 Found 13 hits in this display
Found 13 hits in this display Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Calmodulin-domain protein kinase 1
(Toxoplasma gondii) | BDBM50389688
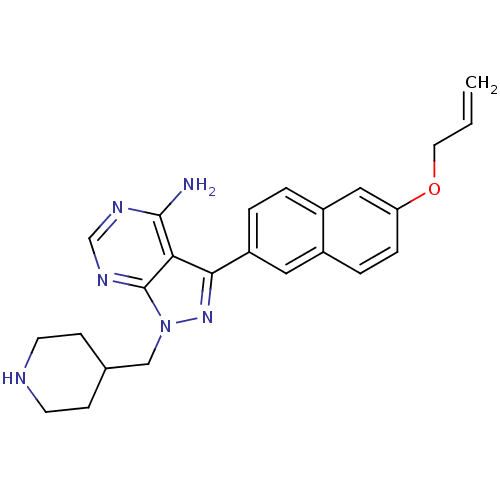
(CHEMBL2069315 | US10544104, Compound 36 | US112479...)Show SMILES Nc1ncnc2n(CC3CCNCC3)nc(-c3ccc4cc(OCC=C)ccc4c3)c12 Show InChI InChI=1S/C24H26N6O/c1-2-11-31-20-6-5-17-12-19(4-3-18(17)13-20)22-21-23(25)27-15-28-24(21)30(29-22)14-16-7-9-26-10-8-16/h2-6,12-13,15-16,26H,1,7-11,14H2,(H2,25,27,28) | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
UNIVERSITY OF WASHINGTON THROUGH ITS CENTER FOR CO
US Patent
| Assay Description
Inhibition of TgCDPK1 and CpCDPK1 was determined using a luminescent kinase assay which measures ATP depletion in the presence of the Syntide 2 pepti... |
US Patent US10544104 (2020)
BindingDB Entry DOI: 10.7270/Q2D79DSF |
More data for this
Ligand-Target Pair | |
cGMP-dependent protein kinase
() | BDBM50389688

(CHEMBL2069315 | US10544104, Compound 36 | US112479...)Show SMILES Nc1ncnc2n(CC3CCNCC3)nc(-c3ccc4cc(OCC=C)ccc4c3)c12 Show InChI InChI=1S/C24H26N6O/c1-2-11-31-20-6-5-17-12-19(4-3-18(17)13-20)22-21-23(25)27-15-28-24(21)30(29-22)14-16-7-9-26-10-8-16/h2-6,12-13,15-16,26H,1,7-11,14H2,(H2,25,27,28) | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of TgCDPK1 and CpCDPK1 was determined using a luminescent kinase assay which measures ATP depletion in the presence of the Syntide 2 pepti... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2K077GQ |
More data for this
Ligand-Target Pair | |
Calmodulin-domain protein kinase 1
(Toxoplasma gondii) | BDBM50389688

(CHEMBL2069315 | US10544104, Compound 36 | US112479...)Show SMILES Nc1ncnc2n(CC3CCNCC3)nc(-c3ccc4cc(OCC=C)ccc4c3)c12 Show InChI InChI=1S/C24H26N6O/c1-2-11-31-20-6-5-17-12-19(4-3-18(17)13-20)22-21-23(25)27-15-28-24(21)30(29-22)14-16-7-9-26-10-8-16/h2-6,12-13,15-16,26H,1,7-11,14H2,(H2,25,27,28) | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Washington Through its Center for Commercialization
US Patent
| Assay Description
Most known kinase inhibitors bind in the ATP-binding pocket of the active site19,20. These inhibitors exploit many of the same hydrophobic contacts a... |
US Patent US9765037 (2017)
BindingDB Entry DOI: 10.7270/Q2B56MVC |
More data for this
Ligand-Target Pair | |
Calmodulin-domain protein kinase 1, putative
(Cryptosporidium parvum (strain Iowa II)) | BDBM50389688

(CHEMBL2069315 | US10544104, Compound 36 | US112479...)Show SMILES Nc1ncnc2n(CC3CCNCC3)nc(-c3ccc4cc(OCC=C)ccc4c3)c12 Show InChI InChI=1S/C24H26N6O/c1-2-11-31-20-6-5-17-12-19(4-3-18(17)13-20)22-21-23(25)27-15-28-24(21)30(29-22)14-16-7-9-26-10-8-16/h2-6,12-13,15-16,26H,1,7-11,14H2,(H2,25,27,28) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a |
UNIVERSITY OF WASHINGTON THROUGH ITS CENTER FOR CO
US Patent
| Assay Description
Inhibition of TgCDPK1 and CpCDPK1 was determined using a luminescent kinase assay which measures ATP depletion in the presence of the Syntide 2 pepti... |
US Patent US10544104 (2020)
BindingDB Entry DOI: 10.7270/Q2D79DSF |
More data for this
Ligand-Target Pair | |
Calcium-dependent protein kinase 1
(Cryptosporidium parvum) | BDBM50389688

(CHEMBL2069315 | US10544104, Compound 36 | US112479...)Show SMILES Nc1ncnc2n(CC3CCNCC3)nc(-c3ccc4cc(OCC=C)ccc4c3)c12 Show InChI InChI=1S/C24H26N6O/c1-2-11-31-20-6-5-17-12-19(4-3-18(17)13-20)22-21-23(25)27-15-28-24(21)30(29-22)14-16-7-9-26-10-8-16/h2-6,12-13,15-16,26H,1,7-11,14H2,(H2,25,27,28) | UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Washington Through its Center for Commercialization
US Patent
| Assay Description
Two types of enzyme assays were developed to follow TgCDPK1 activity, a radiometric scintillation proximity assay measured the labeled γ-phospha... |
US Patent US9765037 (2017)
BindingDB Entry DOI: 10.7270/Q2B56MVC |
More data for this
Ligand-Target Pair | |
Calcium/calmodulin dependent protein kinase with a kinas domain and 4 calmodulin-like EF hands
(Cryptosporidium parvum (strain Iowa II)) | BDBM50389688

(CHEMBL2069315 | US10544104, Compound 36 | US112479...)Show SMILES Nc1ncnc2n(CC3CCNCC3)nc(-c3ccc4cc(OCC=C)ccc4c3)c12 Show InChI InChI=1S/C24H26N6O/c1-2-11-31-20-6-5-17-12-19(4-3-18(17)13-20)22-21-23(25)27-15-28-24(21)30(29-22)14-16-7-9-26-10-8-16/h2-6,12-13,15-16,26H,1,7-11,14H2,(H2,25,27,28) | UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of TgCDPK1 and CpCDPK1 was determined using a luminescent kinase assay which measures ATP depletion in the presence of the Syntide 2 pepti... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2K077GQ |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase HCK
(Homo sapiens (Human)) | BDBM50389688

(CHEMBL2069315 | US10544104, Compound 36 | US112479...)Show SMILES Nc1ncnc2n(CC3CCNCC3)nc(-c3ccc4cc(OCC=C)ccc4c3)c12 Show InChI InChI=1S/C24H26N6O/c1-2-11-31-20-6-5-17-12-19(4-3-18(17)13-20)22-21-23(25)27-15-28-24(21)30(29-22)14-16-7-9-26-10-8-16/h2-6,12-13,15-16,26H,1,7-11,14H2,(H2,25,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
UNIVERSITY OF WASHINGTON THROUGH ITS CENTER FOR CO
US Patent
| Assay Description
Inhibition of TgCDPK1 and CpCDPK1 was determined using a luminescent kinase assay which measures ATP depletion in the presence of the Syntide 2 pepti... |
US Patent US10544104 (2020)
BindingDB Entry DOI: 10.7270/Q2D79DSF |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase Src
(Homo sapiens (Human)) | BDBM50389688

(CHEMBL2069315 | US10544104, Compound 36 | US112479...)Show SMILES Nc1ncnc2n(CC3CCNCC3)nc(-c3ccc4cc(OCC=C)ccc4c3)c12 Show InChI InChI=1S/C24H26N6O/c1-2-11-31-20-6-5-17-12-19(4-3-18(17)13-20)22-21-23(25)27-15-28-24(21)30(29-22)14-16-7-9-26-10-8-16/h2-6,12-13,15-16,26H,1,7-11,14H2,(H2,25,27,28) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Washington
Curated by ChEMBL
| Assay Description
Inhibition of human SRC using Ac-EIYGEFKKK-OH as substrate after 60 mins by phosphorimaging method |
J Med Chem 55: 2416-26 (2012)
Article DOI: 10.1021/jm201713h
BindingDB Entry DOI: 10.7270/Q2P2706V |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase Src
(Homo sapiens (Human)) | BDBM50389688

(CHEMBL2069315 | US10544104, Compound 36 | US112479...)Show SMILES Nc1ncnc2n(CC3CCNCC3)nc(-c3ccc4cc(OCC=C)ccc4c3)c12 Show InChI InChI=1S/C24H26N6O/c1-2-11-31-20-6-5-17-12-19(4-3-18(17)13-20)22-21-23(25)27-15-28-24(21)30(29-22)14-16-7-9-26-10-8-16/h2-6,12-13,15-16,26H,1,7-11,14H2,(H2,25,27,28) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
UNIVERSITY OF WASHINGTON THROUGH ITS CENTER FOR CO
US Patent
| Assay Description
Inhibition of TgCDPK1 and CpCDPK1 was determined using a luminescent kinase assay which measures ATP depletion in the presence of the Syntide 2 pepti... |
US Patent US10544104 (2020)
BindingDB Entry DOI: 10.7270/Q2D79DSF |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL1
(Homo sapiens (Human)) | BDBM50389688

(CHEMBL2069315 | US10544104, Compound 36 | US112479...)Show SMILES Nc1ncnc2n(CC3CCNCC3)nc(-c3ccc4cc(OCC=C)ccc4c3)c12 Show InChI InChI=1S/C24H26N6O/c1-2-11-31-20-6-5-17-12-19(4-3-18(17)13-20)22-21-23(25)27-15-28-24(21)30(29-22)14-16-7-9-26-10-8-16/h2-6,12-13,15-16,26H,1,7-11,14H2,(H2,25,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
UNIVERSITY OF WASHINGTON THROUGH ITS CENTER FOR CO
US Patent
| Assay Description
Inhibition of TgCDPK1 and CpCDPK1 was determined using a luminescent kinase assay which measures ATP depletion in the presence of the Syntide 2 pepti... |
US Patent US10544104 (2020)
BindingDB Entry DOI: 10.7270/Q2D79DSF |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase HCK
(Homo sapiens (Human)) | BDBM50389688

(CHEMBL2069315 | US10544104, Compound 36 | US112479...)Show SMILES Nc1ncnc2n(CC3CCNCC3)nc(-c3ccc4cc(OCC=C)ccc4c3)c12 Show InChI InChI=1S/C24H26N6O/c1-2-11-31-20-6-5-17-12-19(4-3-18(17)13-20)22-21-23(25)27-15-28-24(21)30(29-22)14-16-7-9-26-10-8-16/h2-6,12-13,15-16,26H,1,7-11,14H2,(H2,25,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Washington Through its Center for Commercialization
US Patent
| Assay Description
Inhibition of human tyrosine kinases. |
US Patent US9765037 (2017)
BindingDB Entry DOI: 10.7270/Q2B56MVC |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL1
(Homo sapiens (Human)) | BDBM50389688

(CHEMBL2069315 | US10544104, Compound 36 | US112479...)Show SMILES Nc1ncnc2n(CC3CCNCC3)nc(-c3ccc4cc(OCC=C)ccc4c3)c12 Show InChI InChI=1S/C24H26N6O/c1-2-11-31-20-6-5-17-12-19(4-3-18(17)13-20)22-21-23(25)27-15-28-24(21)30(29-22)14-16-7-9-26-10-8-16/h2-6,12-13,15-16,26H,1,7-11,14H2,(H2,25,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Washington Through its Center for Commercialization
US Patent
| Assay Description
Inhibition of human tyrosine kinases. |
US Patent US9765037 (2017)
BindingDB Entry DOI: 10.7270/Q2B56MVC |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase Src
(Homo sapiens (Human)) | BDBM50389688

(CHEMBL2069315 | US10544104, Compound 36 | US112479...)Show SMILES Nc1ncnc2n(CC3CCNCC3)nc(-c3ccc4cc(OCC=C)ccc4c3)c12 Show InChI InChI=1S/C24H26N6O/c1-2-11-31-20-6-5-17-12-19(4-3-18(17)13-20)22-21-23(25)27-15-28-24(21)30(29-22)14-16-7-9-26-10-8-16/h2-6,12-13,15-16,26H,1,7-11,14H2,(H2,25,27,28) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Washington Through its Center for Commercialization
US Patent
| Assay Description
Inhibition of human tyrosine kinases. |
US Patent US9765037 (2017)
BindingDB Entry DOI: 10.7270/Q2B56MVC |
More data for this
Ligand-Target Pair | |