 Found 64 hits Enz. Inhib. hit(s) with all data for entry = 50007270
Found 64 hits Enz. Inhib. hit(s) with all data for entry = 50007270 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Non-receptor tyrosine-protein kinase TYK2
(Homo sapiens (Human)) | BDBM50507816
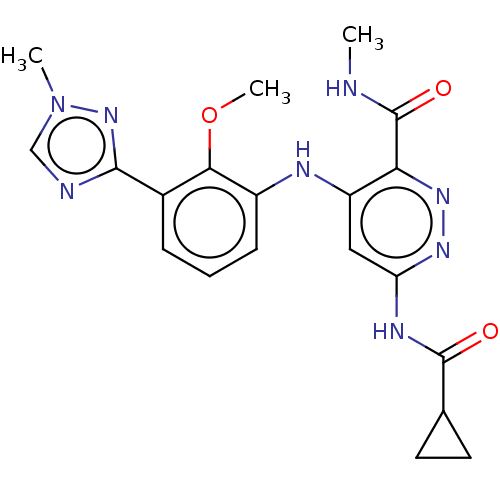
(Bms-986165 | Deucravacitinib)Show SMILES [2H]C([2H])([2H])NC(=O)c1nnc(NC(=O)C2CC2)cc1Nc1cccc(-c2ncn(C)n2)c1OC Show InChI InChI=1S/C20H22N8O3/c1-21-20(30)16-14(9-15(25-26-16)24-19(29)11-7-8-11)23-13-6-4-5-12(17(13)31-3)18-22-10-28(2)27-18/h4-6,9-11H,7-8H2,1-3H3,(H,21,30)(H2,23,24,25,29) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of TYK2 in human Jurkat cells assessed as reduction in IFN-alpha stimulated TYK2 phosphorylation by caspase3/7 reagent-based Western blot ... |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-1
(Homo sapiens (Human)) | BDBM106870
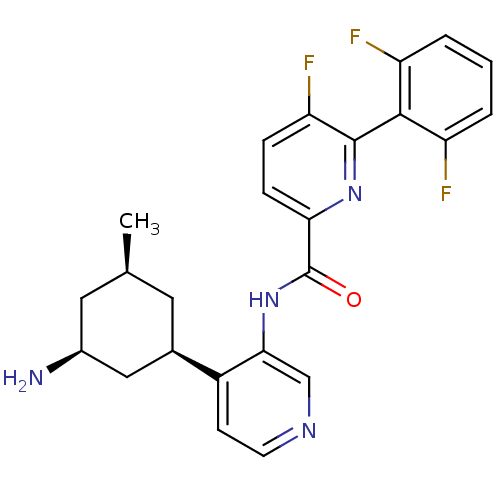
(US8592455, 70)Show SMILES C[C@@H]1C[C@H](N)C[C@@H](C1)c1ccncc1NC(=O)c1ccc(F)c(n1)-c1c(F)cccc1F |r,wU:6.8,3.3,1.0,(-.67,2.69,;-2,1.93,;-3.33,2.69,;-4.67,1.93,;-6,2.69,;-4.67,.38,;-3.33,-.38,;-2,.38,;-3.33,-1.93,;-4.67,-2.69,;-4.67,-4.23,;-3.33,-5,;-2,-4.23,;-2,-2.69,;-.67,-1.93,;.67,-2.69,;.67,-4.23,;2,-1.93,;3.33,-2.69,;4.67,-1.93,;4.67,-.38,;6,.38,;3.33,.38,;2,-.38,;3.33,1.93,;4.67,2.69,;6,1.93,;4.67,4.23,;3.33,5,;2,4.23,;2,2.69,;.67,1.93,)| Show InChI InChI=1S/C24H23F3N4O/c1-13-9-14(11-15(28)10-13)16-7-8-29-12-21(16)31-24(32)20-6-5-19(27)23(30-20)22-17(25)3-2-4-18(22)26/h2-8,12-15H,9-11,28H2,1H3,(H,31,32)/t13-,14+,15-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.00600 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PIM1 (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-3
(Homo sapiens (Human)) | BDBM106870

(US8592455, 70)Show SMILES C[C@@H]1C[C@H](N)C[C@@H](C1)c1ccncc1NC(=O)c1ccc(F)c(n1)-c1c(F)cccc1F |r,wU:6.8,3.3,1.0,(-.67,2.69,;-2,1.93,;-3.33,2.69,;-4.67,1.93,;-6,2.69,;-4.67,.38,;-3.33,-.38,;-2,.38,;-3.33,-1.93,;-4.67,-2.69,;-4.67,-4.23,;-3.33,-5,;-2,-4.23,;-2,-2.69,;-.67,-1.93,;.67,-2.69,;.67,-4.23,;2,-1.93,;3.33,-2.69,;4.67,-1.93,;4.67,-.38,;6,.38,;3.33,.38,;2,-.38,;3.33,1.93,;4.67,2.69,;6,1.93,;4.67,4.23,;3.33,5,;2,4.23,;2,2.69,;.67,1.93,)| Show InChI InChI=1S/C24H23F3N4O/c1-13-9-14(11-15(28)10-13)16-7-8-29-12-21(16)31-24(32)20-6-5-19(27)23(30-20)22-17(25)3-2-4-18(22)26/h2-8,12-15H,9-11,28H2,1H3,(H,31,32)/t13-,14+,15-/m0/s1 | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.00900 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PIM3 (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-2
(Homo sapiens (Human)) | BDBM106870

(US8592455, 70)Show SMILES C[C@@H]1C[C@H](N)C[C@@H](C1)c1ccncc1NC(=O)c1ccc(F)c(n1)-c1c(F)cccc1F |r,wU:6.8,3.3,1.0,(-.67,2.69,;-2,1.93,;-3.33,2.69,;-4.67,1.93,;-6,2.69,;-4.67,.38,;-3.33,-.38,;-2,.38,;-3.33,-1.93,;-4.67,-2.69,;-4.67,-4.23,;-3.33,-5,;-2,-4.23,;-2,-2.69,;-.67,-1.93,;.67,-2.69,;.67,-4.23,;2,-1.93,;3.33,-2.69,;4.67,-1.93,;4.67,-.38,;6,.38,;3.33,.38,;2,-.38,;3.33,1.93,;4.67,2.69,;6,1.93,;4.67,4.23,;3.33,5,;2,4.23,;2,2.69,;.67,1.93,)| Show InChI InChI=1S/C24H23F3N4O/c1-13-9-14(11-15(28)10-13)16-7-8-29-12-21(16)31-24(32)20-6-5-19(27)23(30-20)22-17(25)3-2-4-18(22)26/h2-8,12-15H,9-11,28H2,1H3,(H,31,32)/t13-,14+,15-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.0180 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PIM2 (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Interleukin-1 receptor-associated kinase 4
(Homo sapiens (Human)) | BDBM319575

(5-(((2S,3S,4S)-3-ethyl-4-fluoro-5-oxopyrrolidin-2-...)Show SMILES CC[C@H]1[C@@H](COc2cccc3cc(C(N)=O)c(OC)cc23)NC(=O)[C@H]1F |r| Show InChI InChI=1S/C19H21FN2O4/c1-3-11-14(22-19(24)17(11)20)9-26-15-6-4-5-10-7-13(18(21)23)16(25-2)8-12(10)15/h4-8,11,14,17H,3,9H2,1-2H3,(H2,21,23)(H,22,24)/t11-,14+,17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of IRAK4 (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50192880
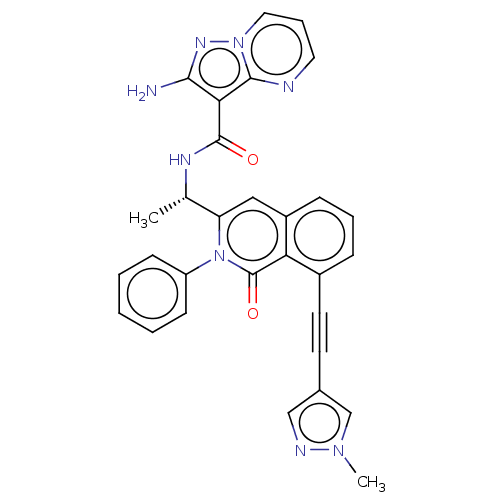
(CHEMBL3984425 | US10329299, Compound 21 | US106752...)Show SMILES C[C@H](NC(=O)c1c(N)nn2cccnc12)c1cc2cccc(C#Cc3cnn(C)c3)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C30H24N8O2/c1-19(34-29(39)26-27(31)35-37-15-7-14-32-28(26)37)24-16-22-9-6-8-21(13-12-20-17-33-36(2)18-20)25(22)30(40)38(24)23-10-4-3-5-11-23/h3-11,14-19H,1-2H3,(H2,31,35)(H,34,39)/t19-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 0.290 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Serine/threonine-protein kinase pim-1
(Homo sapiens (Human)) | BDBM50387298
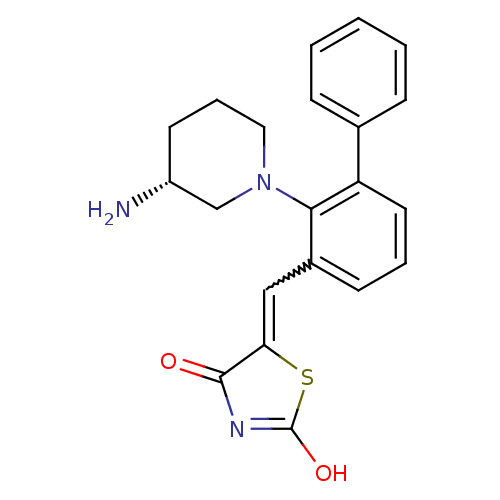
(CHEMBL2048872)Show SMILES N[C@@H]1CCCN(C1)c1c(C=C2SC(O)=NC2=O)cccc1-c1ccccc1 |r,w:9.9,c:14| Show InChI InChI=1S/C21H21N3O2S/c22-16-9-5-11-24(13-16)19-15(12-18-20(25)23-21(26)27-18)8-4-10-17(19)14-6-2-1-3-7-14/h1-4,6-8,10,12,16H,5,9,11,13,22H2,(H,23,25,26)/t16-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PIM1 (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Non-receptor tyrosine-protein kinase TYK2
(Homo sapiens (Human)) | BDBM50507817
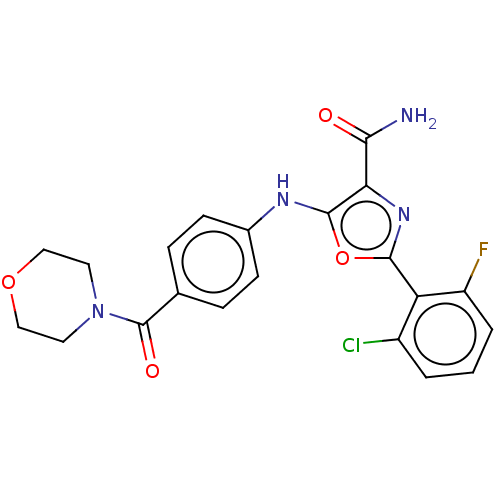
(CHEMBL4476770)Show SMILES NC(=O)c1nc(oc1Nc1ccc(cc1)C(=O)N1CCOCC1)-c1c(F)cccc1Cl Show InChI InChI=1S/C21H18ClFN4O4/c22-14-2-1-3-15(23)16(14)19-26-17(18(24)28)20(31-19)25-13-6-4-12(5-7-13)21(29)27-8-10-30-11-9-27/h1-7,25H,8-11H2,(H2,24,28) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of TYK2 in human Jurkat cells assessed as reduction in IFN-alpha stimulated TYK2 phosphorylation by caspase3/7 reagent-based Western blot ... |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
MAP kinase-interacting serine/threonine-protein kinase 2
(Homo sapiens (Human)) | BDBM50263263

(CHEMBL4073443)Show SMILES Cc1cc(Nc2cc(N)ncn2)c(=O)n2c1C(=O)NC21CCCCC1 Show InChI InChI=1S/C17H20N6O2/c1-10-7-11(21-13-8-12(18)19-9-20-13)16(25)23-14(10)15(24)22-17(23)5-3-2-4-6-17/h7-9H,2-6H2,1H3,(H,22,24)(H3,18,19,20,21) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of MNK2 (unknown origin) expressed in HEK293 cells assessed as decrease in eIF4E phosphorylation at Ser209 residues by Western blot analys... |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Interleukin-1 receptor-associated kinase 4
(Homo sapiens (Human)) | BDBM50040805
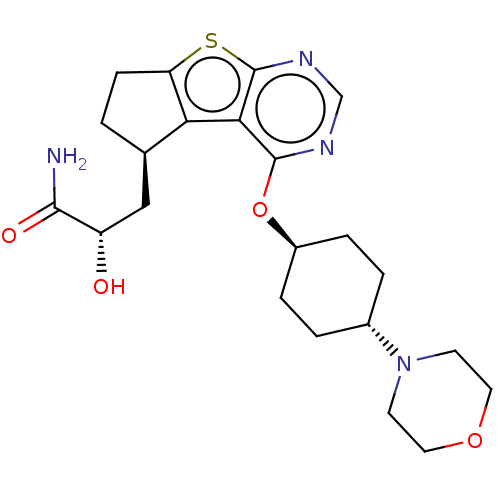
(CHEMBL3361255)Show SMILES NC(=O)[C@@H](O)C[C@H]1CCc2sc3ncnc(O[C@H]4CC[C@@H](CC4)N4CCOCC4)c3c12 |r,wU:20.23,wD:17.16,6.5,3.3,(47.71,-11.7,;46.68,-10.55,;45.18,-10.87,;47.17,-9.09,;48.67,-8.78,;46.14,-7.95,;46.62,-6.48,;48.09,-6.01,;48.1,-4.47,;46.63,-3.99,;45.72,-2.74,;44.25,-3.22,;42.92,-2.45,;41.59,-3.22,;41.59,-4.77,;42.92,-5.54,;42.92,-7.07,;41.59,-7.84,;40.25,-7.07,;38.92,-7.84,;38.92,-9.37,;40.25,-10.15,;41.59,-9.38,;37.58,-10.14,;36.25,-9.35,;34.92,-10.11,;34.91,-11.65,;36.23,-12.43,;37.58,-11.66,;44.25,-4.77,;45.72,-5.23,)| Show InChI InChI=1S/C22H30N4O4S/c23-20(28)16(27)11-13-1-6-17-18(13)19-21(24-12-25-22(19)31-17)30-15-4-2-14(3-5-15)26-7-9-29-10-8-26/h12-16,27H,1-11H2,(H2,23,28)/t13-,14-,15-,16+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of IRAK4 (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Macrophage-stimulating protein receptor
(Homo sapiens (Human)) | BDBM28031
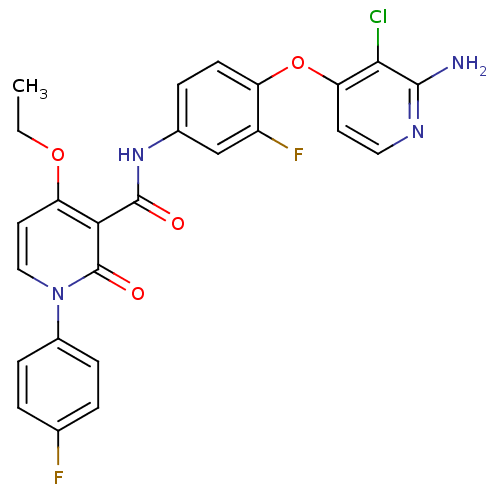
(BMS-777607 | N-{4-[(2-amino-3-chloropyridin-4-yl)o...)Show SMILES CCOc1ccn(-c2ccc(F)cc2)c(=O)c1C(=O)Nc1ccc(Oc2ccnc(N)c2Cl)c(F)c1 Show InChI InChI=1S/C25H19ClF2N4O4/c1-2-35-19-10-12-32(16-6-3-14(27)4-7-16)25(34)21(19)24(33)31-15-5-8-18(17(28)13-15)36-20-9-11-30-23(29)22(20)26/h3-13H,2H2,1H3,(H2,29,30)(H,31,33) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of RON (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-3
(Homo sapiens (Human)) | BDBM50387298

(CHEMBL2048872)Show SMILES N[C@@H]1CCCN(C1)c1c(C=C2SC(O)=NC2=O)cccc1-c1ccccc1 |r,w:9.9,c:14| Show InChI InChI=1S/C21H21N3O2S/c22-16-9-5-11-24(13-16)19-15(12-18-20(25)23-21(26)27-18)8-4-10-17(19)14-6-2-1-3-7-14/h1-4,6-8,10,12,16H,5,9,11,13,22H2,(H,23,25,26)/t16-/m1/s1 | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PIM3 (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
MAP kinase-interacting serine/threonine-protein kinase 1
(Homo sapiens (Human)) | BDBM50263263

(CHEMBL4073443)Show SMILES Cc1cc(Nc2cc(N)ncn2)c(=O)n2c1C(=O)NC21CCCCC1 Show InChI InChI=1S/C17H20N6O2/c1-10-7-11(21-13-8-12(18)19-9-20-13)16(25)23-14(10)15(24)22-17(23)5-3-2-4-6-17/h7-9H,2-6H2,1H3,(H,22,24)(H3,18,19,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of MNK1 (unknown origin) expressed in HEK293 cells assessed as decrease in eIF4E phosphorylation at Ser209 residues by Western blot analys... |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50403068
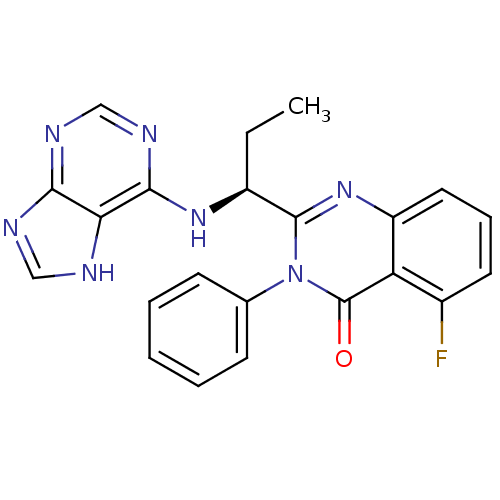
(CHEMBL2216870 | IDELALISIB | US9745321, CAL-101)Show SMILES CC[C@H](Nc1ncnc2nc[nH]c12)c1nc2cccc(F)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C22H18FN7O/c1-2-15(28-20-18-19(25-11-24-18)26-12-27-20)21-29-16-10-6-9-14(23)17(16)22(31)30(21)13-7-4-3-5-8-13/h3-12,15H,2H2,1H3,(H2,24,25,26,27,28)/t15-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50193013

(Duvelisib | INK-1147 | INK-1197 | IPI-145)Show SMILES C[C@H](Nc1ncnc2nc[nH]c12)c1cc2cccc(Cl)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C22H17ClN6O/c1-13(28-21-19-20(25-11-24-19)26-12-27-21)17-10-14-6-5-9-16(23)18(14)22(30)29(17)15-7-3-2-4-8-15/h2-13H,1H3,(H2,24,25,26,27,28)/t13-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-2
(Homo sapiens (Human)) | BDBM50387298

(CHEMBL2048872)Show SMILES N[C@@H]1CCCN(C1)c1c(C=C2SC(O)=NC2=O)cccc1-c1ccccc1 |r,w:9.9,c:14| Show InChI InChI=1S/C21H21N3O2S/c22-16-9-5-11-24(13-16)19-15(12-18-20(25)23-21(26)27-18)8-4-10-17(19)14-6-2-1-3-7-14/h1-4,6-8,10,12,16H,5,9,11,13,22H2,(H,23,25,26)/t16-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PIM2 (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM184556
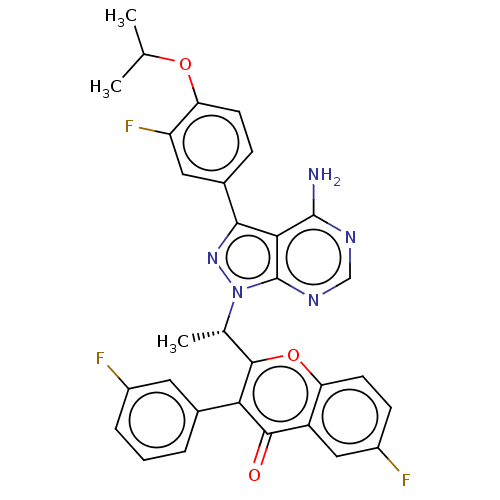
(US9150579, B1)Show SMILES CC(C)Oc1ccc(cc1F)-c1nn([C@@H](C)c2oc3ccc(F)cc3c(=O)c2-c2cccc(F)c2)c2ncnc(N)c12 |r| Show InChI InChI=1S/C31H24F3N5O3/c1-15(2)41-24-9-7-18(12-22(24)34)27-26-30(35)36-14-37-31(26)39(38-27)16(3)29-25(17-5-4-6-19(32)11-17)28(40)21-13-20(33)8-10-23(21)42-29/h4-16H,1-3H3,(H2,35,36,37)/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.20 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Macrophage-stimulating protein receptor
(Homo sapiens (Human)) | BDBM50427138
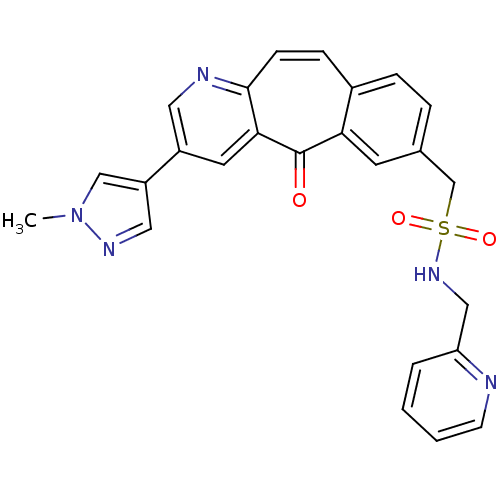
(CHEMBL2323775 | MK-8033)Show SMILES Cn1cc(cn1)-c1cnc2ccc3ccc(CS(=O)(=O)NCc4ccccn4)cc3c(=O)c2c1 Show InChI InChI=1S/C25H21N5O3S/c1-30-15-20(13-28-30)19-11-23-24(27-12-19)8-7-18-6-5-17(10-22(18)25(23)31)16-34(32,33)29-14-21-4-2-3-9-26-21/h2-13,15,29H,14,16H2,1H3 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of RON (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-1
(Homo sapiens (Human)) | BDBM50364776
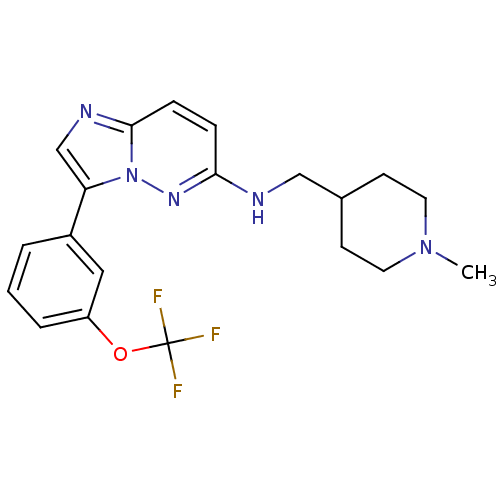
(CHEMBL1952141 | CHEMBL1952329)Show SMILES CN1CCC(CNc2ccc3ncc(-c4cccc(OC(F)(F)F)c4)n3n2)CC1 Show InChI InChI=1S/C20H22F3N5O/c1-27-9-7-14(8-10-27)12-24-18-5-6-19-25-13-17(28(19)26-18)15-3-2-4-16(11-15)29-20(21,22)23/h2-6,11,13-14H,7-10,12H2,1H3,(H,24,26) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PIM1 (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Interleukin-1 receptor-associated kinase 4
(Homo sapiens (Human)) | BDBM50040806
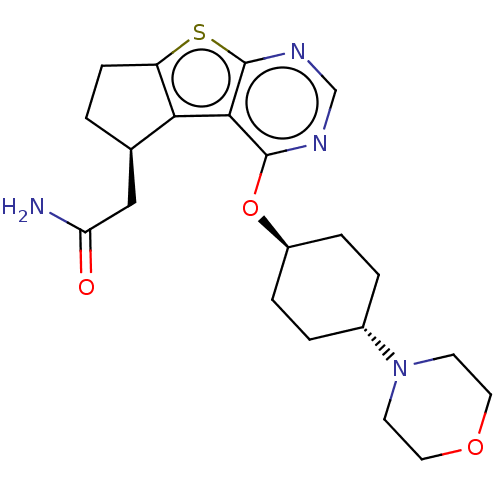
(CHEMBL3361254)Show SMILES NC(=O)C[C@H]1CCc2sc3ncnc(O[C@H]4CC[C@@H](CC4)N4CCOCC4)c3c12 |r,wU:18.21,wD:15.14,4.3,(33.74,-8.14,;32.23,-8.45,;31.75,-9.91,;31.21,-7.3,;31.69,-5.84,;33.16,-5.37,;33.17,-3.83,;31.7,-3.35,;30.79,-2.1,;29.32,-2.58,;27.99,-1.82,;26.66,-2.58,;26.66,-4.13,;27.99,-4.9,;27.99,-6.43,;26.66,-7.2,;25.33,-6.43,;24,-7.19,;23.99,-8.73,;25.32,-9.5,;26.66,-8.74,;22.66,-9.49,;21.33,-8.71,;20,-9.47,;19.98,-11,;21.31,-11.78,;22.65,-11.02,;29.32,-4.13,;30.79,-4.59,)| Show InChI InChI=1S/C21H28N4O3S/c22-17(26)11-13-1-6-16-18(13)19-20(23-12-24-21(19)29-16)28-15-4-2-14(3-5-15)25-7-9-27-10-8-25/h12-15H,1-11H2,(H2,22,26)/t13-,14-,15-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 7.5 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of IRAK4 (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50324735
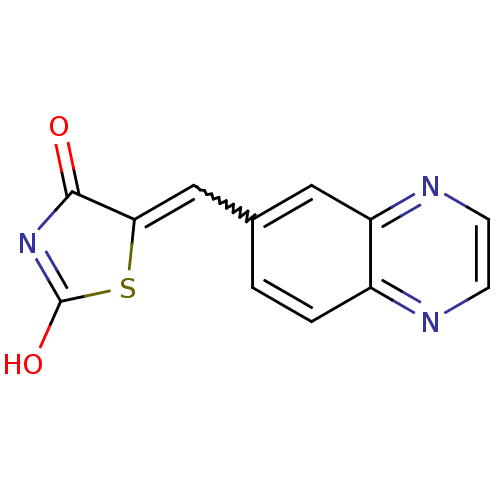
((5E)-5-(QUINOXALIN-6-YLMETHYLENE)-1,3-THIAZOLIDINE...)Show SMILES OC1=NC(=O)C(S1)=Cc1ccc2nccnc2c1 |w:7.8,t:1| Show InChI InChI=1S/C12H7N3O2S/c16-11-10(18-12(17)15-11)6-7-1-2-8-9(5-7)14-4-3-13-8/h1-6H,(H,15,16,17) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
TGF-beta receptor type-1
(Homo sapiens (Human)) | BDBM50015639
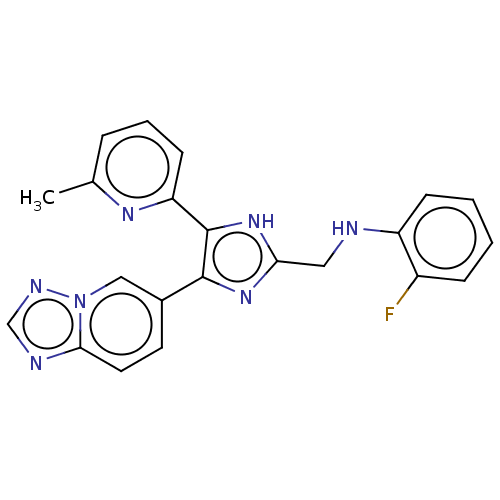
(CHEMBL3260567 | USRE47141, Example 2)Show SMILES Cc1cccc(n1)-c1[nH]c(CNc2ccccc2F)nc1-c1ccc2ncnn2c1 Show InChI InChI=1S/C22H18FN7/c1-14-5-4-8-18(27-14)22-21(15-9-10-20-25-13-26-30(20)12-15)28-19(29-22)11-24-17-7-3-2-6-16(17)23/h2-10,12-13,24H,11H2,1H3,(H,28,29) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of TGFBR1 (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
TGF-beta receptor type-1
(Homo sapiens (Human)) | BDBM50252542
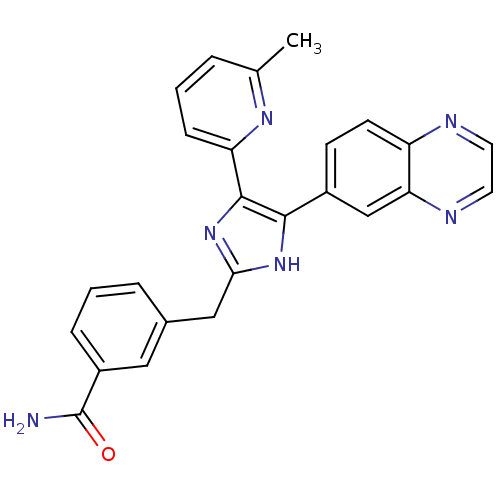
(3-((5-(6-methylpyridin-2-yl)-4-(quinoxalin-6-yl)-1...)Show SMILES Cc1cccc(n1)-c1nc(Cc2cccc(c2)C(N)=O)[nH]c1-c1ccc2nccnc2c1 Show InChI InChI=1S/C25H20N6O/c1-15-4-2-7-20(29-15)24-23(17-8-9-19-21(14-17)28-11-10-27-19)30-22(31-24)13-16-5-3-6-18(12-16)25(26)32/h2-12,14H,13H2,1H3,(H2,26,32)(H,30,31) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of TGFBR1 (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50507818
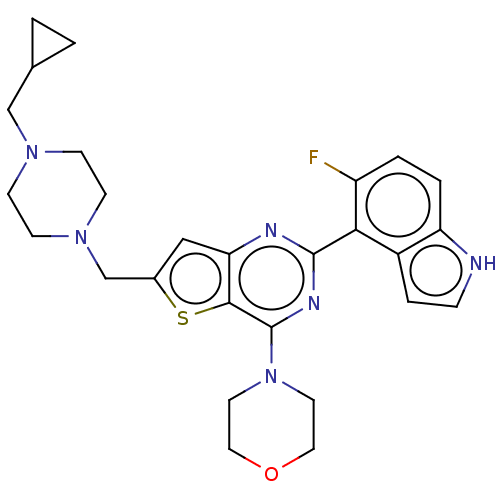
(CHEMBL4450504)Show SMILES Fc1ccc2[nH]ccc2c1-c1nc(N2CCOCC2)c2sc(CN3CCN(CC4CC4)CC3)cc2n1 Show InChI InChI=1S/C27H31FN6OS/c28-21-3-4-22-20(5-6-29-22)24(21)26-30-23-15-19(17-33-9-7-32(8-10-33)16-18-1-2-18)36-25(23)27(31-26)34-11-13-35-14-12-34/h3-6,15,18,29H,1-2,7-14,16-17H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50192880

(CHEMBL3984425 | US10329299, Compound 21 | US106752...)Show SMILES C[C@H](NC(=O)c1c(N)nn2cccnc12)c1cc2cccc(C#Cc3cnn(C)c3)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C30H24N8O2/c1-19(34-29(39)26-27(31)35-37-15-7-14-32-28(26)37)24-16-22-9-6-8-21(13-12-20-17-33-36(2)18-20)25(22)30(40)38(24)23-10-4-3-5-11-23/h3-11,14-19H,1-2H3,(H2,31,35)(H,34,39)/t19-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kalpha (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase JAK1
(Homo sapiens (Human)) | BDBM50507817

(CHEMBL4476770)Show SMILES NC(=O)c1nc(oc1Nc1ccc(cc1)C(=O)N1CCOCC1)-c1c(F)cccc1Cl Show InChI InChI=1S/C21H18ClFN4O4/c22-14-2-1-3-15(23)16(14)19-26-17(18(24)28)20(31-19)25-13-6-4-12(5-7-13)21(29)27-8-10-30-11-9-27/h1-7,25H,8-11H2,(H2,24,28) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of JAK1 in human Jurkat cells assessed as reduction in IFN-alpha stimulated JAK1 phosphorylation by caspase3/7 reagent-based Western blot ... |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50192880

(CHEMBL3984425 | US10329299, Compound 21 | US106752...)Show SMILES C[C@H](NC(=O)c1c(N)nn2cccnc12)c1cc2cccc(C#Cc3cnn(C)c3)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C30H24N8O2/c1-19(34-29(39)26-27(31)35-37-15-7-14-32-28(26)37)24-16-22-9-6-8-21(13-12-20-17-33-36(2)18-20)25(22)30(40)38(24)23-10-4-3-5-11-23/h3-11,14-19H,1-2H3,(H2,31,35)(H,34,39)/t19-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase JAK2
(Homo sapiens (Human)) | BDBM50507817

(CHEMBL4476770)Show SMILES NC(=O)c1nc(oc1Nc1ccc(cc1)C(=O)N1CCOCC1)-c1c(F)cccc1Cl Show InChI InChI=1S/C21H18ClFN4O4/c22-14-2-1-3-15(23)16(14)19-26-17(18(24)28)20(31-19)25-13-6-4-12(5-7-13)21(29)27-8-10-30-11-9-27/h1-7,25H,8-11H2,(H2,24,28) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 26 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of JAK2 in human Jurkat cells assessed as reduction in IFN-alpha stimulated JAK2 phosphorylation by caspase3/7 reagent-based Western blot ... |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50193013

(Duvelisib | INK-1147 | INK-1197 | IPI-145)Show SMILES C[C@H](Nc1ncnc2nc[nH]c12)c1cc2cccc(Cl)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C22H17ClN6O/c1-13(28-21-19-20(25-11-24-19)26-12-27-21)17-10-14-6-5-9-16(23)18(14)22(30)29(17)15-7-3-2-4-8-15/h2-13H,1H3,(H2,24,25,26,27,28)/t13-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50004547
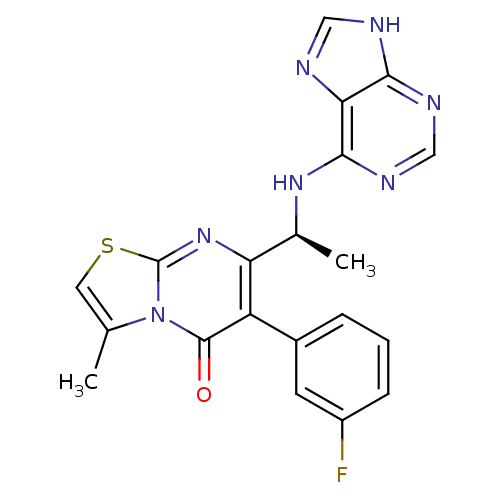
(CHEMBL2216863 | US10065963, Compound 28 | US104280...)Show SMILES C[C@H](Nc1ncnc2[nH]cnc12)c1nc2scc(C)n2c(=O)c1-c1cccc(F)c1 |r| Show InChI InChI=1S/C20H16FN7OS/c1-10-7-30-20-27-15(11(2)26-18-16-17(23-8-22-16)24-9-25-18)14(19(29)28(10)20)12-4-3-5-13(21)6-12/h3-9,11H,1-2H3,(H2,22,23,24,25,26)/t11-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 31 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Interleukin-1 receptor-associated kinase 4
(Homo sapiens (Human)) | BDBM390174
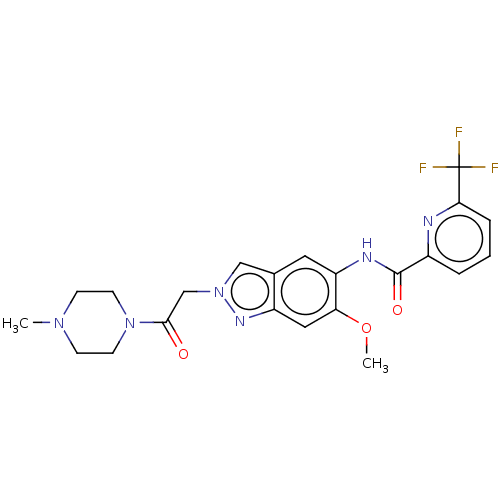
(US9951086, Example 64)Show SMILES COc1cc2nn(CC(=O)N3CCN(C)CC3)cc2cc1NC(=O)c1cccc(n1)C(F)(F)F Show InChI InChI=1S/C22H23F3N6O3/c1-29-6-8-30(9-7-29)20(32)13-31-12-14-10-17(18(34-2)11-16(14)28-31)27-21(33)15-4-3-5-19(26-15)22(23,24)25/h3-5,10-12H,6-9,13H2,1-2H3,(H,27,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 33 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of IRAK4 (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
TGF-beta receptor type-1
(Homo sapiens (Human)) | BDBM50204834

(2-(5-chloro-2-fluorophenyl)-N-(pyridin-4-yl)pterid...)Show InChI InChI=1S/C17H10ClFN6/c18-10-1-2-13(19)12(9-10)15-24-16-14(21-7-8-22-16)17(25-15)23-11-3-5-20-6-4-11/h1-9H,(H,20,22,23,24,25) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 35 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of TGFBR1 (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase JAK3
(Homo sapiens (Human)) | BDBM50507817

(CHEMBL4476770)Show SMILES NC(=O)c1nc(oc1Nc1ccc(cc1)C(=O)N1CCOCC1)-c1c(F)cccc1Cl Show InChI InChI=1S/C21H18ClFN4O4/c22-14-2-1-3-15(23)16(14)19-26-17(18(24)28)20(31-19)25-13-6-4-12(5-7-13)21(29)27-8-10-30-11-9-27/h1-7,25H,8-11H2,(H2,24,28) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 41 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of JAK3 in human Jurkat cells assessed as reduction in IFN-alpha stimulated JAK3 phosphorylation by caspase3/7 reagent-based Western blot ... |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Interleukin-1 receptor-associated kinase 4
(Homo sapiens (Human)) | BDBM50246769
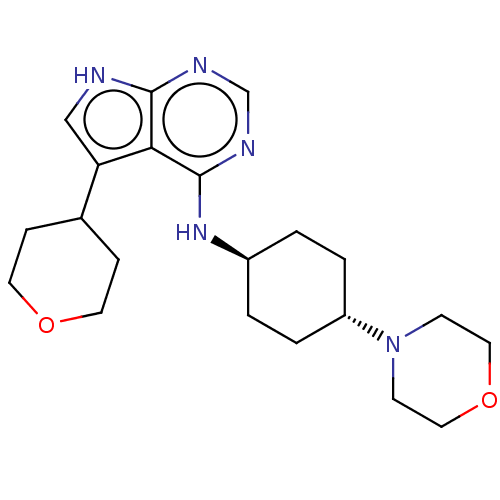
(CHEMBL4064608)Show SMILES C1CC(CCO1)c1c[nH]c2ncnc(N[C@H]3CC[C@@H](CC3)N3CCOCC3)c12 |r,wU:15.15,wD:18.22,(7.34,-15.64,;7.82,-17.1,;6.8,-18.25,;5.29,-17.94,;4.81,-16.48,;5.83,-15.33,;7.28,-19.71,;6.38,-20.97,;7.3,-22.21,;8.77,-21.73,;10.1,-22.5,;11.43,-21.72,;11.43,-20.18,;10.1,-19.41,;10.09,-17.87,;11.43,-17.1,;11.42,-15.56,;12.75,-14.8,;14.09,-15.57,;14.08,-17.11,;12.75,-17.87,;15.42,-14.8,;16.75,-15.58,;18.08,-14.82,;18.09,-13.28,;16.76,-12.5,;15.42,-13.27,;8.76,-20.18,)| Show InChI InChI=1S/C21H31N5O2/c1-3-17(26-7-11-28-12-8-26)4-2-16(1)25-21-19-18(15-5-9-27-10-6-15)13-22-20(19)23-14-24-21/h13-17H,1-12H2,(H2,22,23,24,25)/t16-,17- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 51 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of IRAK4 (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
TGF-beta receptor type-1
(Homo sapiens (Human)) | BDBM50015640
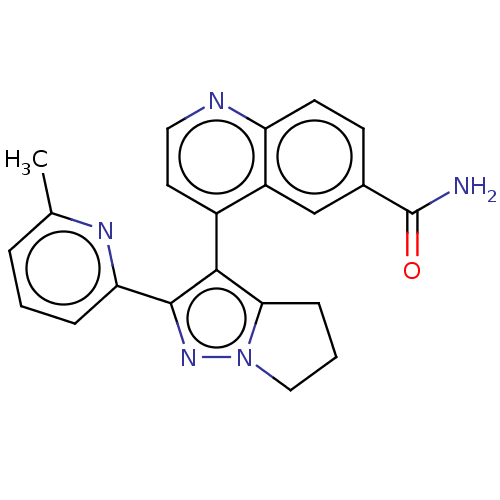
(GALUNISERTIB | LY-2157299)Show SMILES Cc1cccc(n1)-c1nn2CCCc2c1-c1ccnc2ccc(cc12)C(N)=O |(13.61,.14,;12.84,-1.19,;13.61,-2.53,;12.84,-3.86,;11.3,-3.86,;10.53,-2.53,;11.3,-1.19,;8.99,-2.53,;8.08,-1.28,;6.62,-1.76,;5.15,-1.28,;4.25,-2.53,;5.15,-3.77,;6.62,-3.3,;8.08,-3.77,;8.56,-5.24,;10.06,-5.56,;10.54,-7.02,;9.51,-8.17,;8,-7.85,;6.97,-8.99,;5.47,-8.67,;4.99,-7.21,;6.02,-6.06,;7.53,-6.38,;3.48,-6.89,;3.01,-5.42,;2.45,-8.03,)| Show InChI InChI=1S/C22H19N5O/c1-13-4-2-5-18(25-13)21-20(19-6-3-11-27(19)26-21)15-9-10-24-17-8-7-14(22(23)28)12-16(15)17/h2,4-5,7-10,12H,3,6,11H2,1H3,(H2,23,28) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 56 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of TGFBR1 (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50324735

((5E)-5-(QUINOXALIN-6-YLMETHYLENE)-1,3-THIAZOLIDINE...)Show SMILES OC1=NC(=O)C(S1)=Cc1ccc2nccnc2c1 |w:7.8,t:1| Show InChI InChI=1S/C12H7N3O2S/c16-11-10(18-12(17)15-11)6-7-1-2-8-9(5-7)14-4-3-13-8/h1-6H,(H,15,16,17) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 60 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kalpha (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50403068

(CHEMBL2216870 | IDELALISIB | US9745321, CAL-101)Show SMILES CC[C@H](Nc1ncnc2nc[nH]c12)c1nc2cccc(F)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C22H18FN7O/c1-2-15(28-20-18-19(25-11-24-18)26-12-27-20)21-29-16-10-6-9-14(23)17(16)22(31)30(21)13-7-4-3-5-8-13/h3-12,15H,2H2,1H3,(H2,24,25,26,27,28)/t15-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 63 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
MAP kinase-interacting serine/threonine-protein kinase 1
(Homo sapiens (Human)) | BDBM375544
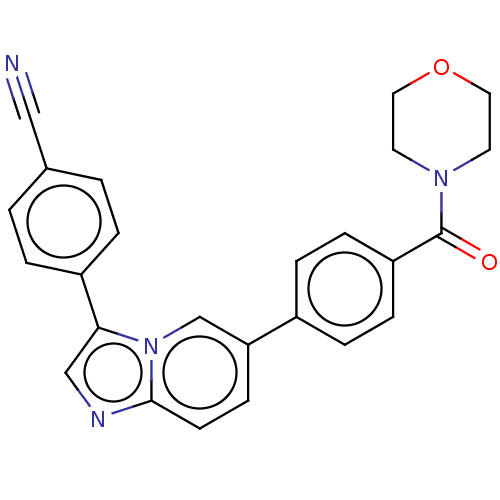
(4-(6-(4-(morpholine-4-carbonyl)phenyl)imidazo[1,2-...)Show SMILES O=C(N1CCOCC1)c1ccc(cc1)-c1ccc2ncc(-c3ccc(cc3)C#N)n2c1 Show InChI InChI=1S/C25H20N4O2/c26-15-18-1-3-20(4-2-18)23-16-27-24-10-9-22(17-29(23)24)19-5-7-21(8-6-19)25(30)28-11-13-31-14-12-28/h1-10,16-17H,11-14H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 64 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of MNK1 (unknown origin) expressed in HEK293 cells assessed as decrease in eIF4E phosphorylation at Ser209 residues by Western blot analys... |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-3
(Homo sapiens (Human)) | BDBM50364776

(CHEMBL1952141 | CHEMBL1952329)Show SMILES CN1CCC(CNc2ccc3ncc(-c4cccc(OC(F)(F)F)c4)n3n2)CC1 Show InChI InChI=1S/C20H22F3N5O/c1-27-9-7-14(8-10-27)12-24-18-5-6-19-25-13-17(28(19)26-18)15-3-2-4-16(11-15)29-20(21,22)23/h2-6,11,13-14H,7-10,12H2,1H3,(H,24,26) | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 69 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PIM3 (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
TGF-beta receptor type-1
(Homo sapiens (Human)) | BDBM50110208

(4-(4-(benzo[d][1,3]dioxol-5-yl)-5-(pyridin-2-yl)-1...)Show SMILES NC(=O)c1ccc(cc1)-c1nc(c([nH]1)-c1ccc2OCOc2c1)-c1ccccn1 Show InChI InChI=1S/C22H16N4O3/c23-21(27)13-4-6-14(7-5-13)22-25-19(20(26-22)16-3-1-2-10-24-16)15-8-9-17-18(11-15)29-12-28-17/h1-11H,12H2,(H2,23,27)(H,25,26) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 70 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of TGFBR1 (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM50192880

(CHEMBL3984425 | US10329299, Compound 21 | US106752...)Show SMILES C[C@H](NC(=O)c1c(N)nn2cccnc12)c1cc2cccc(C#Cc3cnn(C)c3)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C30H24N8O2/c1-19(34-29(39)26-27(31)35-37-15-7-14-32-28(26)37)24-16-22-9-6-8-21(13-12-20-17-33-36(2)18-20)25(22)30(40)38(24)23-10-4-3-5-11-23/h3-11,14-19H,1-2H3,(H2,31,35)(H,34,39)/t19-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 82 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kbeta (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM25084
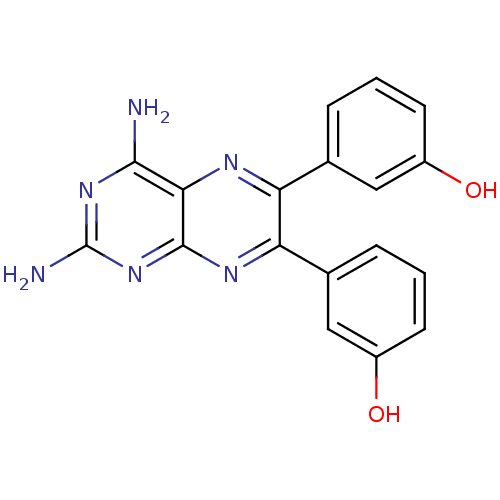
(3-[2,4-diamino-6-(3-hydroxyphenyl)pteridin-7-yl]ph...)Show SMILES Nc1nc(N)c2nc(-c3cccc(O)c3)c(nc2n1)-c1cccc(O)c1 Show InChI InChI=1S/C18H14N6O2/c19-16-15-17(24-18(20)23-16)22-14(10-4-2-6-12(26)8-10)13(21-15)9-3-1-5-11(25)7-9/h1-8,25-26H,(H4,19,20,22,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 83 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM50193013

(Duvelisib | INK-1147 | INK-1197 | IPI-145)Show SMILES C[C@H](Nc1ncnc2nc[nH]c12)c1cc2cccc(Cl)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C22H17ClN6O/c1-13(28-21-19-20(25-11-24-19)26-12-27-21)17-10-14-6-5-9-16(23)18(14)22(30)29(17)15-7-3-2-4-8-15/h2-13H,1H3,(H2,24,25,26,27,28)/t13-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 85 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kbeta (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
MAP kinase-interacting serine/threonine-protein kinase 2
(Homo sapiens (Human)) | BDBM375544

(4-(6-(4-(morpholine-4-carbonyl)phenyl)imidazo[1,2-...)Show SMILES O=C(N1CCOCC1)c1ccc(cc1)-c1ccc2ncc(-c3ccc(cc3)C#N)n2c1 Show InChI InChI=1S/C25H20N4O2/c26-15-18-1-3-20(4-2-18)23-16-27-24-10-9-22(17-29(23)24)19-5-7-21(8-6-19)25(30)28-11-13-31-14-12-28/h1-10,16-17H,11-14H2 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 86 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of MNK2 (unknown origin) expressed in HEK293 cells assessed as decrease in eIF4E phosphorylation at Ser209 residues by Western blot analys... |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM25084

(3-[2,4-diamino-6-(3-hydroxyphenyl)pteridin-7-yl]ph...)Show SMILES Nc1nc(N)c2nc(-c3cccc(O)c3)c(nc2n1)-c1cccc(O)c1 Show InChI InChI=1S/C18H14N6O2/c19-16-15-17(24-18(20)23-16)22-14(10-4-2-6-12(26)8-10)13(21-15)9-3-1-5-11(25)7-9/h1-8,25-26H,(H4,19,20,22,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 235 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50324735

((5E)-5-(QUINOXALIN-6-YLMETHYLENE)-1,3-THIAZOLIDINE...)Show SMILES OC1=NC(=O)C(S1)=Cc1ccc2nccnc2c1 |w:7.8,t:1| Show InChI InChI=1S/C12H7N3O2S/c16-11-10(18-12(17)15-11)6-7-1-2-8-9(5-7)14-4-3-13-8/h1-6H,(H,15,16,17) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-2
(Homo sapiens (Human)) | BDBM50364776

(CHEMBL1952141 | CHEMBL1952329)Show SMILES CN1CCC(CNc2ccc3ncc(-c4cccc(OC(F)(F)F)c4)n3n2)CC1 Show InChI InChI=1S/C20H22F3N5O/c1-27-9-7-14(8-10-27)12-24-18-5-6-19-25-13-17(28(19)26-18)15-3-2-4-16(11-15)29-20(21,22)23/h2-6,11,13-14H,7-10,12H2,1H3,(H,24,26) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 363 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PIM2 (unknown origin) |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50403068

(CHEMBL2216870 | IDELALISIB | US9745321, CAL-101)Show SMILES CC[C@H](Nc1ncnc2nc[nH]c12)c1nc2cccc(F)c2c(=O)n1-c1ccccc1 |r| Show InChI InChI=1S/C22H18FN7O/c1-2-15(28-20-18-19(25-11-24-18)26-12-27-20)21-29-16-10-6-9-14(23)17(16)22(31)30(21)13-7-4-3-5-8-13/h3-12,15H,2H2,1H3,(H2,24,25,26,27,28)/t15-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 800 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of human N-terminal His-tagged PI3Kalpha/p85alpha expressed in Spodoptera frugiperda using phosphatidylinositol as substrate |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
MAP kinase-interacting serine/threonine-protein kinase 1
(Homo sapiens (Human)) | BDBM50130693
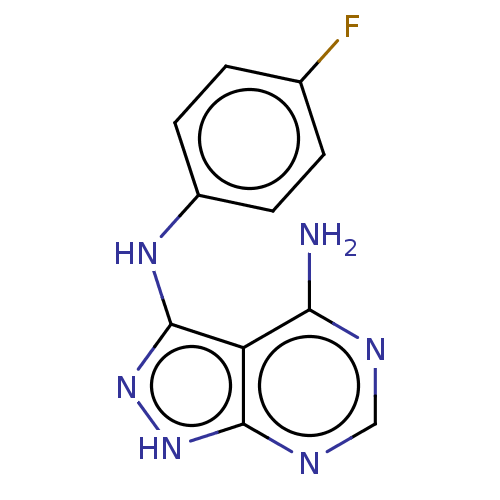
(CGP-57380 | CHEMBL1240885)Show InChI InChI=1S/C11H9FN6/c12-6-1-3-7(4-2-6)16-11-8-9(13)14-5-15-10(8)17-18-11/h1-5H,(H4,13,14,15,16,17,18) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 870 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of MNK1 (unknown origin) expressed in HEK293 cells assessed as decrease in eIF4E phosphorylation at Ser209 residues by Western blot analys... |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM50507818

(CHEMBL4450504)Show SMILES Fc1ccc2[nH]ccc2c1-c1nc(N2CCOCC2)c2sc(CN3CCN(CC4CC4)CC3)cc2n1 Show InChI InChI=1S/C27H31FN6OS/c28-21-3-4-22-20(5-6-29-22)24(21)26-30-23-15-19(17-33-9-7-32(8-10-33)16-18-1-2-18)36-25(23)27(31-26)34-11-13-35-14-12-34/h3-6,15,18,29H,1-2,7-14,16-17H2 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.08E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of human full length recombinant N-terminal His-tagged PI3Kbeta/p85alpha expressed in baculovirus infected Sf21 insect cells |
Eur J Med Chem 163: 413-427 (2019)
Article DOI: 10.1016/j.ejmech.2018.11.072
BindingDB Entry DOI: 10.7270/Q2736V7K |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data