 Found 16 hits in this display
Found 16 hits in this display Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
5-hydroxytryptamine receptor 6
(Homo sapiens (Human)) | BDBM21358
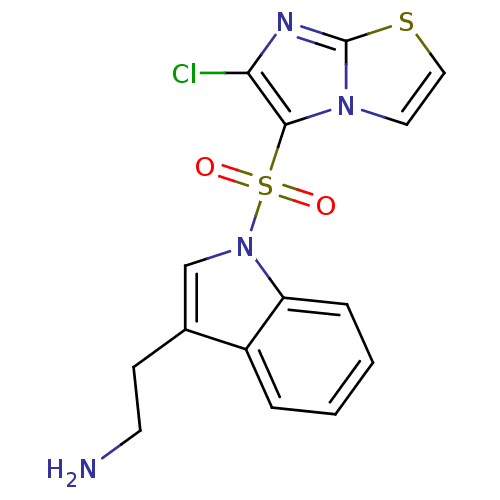
(2-[1-({6-chloroimidazo[2,1-b][1,3]thiazole-5-}sulf...)Show SMILES NCCc1cn(c2ccccc12)S(=O)(=O)c1c(Cl)nc2sccn12 Show InChI InChI=1S/C15H13ClN4O2S2/c16-13-14(19-7-8-23-15(19)18-13)24(21,22)20-9-10(5-6-17)11-3-1-2-4-12(11)20/h1-4,7-9H,5-6,17H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2 | -49.2 | n/a | n/a | 6.5 | n/a | n/a | 7.4 | 22 |
Wyeth Research
| Assay Description
IC50 values for each test compound were determined from nonlinear regression analysis of data collected from ligand binding experiments. The inhibiti... |
J Med Chem 50: 5535-8 (2007)
Article DOI: 10.1021/jm070521y
BindingDB Entry DOI: 10.7270/Q23R0R5G |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 6
(Homo sapiens (Human)) | BDBM21358

(2-[1-({6-chloroimidazo[2,1-b][1,3]thiazole-5-}sulf...)Show SMILES NCCc1cn(c2ccccc12)S(=O)(=O)c1c(Cl)nc2sccn12 Show InChI InChI=1S/C15H13ClN4O2S2/c16-13-14(19-7-8-23-15(19)18-13)24(21,22)20-9-10(5-6-17)11-3-1-2-4-12(11)20/h1-4,7-9H,5-6,17H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona
Curated by ChEMBL
| Assay Description
Displacement of [3H]LSD from human 5HT6 receptor expressed in HEK293 cells by liquid scintillation counting |
J Med Chem 52: 675-87 (2009)
Article DOI: 10.1021/jm8009469
BindingDB Entry DOI: 10.7270/Q2SN09V7 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 6
(Homo sapiens (Human)) | BDBM21358

(2-[1-({6-chloroimidazo[2,1-b][1,3]thiazole-5-}sulf...)Show SMILES NCCc1cn(c2ccccc12)S(=O)(=O)c1c(Cl)nc2sccn12 Show InChI InChI=1S/C15H13ClN4O2S2/c16-13-14(19-7-8-23-15(19)18-13)24(21,22)20-9-10(5-6-17)11-3-1-2-4-12(11)20/h1-4,7-9H,5-6,17H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona
Curated by ChEMBL
| Assay Description
Displacement of [3H]LSD from human cloned 5HT6 receptor expressed in human HeLa cells |
J Med Chem 52: 6153-7 (2009)
Article DOI: 10.1021/jm900796p
BindingDB Entry DOI: 10.7270/Q2BG2P3X |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 6
(Homo sapiens (Human)) | BDBM21358

(2-[1-({6-chloroimidazo[2,1-b][1,3]thiazole-5-}sulf...)Show SMILES NCCc1cn(c2ccccc12)S(=O)(=O)c1c(Cl)nc2sccn12 Show InChI InChI=1S/C15H13ClN4O2S2/c16-13-14(19-7-8-23-15(19)18-13)24(21,22)20-9-10(5-6-17)11-3-1-2-4-12(11)20/h1-4,7-9H,5-6,17H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Caen Basse-Normandie
Curated by ChEMBL
| Assay Description
Displacement of [3H]lysergic acid diethylamide from human 5-HT6 receptor expressed in HeLa cells after 120 mins by scintillation counter |
J Med Chem 58: 7901-12 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00179
BindingDB Entry DOI: 10.7270/Q2M90BGQ |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 6
(Homo sapiens (Human)) | BDBM21358

(2-[1-({6-chloroimidazo[2,1-b][1,3]thiazole-5-}sulf...)Show SMILES NCCc1cn(c2ccccc12)S(=O)(=O)c1c(Cl)nc2sccn12 Show InChI InChI=1S/C15H13ClN4O2S2/c16-13-14(19-7-8-23-15(19)18-13)24(21,22)20-9-10(5-6-17)11-3-1-2-4-12(11)20/h1-4,7-9H,5-6,17H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2 | -51.6 | n/a | n/a | 6.5 | n/a | n/a | 7.4 | 37 |
Universitat de Barcelona
| Assay Description
Radioligand binding assays were performed using membranes from HEK-293 transfected with human 5-HT6 receptor. In these membranes the receptor concent... |
Bioorg Med Chem 17: 7387-97 (2009)
Article DOI: 10.1016/j.bmc.2009.08.006
BindingDB Entry DOI: 10.7270/Q2RR1WKS |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 6
(Homo sapiens (Human)) | BDBM21358

(2-[1-({6-chloroimidazo[2,1-b][1,3]thiazole-5-}sulf...)Show SMILES NCCc1cn(c2ccccc12)S(=O)(=O)c1c(Cl)nc2sccn12 Show InChI InChI=1S/C15H13ClN4O2S2/c16-13-14(19-7-8-23-15(19)18-13)24(21,22)20-9-10(5-6-17)11-3-1-2-4-12(11)20/h1-4,7-9H,5-6,17H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2021.128275
BindingDB Entry DOI: 10.7270/Q2VQ36SJ |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2C
(Homo sapiens (Human)) | BDBM21358

(2-[1-({6-chloroimidazo[2,1-b][1,3]thiazole-5-}sulf...)Show SMILES NCCc1cn(c2ccccc12)S(=O)(=O)c1c(Cl)nc2sccn12 Show InChI InChI=1S/C15H13ClN4O2S2/c16-13-14(19-7-8-23-15(19)18-13)24(21,22)20-9-10(5-6-17)11-3-1-2-4-12(11)20/h1-4,7-9H,5-6,17H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 124 | -39.0 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Wyeth Research
| Assay Description
IC50 values for each test compound were determined from nonlinear regression analysis of data collected from ligand binding experiments. The inhibiti... |
J Med Chem 50: 5535-8 (2007)
Article DOI: 10.1021/jm070521y
BindingDB Entry DOI: 10.7270/Q23R0R5G |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2B
(Homo sapiens (Human)) | BDBM21358

(2-[1-({6-chloroimidazo[2,1-b][1,3]thiazole-5-}sulf...)Show SMILES NCCc1cn(c2ccccc12)S(=O)(=O)c1c(Cl)nc2sccn12 Show InChI InChI=1S/C15H13ClN4O2S2/c16-13-14(19-7-8-23-15(19)18-13)24(21,22)20-9-10(5-6-17)11-3-1-2-4-12(11)20/h1-4,7-9H,5-6,17H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 458 | -35.8 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Wyeth Research
| Assay Description
IC50 values for each test compound were determined from nonlinear regression analysis of data collected from ligand binding experiments. The inhibiti... |
J Med Chem 50: 5535-8 (2007)
Article DOI: 10.1021/jm070521y
BindingDB Entry DOI: 10.7270/Q23R0R5G |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 7
(Homo sapiens (Human)) | BDBM21358

(2-[1-({6-chloroimidazo[2,1-b][1,3]thiazole-5-}sulf...)Show SMILES NCCc1cn(c2ccccc12)S(=O)(=O)c1c(Cl)nc2sccn12 Show InChI InChI=1S/C15H13ClN4O2S2/c16-13-14(19-7-8-23-15(19)18-13)24(21,22)20-9-10(5-6-17)11-3-1-2-4-12(11)20/h1-4,7-9H,5-6,17H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 679 | -34.9 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Wyeth Research
| Assay Description
IC50 values for each test compound were determined from nonlinear regression analysis of data collected from ligand binding experiments. The inhibiti... |
J Med Chem 50: 5535-8 (2007)
Article DOI: 10.1021/jm070521y
BindingDB Entry DOI: 10.7270/Q23R0R5G |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM21358

(2-[1-({6-chloroimidazo[2,1-b][1,3]thiazole-5-}sulf...)Show SMILES NCCc1cn(c2ccccc12)S(=O)(=O)c1c(Cl)nc2sccn12 Show InChI InChI=1S/C15H13ClN4O2S2/c16-13-14(19-7-8-23-15(19)18-13)24(21,22)20-9-10(5-6-17)11-3-1-2-4-12(11)20/h1-4,7-9H,5-6,17H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.30E+4 | n/a | n/a | n/a | n/a | 7.4 | 37 |
Wyeth Research
| Assay Description
The effect of compound on cytochrome P450 (CYP) enzyme catalytic activity was determined in human liver microsomes, using a cocktail of probe substra... |
J Med Chem 50: 5535-8 (2007)
Article DOI: 10.1021/jm070521y
BindingDB Entry DOI: 10.7270/Q23R0R5G |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM21358

(2-[1-({6-chloroimidazo[2,1-b][1,3]thiazole-5-}sulf...)Show SMILES NCCc1cn(c2ccccc12)S(=O)(=O)c1c(Cl)nc2sccn12 Show InChI InChI=1S/C15H13ClN4O2S2/c16-13-14(19-7-8-23-15(19)18-13)24(21,22)20-9-10(5-6-17)11-3-1-2-4-12(11)20/h1-4,7-9H,5-6,17H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.10E+4 | n/a | n/a | n/a | n/a | 7.4 | 37 |
Wyeth Research
| Assay Description
The effect of compound on cytochrome P450 (CYP) enzyme catalytic activity was determined in human liver microsomes, using a cocktail of probe substra... |
J Med Chem 50: 5535-8 (2007)
Article DOI: 10.1021/jm070521y
BindingDB Entry DOI: 10.7270/Q23R0R5G |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM21358

(2-[1-({6-chloroimidazo[2,1-b][1,3]thiazole-5-}sulf...)Show SMILES NCCc1cn(c2ccccc12)S(=O)(=O)c1c(Cl)nc2sccn12 Show InChI InChI=1S/C15H13ClN4O2S2/c16-13-14(19-7-8-23-15(19)18-13)24(21,22)20-9-10(5-6-17)11-3-1-2-4-12(11)20/h1-4,7-9H,5-6,17H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.12E+5 | n/a | n/a | n/a | n/a | 7.4 | 37 |
Wyeth Research
| Assay Description
The effect of compound on cytochrome P450 (CYP) enzyme catalytic activity was determined in human liver microsomes, using a cocktail of probe substra... |
J Med Chem 50: 5535-8 (2007)
Article DOI: 10.1021/jm070521y
BindingDB Entry DOI: 10.7270/Q23R0R5G |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM21358

(2-[1-({6-chloroimidazo[2,1-b][1,3]thiazole-5-}sulf...)Show SMILES NCCc1cn(c2ccccc12)S(=O)(=O)c1c(Cl)nc2sccn12 Show InChI InChI=1S/C15H13ClN4O2S2/c16-13-14(19-7-8-23-15(19)18-13)24(21,22)20-9-10(5-6-17)11-3-1-2-4-12(11)20/h1-4,7-9H,5-6,17H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.49E+5 | n/a | n/a | n/a | n/a | 7.4 | 37 |
Wyeth Research
| Assay Description
The effect of compound on cytochrome P450 (CYP) enzyme catalytic activity was determined in human liver microsomes, using a cocktail of probe substra... |
J Med Chem 50: 5535-8 (2007)
Article DOI: 10.1021/jm070521y
BindingDB Entry DOI: 10.7270/Q23R0R5G |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM21358

(2-[1-({6-chloroimidazo[2,1-b][1,3]thiazole-5-}sulf...)Show SMILES NCCc1cn(c2ccccc12)S(=O)(=O)c1c(Cl)nc2sccn12 Show InChI InChI=1S/C15H13ClN4O2S2/c16-13-14(19-7-8-23-15(19)18-13)24(21,22)20-9-10(5-6-17)11-3-1-2-4-12(11)20/h1-4,7-9H,5-6,17H2 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.96E+5 | n/a | n/a | n/a | n/a | 7.4 | 37 |
Wyeth Research
| Assay Description
The effect of compound on cytochrome P450 (CYP) enzyme catalytic activity was determined in human liver microsomes, using a cocktail of probe substra... |
J Med Chem 50: 5535-8 (2007)
Article DOI: 10.1021/jm070521y
BindingDB Entry DOI: 10.7270/Q23R0R5G |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM21358

(2-[1-({6-chloroimidazo[2,1-b][1,3]thiazole-5-}sulf...)Show SMILES NCCc1cn(c2ccccc12)S(=O)(=O)c1c(Cl)nc2sccn12 Show InChI InChI=1S/C15H13ClN4O2S2/c16-13-14(19-7-8-23-15(19)18-13)24(21,22)20-9-10(5-6-17)11-3-1-2-4-12(11)20/h1-4,7-9H,5-6,17H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+5 | n/a | n/a | n/a | n/a | 7.4 | 37 |
Wyeth Research
| Assay Description
The effect of compound on cytochrome P450 (CYP) enzyme catalytic activity was determined in human liver microsomes, using a cocktail of probe substra... |
J Med Chem 50: 5535-8 (2007)
Article DOI: 10.1021/jm070521y
BindingDB Entry DOI: 10.7270/Q23R0R5G |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 6
(Homo sapiens (Human)) | BDBM21358

(2-[1-({6-chloroimidazo[2,1-b][1,3]thiazole-5-}sulf...)Show SMILES NCCc1cn(c2ccccc12)S(=O)(=O)c1c(Cl)nc2sccn12 Show InChI InChI=1S/C15H13ClN4O2S2/c16-13-14(19-7-8-23-15(19)18-13)24(21,22)20-9-10(5-6-17)11-3-1-2-4-12(11)20/h1-4,7-9H,5-6,17H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 6.5 | n/a | n/a | n/a | n/a |
Universitat de Barcelona
Curated by ChEMBL
| Assay Description
Agonist activity at human 5HT6 receptor expressed in HeLa cells assessed as induction of cAMP production after 10 mins by radioimmunoassay |
J Med Chem 52: 6153-7 (2009)
Article DOI: 10.1021/jm900796p
BindingDB Entry DOI: 10.7270/Q2BG2P3X |
More data for this
Ligand-Target Pair | |