)10-7(18)6(17)4(27-10)2-26-31(22,23)29-32(24,25)28-30(19,20)21/h3-4,6-7,10,17-18H,2H2,1H3,(H,22,23)(H,24,25)(H2,12,14,15)(H2,19,20,21)/t4-,6-,7-,10-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 8.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 407-16 (2004)
Article DOI: 10.1124/jpet.103.064907
BindingDB Entry DOI: 10.7270/Q2SQ8XZR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
S-adenosylmethionine synthase isoform type-1/type-2
(Rattus norvegicus) | BDBM50118232

(2-MeSATP | ATP, 2-meS | CHEMBL336208)Show SMILES CSc1nc(N)c2ncn([C@@H]3O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]3O)c2n1 Show InChI InChI=1S/C11H18N5O13P3S/c1-33-11-14-8(12)5-9(15-11)16(3-13-5)10-7(18)6(17)4(27-10)2-26-31(22,23)29-32(24,25)28-30(19,20)21/h3-4,6-7,10,17-18H,2H2,1H3,(H,22,23)(H,24,25)(H2,12,14,15)(H2,19,20,21)/t4-,6-,7-,10-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| 1.10E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory constant against rat Methionine adenosyltransferase was reported |
J Med Chem 29: 318-22 (1986)
BindingDB Entry DOI: 10.7270/Q2DF6RSH |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-2
(Rattus norvegicus) | BDBM50118232

(2-MeSATP | ATP, 2-meS | CHEMBL336208)Show SMILES CSc1nc(N)c2ncn([C@@H]3O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]3O)c2n1 Show InChI InChI=1S/C11H18N5O13P3S/c1-33-11-14-8(12)5-9(15-11)16(3-13-5)10-7(18)6(17)4(27-10)2-26-31(22,23)29-32(24,25)28-30(19,20)21/h3-4,6-7,10,17-18H,2H2,1H3,(H,22,23)(H,24,25)(H2,12,14,15)(H2,19,20,21)/t4-,6-,7-,10-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| 6.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory constant against rat kidney Methionine adenosyltransferase II |
J Med Chem 29: 318-22 (1986)
BindingDB Entry DOI: 10.7270/Q2DF6RSH |
More data for this
Ligand-Target Pair | |
P2Y purinoceptor 6
(Homo sapiens (Human)) | BDBM50118232

(2-MeSATP | ATP, 2-meS | CHEMBL336208)Show SMILES CSc1nc(N)c2ncn([C@@H]3O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]3O)c2n1 Show InChI InChI=1S/C11H18N5O13P3S/c1-33-11-14-8(12)5-9(15-11)16(3-13-5)10-7(18)6(17)4(27-10)2-26-31(22,23)29-32(24,25)28-30(19,20)21/h3-4,6-7,10,17-18H,2H2,1H3,(H,22,23)(H,24,25)(H2,12,14,15)(H2,19,20,21)/t4-,6-,7-,10-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 1.00E+5 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
The compound was evaluated for agonist activity against phospholipase C coupled recombinant human P2Y purinoceptor 6 (P2Y6) |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 6
(Rattus norvegicus) | BDBM50118232

(2-MeSATP | ATP, 2-meS | CHEMBL336208)Show SMILES CSc1nc(N)c2ncn([C@@H]3O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]3O)c2n1 Show InChI InChI=1S/C11H18N5O13P3S/c1-33-11-14-8(12)5-9(15-11)16(3-13-5)10-7(18)6(17)4(27-10)2-26-31(22,23)29-32(24,25)28-30(19,20)21/h3-4,6-7,10,17-18H,2H2,1H3,(H,22,23)(H,24,25)(H2,12,14,15)(H2,19,20,21)/t4-,6-,7-,10-/m1/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 600 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
Antagonist activity against recombinant rat P2X purinoceptor 6 (P2X6 ) |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 4
(RAT) | BDBM50118232

(2-MeSATP | ATP, 2-meS | CHEMBL336208)Show SMILES CSc1nc(N)c2ncn([C@@H]3O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]3O)c2n1 Show InChI InChI=1S/C11H18N5O13P3S/c1-33-11-14-8(12)5-9(15-11)16(3-13-5)10-7(18)6(17)4(27-10)2-26-31(22,23)29-32(24,25)28-30(19,20)21/h3-4,6-7,10,17-18H,2H2,1H3,(H,22,23)(H,24,25)(H2,12,14,15)(H2,19,20,21)/t4-,6-,7-,10-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 7.40E+3 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
The compound was evaluated for antagonist activity against recombinant rat P2X purinoceptor 4 (P2X4) 3 uM |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 1
(Homo sapiens (Human)) | BDBM50118232

(2-MeSATP | ATP, 2-meS | CHEMBL336208)Show SMILES CSc1nc(N)c2ncn([C@@H]3O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]3O)c2n1 Show InChI InChI=1S/C11H18N5O13P3S/c1-33-11-14-8(12)5-9(15-11)16(3-13-5)10-7(18)6(17)4(27-10)2-26-31(22,23)29-32(24,25)28-30(19,20)21/h3-4,6-7,10,17-18H,2H2,1H3,(H,22,23)(H,24,25)(H2,12,14,15)(H2,19,20,21)/t4-,6-,7-,10-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 54 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
The compound was evaluated for antagonist activity against recombinant human P2X purinoceptor 1 (P2X1 ) |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 4
(Homo sapiens (Human)) | BDBM50118232

(2-MeSATP | ATP, 2-meS | CHEMBL336208)Show SMILES CSc1nc(N)c2ncn([C@@H]3O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]3O)c2n1 Show InChI InChI=1S/C11H18N5O13P3S/c1-33-11-14-8(12)5-9(15-11)16(3-13-5)10-7(18)6(17)4(27-10)2-26-31(22,23)29-32(24,25)28-30(19,20)21/h3-4,6-7,10,17-18H,2H2,1H3,(H,22,23)(H,24,25)(H2,12,14,15)(H2,19,20,21)/t4-,6-,7-,10-/m1/s1 | KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
Antagonist activity against recombinant human P2X purinoceptor 4 (P2X4) |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 5
(Rattus norvegicus) | BDBM50118232

(2-MeSATP | ATP, 2-meS | CHEMBL336208)Show SMILES CSc1nc(N)c2ncn([C@@H]3O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]3O)c2n1 Show InChI InChI=1S/C11H18N5O13P3S/c1-33-11-14-8(12)5-9(15-11)16(3-13-5)10-7(18)6(17)4(27-10)2-26-31(22,23)29-32(24,25)28-30(19,20)21/h3-4,6-7,10,17-18H,2H2,1H3,(H,22,23)(H,24,25)(H2,12,14,15)(H2,19,20,21)/t4-,6-,7-,10-/m1/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
Antagonist activity against recombinant rat P2X purinoceptor 5 (P2X5) |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2Y purinoceptor 1
(Meleagris gallopavo) | BDBM50118232

(2-MeSATP | ATP, 2-meS | CHEMBL336208)Show SMILES CSc1nc(N)c2ncn([C@@H]3O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]3O)c2n1 Show InChI InChI=1S/C11H18N5O13P3S/c1-33-11-14-8(12)5-9(15-11)16(3-13-5)10-7(18)6(17)4(27-10)2-26-31(22,23)29-32(24,25)28-30(19,20)21/h3-4,6-7,10,17-18H,2H2,1H3,(H,22,23)(H,24,25)(H2,12,14,15)(H2,19,20,21)/t4-,6-,7-,10-/m1/s1 | MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 8 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
Activation of Purinoceptor P2Y1-mediated phospholipase C in turkey erythrocyte membranes |
J Med Chem 45: 2090-100 (2002)
BindingDB Entry DOI: 10.7270/Q25H7GZB |
More data for this
Ligand-Target Pair | |
P2Y purinoceptor 1
(Homo sapiens (Human)) | BDBM50118232

(2-MeSATP | ATP, 2-meS | CHEMBL336208)Show SMILES CSc1nc(N)c2ncn([C@@H]3O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]3O)c2n1 Show InChI InChI=1S/C11H18N5O13P3S/c1-33-11-14-8(12)5-9(15-11)16(3-13-5)10-7(18)6(17)4(27-10)2-26-31(22,23)29-32(24,25)28-30(19,20)21/h3-4,6-7,10,17-18H,2H2,1H3,(H,22,23)(H,24,25)(H2,12,14,15)(H2,19,20,21)/t4-,6-,7-,10-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 34 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
Accumulation of inositol phosphate in 1321N1 astrocytoma cells expressing human P2Y1 purinoceptor |
J Med Chem 45: 2090-100 (2002)
BindingDB Entry DOI: 10.7270/Q25H7GZB |
More data for this
Ligand-Target Pair | |
P2Y purinoceptor 1
(Rattus norvegicus) | BDBM50422408
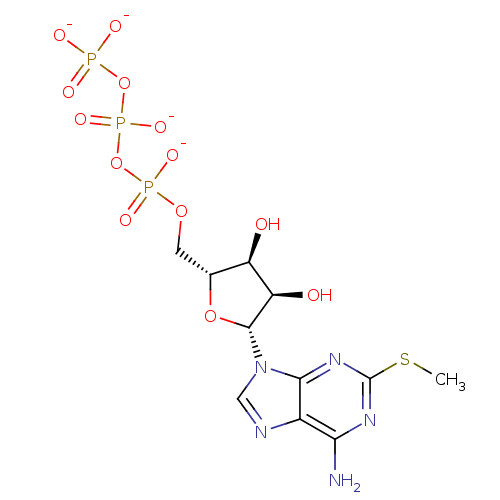
(CHEMBL2364736)Show SMILES CSc1nc(N)c2ncn([C@@H]3O[C@H](COP([O-])(=O)OP([O-])(=O)OP([O-])([O-])=O)[C@@H](O)[C@H]3O)c2n1 |r| Show InChI InChI=1S/C11H18N5O13P3S/c1-33-11-14-8(12)5-9(15-11)16(3-13-5)10-7(18)6(17)4(27-10)2-26-31(22,23)29-32(24,25)28-30(19,20)21/h3-4,6-7,10,17-18H,2H2,1H3,(H,22,23)(H,24,25)(H2,12,14,15)(H2,19,20,21)/p-4/t4-,6-,7-,10-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 4.5 | n/a | n/a | n/a | n/a |
Bar-Ilan University
Curated by ChEMBL
| Assay Description
Agonists activity was evaluated by release of [Ca2+] release of HEK 293 cells stably transfected with rat-brain P2Y purinoceptor 1 (P2Y1-R) |
J Med Chem 45: 5384-96 (2002)
BindingDB Entry DOI: 10.7270/Q2M909DD |
More data for this
Ligand-Target Pair | |
P2Y purinoceptor 1
(Rattus norvegicus) | BDBM50118232

(2-MeSATP | ATP, 2-meS | CHEMBL336208)Show SMILES CSc1nc(N)c2ncn([C@@H]3O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]3O)c2n1 Show InChI InChI=1S/C11H18N5O13P3S/c1-33-11-14-8(12)5-9(15-11)16(3-13-5)10-7(18)6(17)4(27-10)2-26-31(22,23)29-32(24,25)28-30(19,20)21/h3-4,6-7,10,17-18H,2H2,1H3,(H,22,23)(H,24,25)(H2,12,14,15)(H2,19,20,21)/t4-,6-,7-,10-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1 | n/a | n/a | n/a | n/a |
Bar-Ilan University
Curated by ChEMBL
| Assay Description
Concentration required for calcium mobilization at rat purinergic 2Y1 receptor expressed in HEK 293 cells |
J Med Chem 47: 4405-16 (2004)
Article DOI: 10.1021/jm049771u
BindingDB Entry DOI: 10.7270/Q2PC334S |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 4
(Mus musculus) | BDBM50118232

(2-MeSATP | ATP, 2-meS | CHEMBL336208)Show SMILES CSc1nc(N)c2ncn([C@@H]3O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]3O)c2n1 Show InChI InChI=1S/C11H18N5O13P3S/c1-33-11-14-8(12)5-9(15-11)16(3-13-5)10-7(18)6(17)4(27-10)2-26-31(22,23)29-32(24,25)28-30(19,20)21/h3-4,6-7,10,17-18H,2H2,1H3,(H,22,23)(H,24,25)(H2,12,14,15)(H2,19,20,21)/t4-,6-,7-,10-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Antagonist activity at mouse P2X4 receptor by cell-based calcium influx assay |
J Med Chem 55: 9576-88 (2012)
Article DOI: 10.1021/jm300845v
BindingDB Entry DOI: 10.7270/Q2XK8GPN |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 4
(RAT) | BDBM50118232

(2-MeSATP | ATP, 2-meS | CHEMBL336208)Show SMILES CSc1nc(N)c2ncn([C@@H]3O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]3O)c2n1 Show InChI InChI=1S/C11H18N5O13P3S/c1-33-11-14-8(12)5-9(15-11)16(3-13-5)10-7(18)6(17)4(27-10)2-26-31(22,23)29-32(24,25)28-30(19,20)21/h3-4,6-7,10,17-18H,2H2,1H3,(H,22,23)(H,24,25)(H2,12,14,15)(H2,19,20,21)/t4-,6-,7-,10-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Antagonist activity at rat P2X4 receptor by cell-based calcium influx assay |
J Med Chem 55: 9576-88 (2012)
Article DOI: 10.1021/jm300845v
BindingDB Entry DOI: 10.7270/Q2XK8GPN |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 4
(Homo sapiens (Human)) | BDBM50118232

(2-MeSATP | ATP, 2-meS | CHEMBL336208)Show SMILES CSc1nc(N)c2ncn([C@@H]3O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]3O)c2n1 Show InChI InChI=1S/C11H18N5O13P3S/c1-33-11-14-8(12)5-9(15-11)16(3-13-5)10-7(18)6(17)4(27-10)2-26-31(22,23)29-32(24,25)28-30(19,20)21/h3-4,6-7,10,17-18H,2H2,1H3,(H,22,23)(H,24,25)(H2,12,14,15)(H2,19,20,21)/t4-,6-,7-,10-/m1/s1 | KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Antagonist activity at human P2X4 receptor expressed in 1321N1 cells assessed as inhibition of ATP-induced cytosolic calcium influx compound preincub... |
J Med Chem 55: 9576-88 (2012)
Article DOI: 10.1021/jm300845v
BindingDB Entry DOI: 10.7270/Q2XK8GPN |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 2
(RAT) | BDBM50118232

(2-MeSATP | ATP, 2-meS | CHEMBL336208)Show SMILES CSc1nc(N)c2ncn([C@@H]3O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]3O)c2n1 Show InChI InChI=1S/C11H18N5O13P3S/c1-33-11-14-8(12)5-9(15-11)16(3-13-5)10-7(18)6(17)4(27-10)2-26-31(22,23)29-32(24,25)28-30(19,20)21/h3-4,6-7,10,17-18H,2H2,1H3,(H,22,23)(H,24,25)(H2,12,14,15)(H2,19,20,21)/t4-,6-,7-,10-/m1/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
The compound was evaluated for antagonist activity against recombinant rat receptor P2X purinoceptor 2 (P2X2) at 10 uM, expressed in Xenopus oocytes |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2Y purinoceptor 11
(Homo sapiens (Human)) | BDBM50118232

(2-MeSATP | ATP, 2-meS | CHEMBL336208)Show SMILES CSc1nc(N)c2ncn([C@@H]3O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]3O)c2n1 Show InChI InChI=1S/C11H18N5O13P3S/c1-33-11-14-8(12)5-9(15-11)16(3-13-5)10-7(18)6(17)4(27-10)2-26-31(22,23)29-32(24,25)28-30(19,20)21/h3-4,6-7,10,17-18H,2H2,1H3,(H,22,23)(H,24,25)(H2,12,14,15)(H2,19,20,21)/t4-,6-,7-,10-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 5.00E+4 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
The compound was evaluated for antagonist activity against phospholipase C coupled human P2Y purinoceptor 11 (P2Y11) |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 3
(Homo sapiens (Human)) | BDBM50118232

(2-MeSATP | ATP, 2-meS | CHEMBL336208)Show SMILES CSc1nc(N)c2ncn([C@@H]3O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]3O)c2n1 Show InChI InChI=1S/C11H18N5O13P3S/c1-33-11-14-8(12)5-9(15-11)16(3-13-5)10-7(18)6(17)4(27-10)2-26-31(22,23)29-32(24,25)28-30(19,20)21/h3-4,6-7,10,17-18H,2H2,1H3,(H,22,23)(H,24,25)(H2,12,14,15)(H2,19,20,21)/t4-,6-,7-,10-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| PDB
PubMed
| n/a | n/a | n/a | n/a | 350 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
The compound was evaluated for antagonist activity against recombinant rat P2X purinoceptor 3 (P2X3) at 10 uM, expressed in Xenopus oocytes |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
P2Y purinoceptor 1
(Meleagris gallopavo) | BDBM50118232

(2-MeSATP | ATP, 2-meS | CHEMBL336208)Show SMILES CSc1nc(N)c2ncn([C@@H]3O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]3O)c2n1 Show InChI InChI=1S/C11H18N5O13P3S/c1-33-11-14-8(12)5-9(15-11)16(3-13-5)10-7(18)6(17)4(27-10)2-26-31(22,23)29-32(24,25)28-30(19,20)21/h3-4,6-7,10,17-18H,2H2,1H3,(H,22,23)(H,24,25)(H2,12,14,15)(H2,19,20,21)/t4-,6-,7-,10-/m1/s1 | MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 8 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
Evaluated for agonist activity against phospholipase C coupled P2Y purinoceptor 1 (P2Y1) of turkey erythrocytes |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2Y purinoceptor 12
(Rattus norvegicus) | BDBM50118232

(2-MeSATP | ATP, 2-meS | CHEMBL336208)Show SMILES CSc1nc(N)c2ncn([C@@H]3O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]3O)c2n1 Show InChI InChI=1S/C11H18N5O13P3S/c1-33-11-14-8(12)5-9(15-11)16(3-13-5)10-7(18)6(17)4(27-10)2-26-31(22,23)29-32(24,25)28-30(19,20)21/h3-4,6-7,10,17-18H,2H2,1H3,(H,22,23)(H,24,25)(H2,12,14,15)(H2,19,20,21)/t4-,6-,7-,10-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| PDB
PubMed
| n/a | n/a | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
Antagonist activity against phospholipase C coupled rat P2Y purinoceptor 12 (P2Y12) |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |