 Found 39 hits Enz. Inhib. hit(s) with all data for entry = 50013265
Found 39 hits Enz. Inhib. hit(s) with all data for entry = 50013265 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cholinesterase
(Homo sapiens (Human)) | BDBM50564199
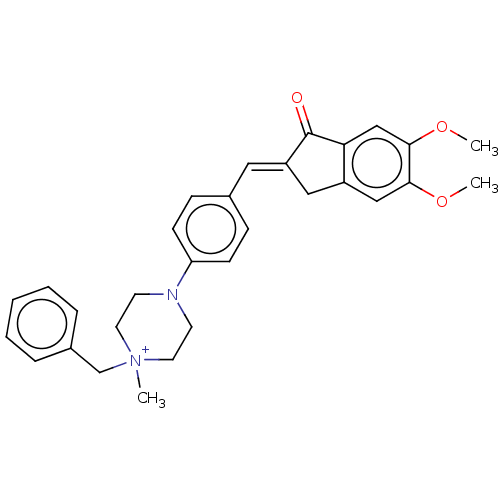
(CHEMBL4800260)Show SMILES [Cl-].COc1cc2C\C(=C/c3ccc(cc3)N3CC[N+](C)(Cc4ccccc4)CC3)C(=O)c2cc1OC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Non competitive inhibition of human plasma BuChE using varying levels of butyrylthiocholine iodide as substrate by Lineweaver-Burk plot analysis |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50564203
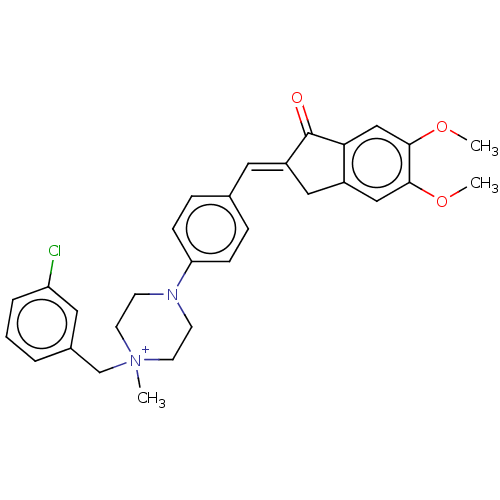
(CHEMBL4791075)Show SMILES [Br-].COc1cc2C\C(=C/c3ccc(cc3)N3CC[N+](C)(Cc4cccc(Cl)c4)CC3)C(=O)c2cc1OC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Non competitive inhibition of human plasma BuChE using varying levels of butyrylthiocholine iodide as substrate by Lineweaver-Burk plot analysis |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50564199

(CHEMBL4800260)Show SMILES [Cl-].COc1cc2C\C(=C/c3ccc(cc3)N3CC[N+](C)(Cc4ccccc4)CC3)C(=O)c2cc1OC | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Non competitive inhibition of human erythrocyte AChE using varying levels of acetylthiocholine iodide as substrate by Lineweaver-Burk plot analysis |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50564203

(CHEMBL4791075)Show SMILES [Br-].COc1cc2C\C(=C/c3ccc(cc3)N3CC[N+](C)(Cc4cccc(Cl)c4)CC3)C(=O)c2cc1OC | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Mixed type inhibition of human erythrocyte AChE using varying levels of acetylthiocholine iodide as substrate by Lineweaver-Burk plot analysis |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50564213
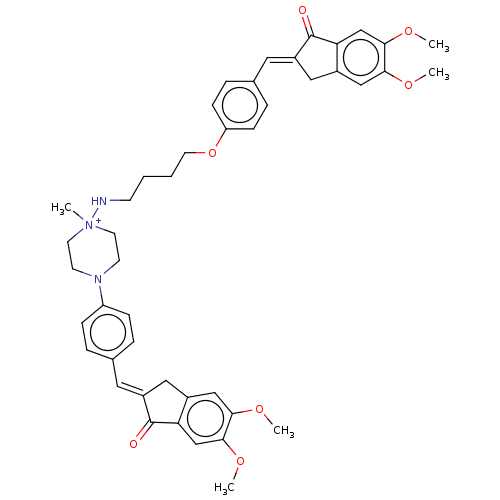
(CHEMBL4780352)Show SMILES [Br-].COc1cc2C\C(=C/c3ccc(OCCCCN[N+]4(C)CCN(CC4)c4ccc(\C=C5/Cc6cc(OC)c(OC)cc6C5=O)cc4)cc3)C(=O)c2cc1OC | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human erythrocyte AChE using acetylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM8960

((+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimet...)Show SMILES COc1cc2CC(CC3CCN(Cc4ccccc4)CC3)C(=O)c2cc1OC Show InChI InChI=1S/C24H29NO3/c1-27-22-14-19-13-20(24(26)21(19)15-23(22)28-2)12-17-8-10-25(11-9-17)16-18-6-4-3-5-7-18/h3-7,14-15,17,20H,8-13,16H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human erythrocyte AChE using acetylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50564198
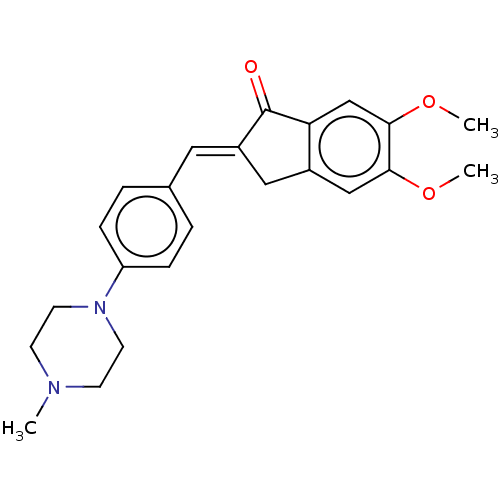
(CHEMBL4786658)Show SMILES COc1cc2C\C(=C/c3ccc(cc3)N3CCN(C)CC3)C(=O)c2cc1OC | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 320 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human erythrocyte AChE using acetylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50564203

(CHEMBL4791075)Show SMILES [Br-].COc1cc2C\C(=C/c3ccc(cc3)N3CC[N+](C)(Cc4cccc(Cl)c4)CC3)C(=O)c2cc1OC | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 320 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human erythrocyte AChE using acetylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50564199

(CHEMBL4800260)Show SMILES [Cl-].COc1cc2C\C(=C/c3ccc(cc3)N3CC[N+](C)(Cc4ccccc4)CC3)C(=O)c2cc1OC | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 370 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human erythrocyte AChE using acetylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50564205

(CHEMBL4784191)Show SMILES [Cl-].COc1cc2C\C(=C/c3ccc(cc3)N3CC[N+](C)(Cc4ccccn4)CC3)C(=O)c2cc1OC | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 380 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human erythrocyte AChE using acetylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50564199

(CHEMBL4800260)Show SMILES [Cl-].COc1cc2C\C(=C/c3ccc(cc3)N3CC[N+](C)(Cc4ccccc4)CC3)C(=O)c2cc1OC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 430 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human plasma BuChE using butyrylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50564203

(CHEMBL4791075)Show SMILES [Br-].COc1cc2C\C(=C/c3ccc(cc3)N3CC[N+](C)(Cc4cccc(Cl)c4)CC3)C(=O)c2cc1OC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 430 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human plasma BuChE using butyrylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50564210
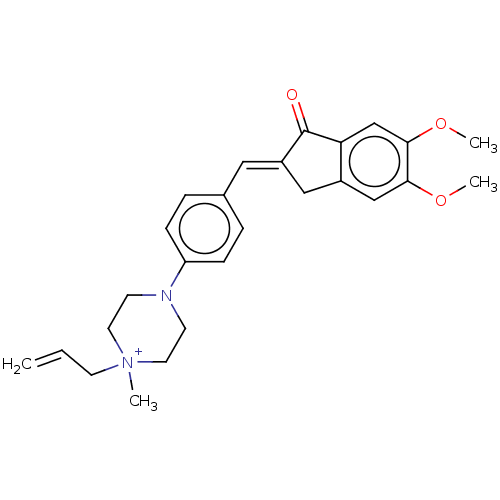
(CHEMBL4798336)Show SMILES [Cl-].COc1cc2C\C(=C/c3ccc(cc3)N3CC[N+](C)(CC=C)CC3)C(=O)c2cc1OC | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 430 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human erythrocyte AChE using acetylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50564204

(CHEMBL4787855)Show SMILES [Cl-].COc1cc2C\C(=C/c3ccc(cc3)N3CC[N+](C)(Cc4ccccc4Cl)CC3)C(=O)c2cc1OC | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 480 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human erythrocyte AChE using acetylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50564202

(CHEMBL4794208)Show SMILES [Br-].COc1cc2C\C(=C/c3ccc(cc3)N3CC[N+](C)(Cc4ccccc4Br)CC3)C(=O)c2cc1OC | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 540 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human erythrocyte AChE using acetylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50564208
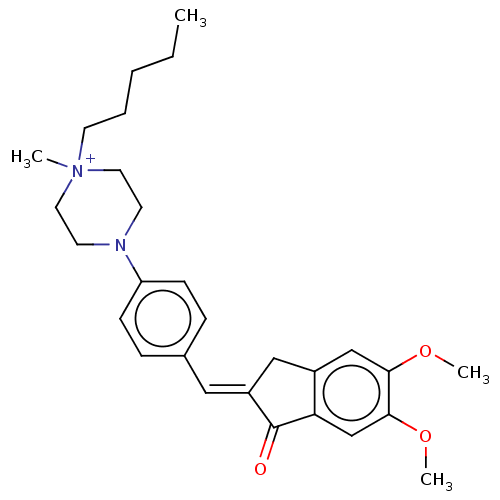
(CHEMBL4797677)Show SMILES [Br-].CCCCC[N+]1(C)CCN(CC1)c1ccc(\C=C2/Cc3cc(OC)c(OC)cc3C2=O)cc1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 540 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human erythrocyte AChE using acetylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50564207

(CHEMBL4776999)Show SMILES [Br-].CCCC[N+]1(C)CCN(CC1)c1ccc(\C=C2/Cc3cc(OC)c(OC)cc3C2=O)cc1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 540 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human erythrocyte AChE using acetylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50564201

(CHEMBL4778690)Show SMILES [Br-].COc1cc2C\C(=C/c3ccc(cc3)N3CC[N+](C)(Cc4cccc(Br)c4)CC3)C(=O)c2cc1OC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 650 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human plasma BuChE using butyrylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50564213

(CHEMBL4780352)Show SMILES [Br-].COc1cc2C\C(=C/c3ccc(OCCCCN[N+]4(C)CCN(CC4)c4ccc(\C=C5/Cc6cc(OC)c(OC)cc6C5=O)cc4)cc3)C(=O)c2cc1OC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 830 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human plasma BuChE using butyrylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50564211

(CHEMBL4790642)Show SMILES [Cl-].COc1cc2C\C(=C/c3ccc(cc3)N3CC[N+](C)(CCO)CC3)C(=O)c2cc1OC | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human erythrocyte AChE using acetylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50564202

(CHEMBL4794208)Show SMILES [Br-].COc1cc2C\C(=C/c3ccc(cc3)N3CC[N+](C)(Cc4ccccc4Br)CC3)C(=O)c2cc1OC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human plasma BuChE using butyrylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50564201

(CHEMBL4778690)Show SMILES [Br-].COc1cc2C\C(=C/c3ccc(cc3)N3CC[N+](C)(Cc4cccc(Br)c4)CC3)C(=O)c2cc1OC | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.99E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human erythrocyte AChE using acetylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50564206
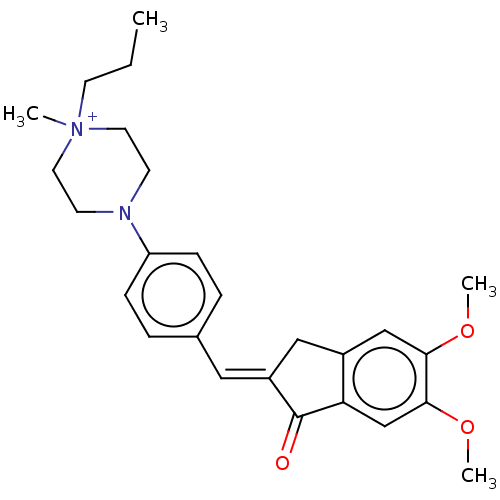
(CHEMBL4794690)Show SMILES [Br-].CCC[N+]1(C)CCN(CC1)c1ccc(\C=C2/Cc3cc(OC)c(OC)cc3C2=O)cc1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.15E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human erythrocyte AChE using acetylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50564204

(CHEMBL4787855)Show SMILES [Cl-].COc1cc2C\C(=C/c3ccc(cc3)N3CC[N+](C)(Cc4ccccc4Cl)CC3)C(=O)c2cc1OC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.16E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human plasma BuChE using butyrylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50564200
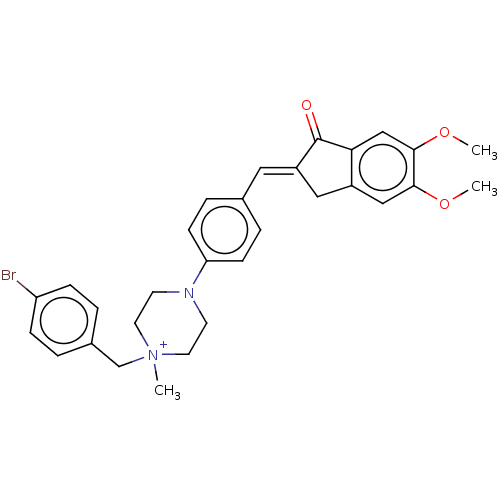
(CHEMBL4780628)Show SMILES [Br-].COc1cc2C\C(=C/c3ccc(cc3)N3CC[N+](C)(Cc4ccc(Br)cc4)CC3)C(=O)c2cc1OC | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.55E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human erythrocyte AChE using acetylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM10404

((1S,12S,14R)-9-methoxy-4-methyl-11-oxa-4-azatetrac...)Show SMILES COc1ccc2CN(C)CC[C@@]34C=C[C@H](O)C[C@@H]3Oc1c24 |r,c:12| Show InChI InChI=1S/C17H21NO3/c1-18-8-7-17-6-5-12(19)9-14(17)21-16-13(20-2)4-3-11(10-18)15(16)17/h3-6,12,14,19H,7-10H2,1-2H3/t12-,14-,17-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human erythrocyte AChE using acetylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cholinesterase
(Homo sapiens (Human)) | BDBM50564205

(CHEMBL4784191)Show SMILES [Cl-].COc1cc2C\C(=C/c3ccc(cc3)N3CC[N+](C)(Cc4ccccn4)CC3)C(=O)c2cc1OC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.24E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human plasma BuChE using butyrylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50564208

(CHEMBL4797677)Show SMILES [Br-].CCCCC[N+]1(C)CCN(CC1)c1ccc(\C=C2/Cc3cc(OC)c(OC)cc3C2=O)cc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.24E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human plasma BuChE using butyrylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50564209

(CHEMBL4794354)Show SMILES [Br-].CCCCCC[N+]1(C)CCN(CC1)c1ccc(\C=C2/Cc3cc(OC)c(OC)cc3C2=O)cc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human plasma BuChE using butyrylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50564209

(CHEMBL4794354)Show SMILES [Br-].CCCCCC[N+]1(C)CCN(CC1)c1ccc(\C=C2/Cc3cc(OC)c(OC)cc3C2=O)cc1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4.25E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human erythrocyte AChE using acetylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50564200

(CHEMBL4780628)Show SMILES [Br-].COc1cc2C\C(=C/c3ccc(cc3)N3CC[N+](C)(Cc4ccc(Br)cc4)CC3)C(=O)c2cc1OC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human plasma BuChE using butyrylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50564211

(CHEMBL4790642)Show SMILES [Cl-].COc1cc2C\C(=C/c3ccc(cc3)N3CC[N+](C)(CCO)CC3)C(=O)c2cc1OC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6.48E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human plasma BuChE using butyrylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM8960

((+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimet...)Show SMILES COc1cc2CC(CC3CCN(Cc4ccccc4)CC3)C(=O)c2cc1OC Show InChI InChI=1S/C24H29NO3/c1-27-22-14-19-13-20(24(26)21(19)15-23(22)28-2)12-17-8-10-25(11-9-17)16-18-6-4-3-5-7-18/h3-7,14-15,17,20H,8-13,16H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.44E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human plasma BuChE using butyrylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50564212
(CHEMBL4794250)Show SMILES [Br-].COc1cc2C\C(=C/c3ccc(OCCN[N+]4(C)CCN(CC4)c4ccc(\C=C5/Cc6cc(OC)c(OC)cc6C5=O)cc4)cc3)C(=O)c2cc1OC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human plasma BuChE using butyrylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50564206

(CHEMBL4794690)Show SMILES [Br-].CCC[N+]1(C)CCN(CC1)c1ccc(\C=C2/Cc3cc(OC)c(OC)cc3C2=O)cc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.02E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human plasma BuChE using butyrylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50564207

(CHEMBL4776999)Show SMILES [Br-].CCCC[N+]1(C)CCN(CC1)c1ccc(\C=C2/Cc3cc(OC)c(OC)cc3C2=O)cc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.16E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human plasma BuChE using butyrylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50564212

(CHEMBL4794250)Show SMILES [Br-].COc1cc2C\C(=C/c3ccc(OCCN[N+]4(C)CCN(CC4)c4ccc(\C=C5/Cc6cc(OC)c(OC)cc6C5=O)cc4)cc3)C(=O)c2cc1OC | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human erythrocyte AChE using acetylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50564210

(CHEMBL4798336)Show SMILES [Cl-].COc1cc2C\C(=C/c3ccc(cc3)N3CC[N+](C)(CC=C)CC3)C(=O)c2cc1OC | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.52E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human plasma BuChE using butyrylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM10404

((1S,12S,14R)-9-methoxy-4-methyl-11-oxa-4-azatetrac...)Show SMILES COc1ccc2CN(C)CC[C@@]34C=C[C@H](O)C[C@@H]3Oc1c24 |r,c:12| Show InChI InChI=1S/C17H21NO3/c1-18-8-7-17-6-5-12(19)9-14(17)21-16-13(20-2)4-3-11(10-18)15(16)17/h3-6,12,14,19H,7-10H2,1-2H3/t12-,14-,17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | 3.16E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human plasma BuChE using butyrylthiocholine as substrate measured after 5 mins by Ellman's method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112140
BindingDB Entry DOI: 10.7270/Q2RV0SGV |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data