 Found 12 hits in this display
Found 12 hits in this display Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
S-adenosylmethionine synthase isoform type-2
(Rattus norvegicus) | BDBM50118221
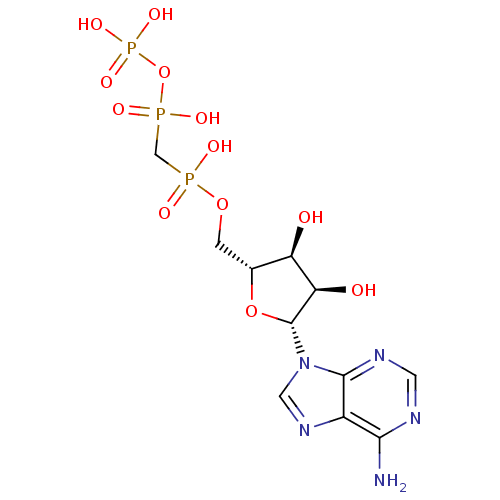
(9H-purine derivative | CHEMBL132722 | DIPHOSPHOMET...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)CP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O Show InChI InChI=1S/C11H18N5O12P3/c12-9-6-10(14-2-13-9)16(3-15-6)11-8(18)7(17)5(27-11)1-26-29(19,20)4-30(21,22)28-31(23,24)25/h2-3,5,7-8,11,17-18H,1,4H2,(H,19,20)(H,21,22)(H2,12,13,14)(H2,23,24,25)/t5-,7-,8-,11-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Similars
| PubMed
| 3.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory constant against rat kidney Methionine adenosyltransferase II |
J Med Chem 29: 318-22 (1986)
BindingDB Entry DOI: 10.7270/Q2DF6RSH |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-1/type-2
(Rattus norvegicus) | BDBM50118221

(9H-purine derivative | CHEMBL132722 | DIPHOSPHOMET...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)CP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O Show InChI InChI=1S/C11H18N5O12P3/c12-9-6-10(14-2-13-9)16(3-15-6)11-8(18)7(17)5(27-11)1-26-29(19,20)4-30(21,22)28-31(23,24)25/h2-3,5,7-8,11,17-18H,1,4H2,(H,19,20)(H,21,22)(H2,12,13,14)(H2,23,24,25)/t5-,7-,8-,11-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Similars
| PubMed
| 3.40E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory constant against rat Methionine adenosyltransferase was reported |
J Med Chem 29: 318-22 (1986)
BindingDB Entry DOI: 10.7270/Q2DF6RSH |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 1
(RAT) | BDBM50118221

(9H-purine derivative | CHEMBL132722 | DIPHOSPHOMET...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)CP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O Show InChI InChI=1S/C11H18N5O12P3/c12-9-6-10(14-2-13-9)16(3-15-6)11-8(18)7(17)5(27-11)1-26-29(19,20)4-30(21,22)28-31(23,24)25/h2-3,5,7-8,11,17-18H,1,4H2,(H,19,20)(H,21,22)(H2,12,13,14)(H2,23,24,25)/t5-,7-,8-,11-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Urbino Carlo Bo
Curated by ChEMBL
| Assay Description
Inhibitory concentration against human Adenosine A3 receptor expressed in HEK293 cells using 0.1 nM [3H]AB-MECA |
J Med Chem 48: 6887-96 (2005)
Article DOI: 10.1021/jm058018d
BindingDB Entry DOI: 10.7270/Q28C9X1X |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 1
(Homo sapiens (Human)) | BDBM50118221

(9H-purine derivative | CHEMBL132722 | DIPHOSPHOMET...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)CP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O Show InChI InChI=1S/C11H18N5O12P3/c12-9-6-10(14-2-13-9)16(3-15-6)11-8(18)7(17)5(27-11)1-26-29(19,20)4-30(21,22)28-31(23,24)25/h2-3,5,7-8,11,17-18H,1,4H2,(H,19,20)(H,21,22)(H2,12,13,14)(H2,23,24,25)/t5-,7-,8-,11-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 200 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
The compound was evaluated for antagonist activity against recombinant human P2X purinoceptor 1 (P2X1 ) |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 5
(Rattus norvegicus) | BDBM50118221

(9H-purine derivative | CHEMBL132722 | DIPHOSPHOMET...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)CP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O Show InChI InChI=1S/C11H18N5O12P3/c12-9-6-10(14-2-13-9)16(3-15-6)11-8(18)7(17)5(27-11)1-26-29(19,20)4-30(21,22)28-31(23,24)25/h2-3,5,7-8,11,17-18H,1,4H2,(H,19,20)(H,21,22)(H2,12,13,14)(H2,23,24,25)/t5-,7-,8-,11-/m1/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | >7.00E+3 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
Antagonist activity against recombinant rat P2X purinoceptor 5 (P2X5) |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 2
(RAT) | BDBM50118221

(9H-purine derivative | CHEMBL132722 | DIPHOSPHOMET...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)CP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O Show InChI InChI=1S/C11H18N5O12P3/c12-9-6-10(14-2-13-9)16(3-15-6)11-8(18)7(17)5(27-11)1-26-29(19,20)4-30(21,22)28-31(23,24)25/h2-3,5,7-8,11,17-18H,1,4H2,(H,19,20)(H,21,22)(H2,12,13,14)(H2,23,24,25)/t5-,7-,8-,11-/m1/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 1.00E+5 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
The compound was evaluated for antagonist activity against recombinant rat receptor P2X purinoceptor 2 (P2X2) at 10 uM, expressed in Xenopus oocytes |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 3
(Homo sapiens (Human)) | BDBM50118221

(9H-purine derivative | CHEMBL132722 | DIPHOSPHOMET...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)CP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O Show InChI InChI=1S/C11H18N5O12P3/c12-9-6-10(14-2-13-9)16(3-15-6)11-8(18)7(17)5(27-11)1-26-29(19,20)4-30(21,22)28-31(23,24)25/h2-3,5,7-8,11,17-18H,1,4H2,(H,19,20)(H,21,22)(H2,12,13,14)(H2,23,24,25)/t5-,7-,8-,11-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 740 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
The compound was evaluated for antagonist activity against recombinant rat P2X purinoceptor 3 (P2X3) at 10 uM, expressed in Xenopus oocytes |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 4
(RAT) | BDBM50118221

(9H-purine derivative | CHEMBL132722 | DIPHOSPHOMET...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)CP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O Show InChI InChI=1S/C11H18N5O12P3/c12-9-6-10(14-2-13-9)16(3-15-6)11-8(18)7(17)5(27-11)1-26-29(19,20)4-30(21,22)28-31(23,24)25/h2-3,5,7-8,11,17-18H,1,4H2,(H,19,20)(H,21,22)(H2,12,13,14)(H2,23,24,25)/t5-,7-,8-,11-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 3.30E+4 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
The compound was evaluated for antagonist activity against recombinant rat P2X purinoceptor 4 (P2X4) 3 uM |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2Y purinoceptor 12
(Rattus norvegicus) | BDBM50118221

(9H-purine derivative | CHEMBL132722 | DIPHOSPHOMET...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)CP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O Show InChI InChI=1S/C11H18N5O12P3/c12-9-6-10(14-2-13-9)16(3-15-6)11-8(18)7(17)5(27-11)1-26-29(19,20)4-30(21,22)28-31(23,24)25/h2-3,5,7-8,11,17-18H,1,4H2,(H,19,20)(H,21,22)(H2,12,13,14)(H2,23,24,25)/t5-,7-,8-,11-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
Antagonist activity against phospholipase C coupled rat P2Y purinoceptor 12 (P2Y12) |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 7
(Homo sapiens (Human)) | BDBM50118221

(9H-purine derivative | CHEMBL132722 | DIPHOSPHOMET...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)CP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O Show InChI InChI=1S/C11H18N5O12P3/c12-9-6-10(14-2-13-9)16(3-15-6)11-8(18)7(17)5(27-11)1-26-29(19,20)4-30(21,22)28-31(23,24)25/h2-3,5,7-8,11,17-18H,1,4H2,(H,19,20)(H,21,22)(H2,12,13,14)(H2,23,24,25)/t5-,7-,8-,11-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
Antagonist activity against recombinant human P2X purinoceptor 7 (P2X7) |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2Y purinoceptor 1
(Meleagris gallopavo) | BDBM50118221

(9H-purine derivative | CHEMBL132722 | DIPHOSPHOMET...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)CP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O Show InChI InChI=1S/C11H18N5O12P3/c12-9-6-10(14-2-13-9)16(3-15-6)11-8(18)7(17)5(27-11)1-26-29(19,20)4-30(21,22)28-31(23,24)25/h2-3,5,7-8,11,17-18H,1,4H2,(H,19,20)(H,21,22)(H2,12,13,14)(H2,23,24,25)/t5-,7-,8-,11-/m1/s1 | MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
Evaluated for agonist activity against phospholipase C coupled P2Y purinoceptor 1 (P2Y1) of turkey erythrocytes |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 6
(Rattus norvegicus) | BDBM50118221

(9H-purine derivative | CHEMBL132722 | DIPHOSPHOMET...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)CP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O Show InChI InChI=1S/C11H18N5O12P3/c12-9-6-10(14-2-13-9)16(3-15-6)11-8(18)7(17)5(27-11)1-26-29(19,20)4-30(21,22)28-31(23,24)25/h2-3,5,7-8,11,17-18H,1,4H2,(H,19,20)(H,21,22)(H2,12,13,14)(H2,23,24,25)/t5-,7-,8-,11-/m1/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | >8.00E+3 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
Antagonist activity against recombinant rat P2X purinoceptor 6 (P2X6 ) |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |