 Found 34 hits Enz. Inhib. hit(s) with all data for entry = 50039975
Found 34 hits Enz. Inhib. hit(s) with all data for entry = 50039975 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM8960

((+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimet...)Show SMILES COc1cc2CC(CC3CCN(Cc4ccccc4)CC3)C(=O)c2cc1OC Show InChI InChI=1S/C24H29NO3/c1-27-22-14-19-13-20(24(26)21(19)15-23(22)28-2)12-17-8-10-25(11-9-17)16-18-6-4-3-5-7-18/h3-7,14-15,17,20H,8-13,16H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of human AChE using acetylthiocholine iodide as substrate after 5 mins by DTNB method |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50387090
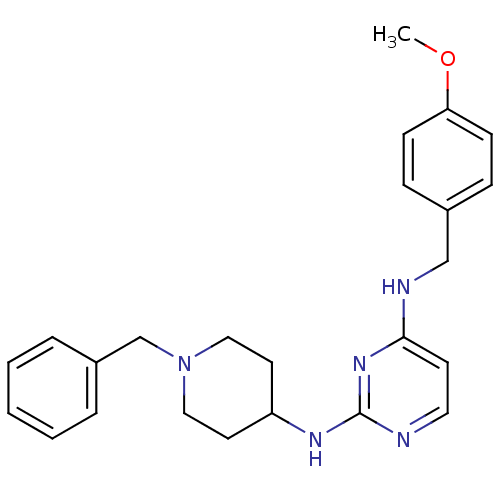
(CHEMBL2047229)Show SMILES COc1ccc(CNc2ccnc(NC3CCN(Cc4ccccc4)CC3)n2)cc1 Show InChI InChI=1S/C24H29N5O/c1-30-22-9-7-19(8-10-22)17-26-23-11-14-25-24(28-23)27-21-12-15-29(16-13-21)18-20-5-3-2-4-6-20/h2-11,14,21H,12-13,15-18H2,1H3,(H2,25,26,27,28) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 600 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50387094
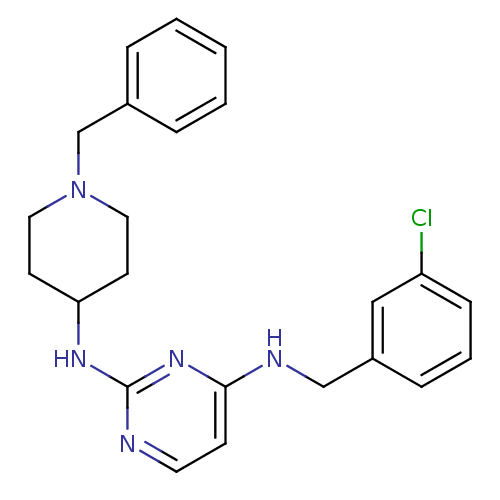
(CHEMBL2047224)Show SMILES Clc1cccc(CNc2ccnc(NC3CCN(Cc4ccccc4)CC3)n2)c1 Show InChI InChI=1S/C23H26ClN5/c24-20-8-4-7-19(15-20)16-26-22-9-12-25-23(28-22)27-21-10-13-29(14-11-21)17-18-5-2-1-3-6-18/h1-9,12,15,21H,10-11,13-14,16-17H2,(H2,25,26,27,28) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 600 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50387092
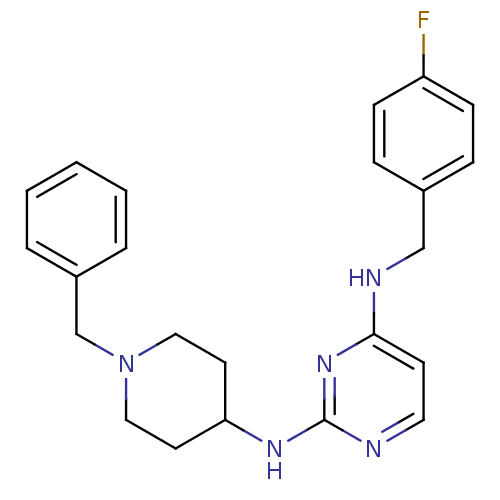
(CHEMBL2047227)Show SMILES Fc1ccc(CNc2ccnc(NC3CCN(Cc4ccccc4)CC3)n2)cc1 Show InChI InChI=1S/C23H26FN5/c24-20-8-6-18(7-9-20)16-26-22-10-13-25-23(28-22)27-21-11-14-29(15-12-21)17-19-4-2-1-3-5-19/h1-10,13,21H,11-12,14-17H2,(H2,25,26,27,28) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 700 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50387088
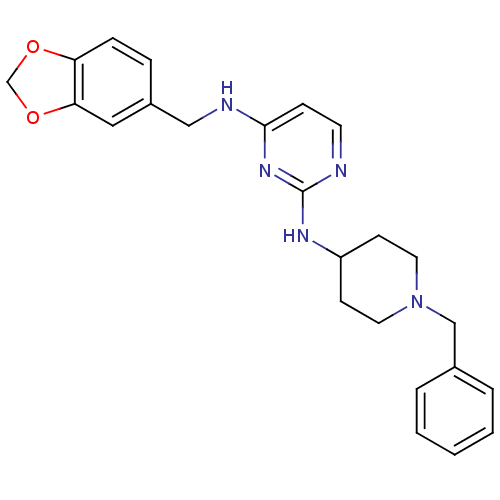
(CHEMBL2047375)Show SMILES C(Nc1ccnc(NC2CCN(Cc3ccccc3)CC2)n1)c1ccc2OCOc2c1 Show InChI InChI=1S/C24H27N5O2/c1-2-4-18(5-3-1)16-29-12-9-20(10-13-29)27-24-25-11-8-23(28-24)26-15-19-6-7-21-22(14-19)31-17-30-21/h1-8,11,14,20H,9-10,12-13,15-17H2,(H2,25,26,27,28) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50387095
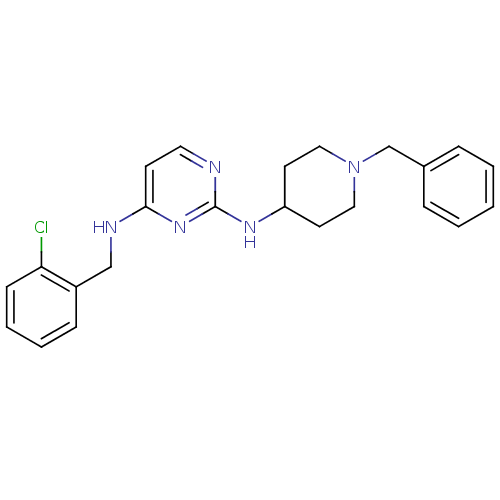
(CHEMBL2047223)Show InChI InChI=1S/C23H26ClN5/c24-21-9-5-4-8-19(21)16-26-22-10-13-25-23(28-22)27-20-11-14-29(15-12-20)17-18-6-2-1-3-7-18/h1-10,13,20H,11-12,14-17H2,(H2,25,26,27,28) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM50387092

(CHEMBL2047227)Show SMILES Fc1ccc(CNc2ccnc(NC3CCN(Cc4ccccc4)CC3)n2)cc1 Show InChI InChI=1S/C23H26FN5/c24-20-8-6-18(7-9-20)16-26-22-10-13-25-23(28-22)27-21-11-14-29(15-12-21)17-19-4-2-1-3-5-19/h1-10,13,21H,11-12,14-17H2,(H2,25,26,27,28) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of equine BuChE using butyrylthiocholine iodide as substrate after 5 mins by DTNB method |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM50387095

(CHEMBL2047223)Show InChI InChI=1S/C23H26ClN5/c24-21-9-5-4-8-19(21)16-26-22-10-13-25-23(28-22)27-20-11-14-29(15-12-20)17-18-6-2-1-3-7-18/h1-10,13,20H,11-12,14-17H2,(H2,25,26,27,28) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of equine BuChE using butyrylthiocholine iodide as substrate after 5 mins by DTNB method |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM50387091
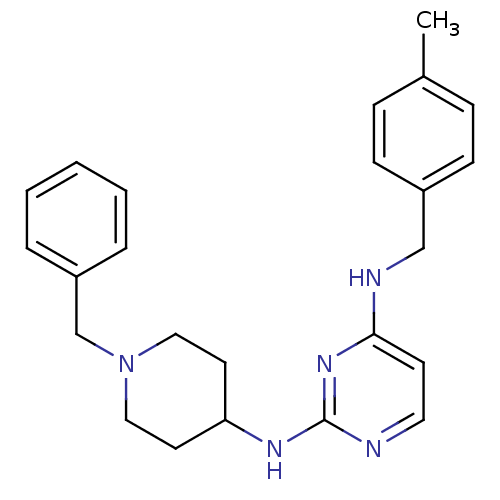
(CHEMBL2047228)Show SMILES Cc1ccc(CNc2ccnc(NC3CCN(Cc4ccccc4)CC3)n2)cc1 Show InChI InChI=1S/C24H29N5/c1-19-7-9-20(10-8-19)17-26-23-11-14-25-24(28-23)27-22-12-15-29(16-13-22)18-21-5-3-2-4-6-21/h2-11,14,22H,12-13,15-18H2,1H3,(H2,25,26,27,28) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of equine BuChE using butyrylthiocholine iodide as substrate after 5 mins by DTNB method |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM50387094

(CHEMBL2047224)Show SMILES Clc1cccc(CNc2ccnc(NC3CCN(Cc4ccccc4)CC3)n2)c1 Show InChI InChI=1S/C23H26ClN5/c24-20-8-4-7-19(15-20)16-26-22-9-12-25-23(28-22)27-21-10-13-29(14-11-21)17-18-5-2-1-3-6-18/h1-9,12,15,21H,10-11,13-14,16-17H2,(H2,25,26,27,28) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of equine BuChE using butyrylthiocholine iodide as substrate after 5 mins by DTNB method |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM50387093

(CHEMBL2047225)Show SMILES Clc1ccc(CNc2ccnc(NC3CCN(Cc4ccccc4)CC3)n2)cc1 Show InChI InChI=1S/C23H26ClN5/c24-20-8-6-18(7-9-20)16-26-22-10-13-25-23(28-22)27-21-11-14-29(15-12-21)17-19-4-2-1-3-5-19/h1-10,13,21H,11-12,14-17H2,(H2,25,26,27,28) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of equine BuChE using butyrylthiocholine iodide as substrate after 5 mins by DTNB method |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM10404

((1S,12S,14R)-9-methoxy-4-methyl-11-oxa-4-azatetrac...)Show SMILES COc1ccc2CN(C)CC[C@@]34C=C[C@H](O)C[C@@H]3Oc1c24 |r,c:12| Show InChI InChI=1S/C17H21NO3/c1-18-8-7-17-6-5-12(19)9-14(17)21-16-13(20-2)4-3-11(10-18)15(16)17/h3-6,12,14,19H,7-10H2,1-2H3/t12-,14-,17-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of human AChE using acetylthiocholine iodide as substrate after 5 mins by DTNB method |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50387093

(CHEMBL2047225)Show SMILES Clc1ccc(CNc2ccnc(NC3CCN(Cc4ccccc4)CC3)n2)cc1 Show InChI InChI=1S/C23H26ClN5/c24-20-8-6-18(7-9-20)16-26-22-10-13-25-23(28-22)27-21-11-14-29(15-12-21)17-19-4-2-1-3-5-19/h1-10,13,21H,11-12,14-17H2,(H2,25,26,27,28) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM8960

((+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimet...)Show SMILES COc1cc2CC(CC3CCN(Cc4ccccc4)CC3)C(=O)c2cc1OC Show InChI InChI=1S/C24H29NO3/c1-27-22-14-19-13-20(24(26)21(19)15-23(22)28-2)12-17-8-10-25(11-9-17)16-18-6-4-3-5-7-18/h3-7,14-15,17,20H,8-13,16H2,1-2H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM8960

((+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimet...)Show SMILES COc1cc2CC(CC3CCN(Cc4ccccc4)CC3)C(=O)c2cc1OC Show InChI InChI=1S/C24H29NO3/c1-27-22-14-19-13-20(24(26)21(19)15-23(22)28-2)12-17-8-10-25(11-9-17)16-18-6-4-3-5-7-18/h3-7,14-15,17,20H,8-13,16H2,1-2H3 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of equine BuChE using butyrylthiocholine iodide as substrate after 5 mins by DTNB method |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM50387088

(CHEMBL2047375)Show SMILES C(Nc1ccnc(NC2CCN(Cc3ccccc3)CC2)n1)c1ccc2OCOc2c1 Show InChI InChI=1S/C24H27N5O2/c1-2-4-18(5-3-1)16-29-12-9-20(10-13-29)27-24-25-11-8-23(28-24)26-15-19-6-7-21-22(14-19)31-17-30-21/h1-8,11,14,20H,9-10,12-13,15-17H2,(H2,25,26,27,28) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of equine BuChE using butyrylthiocholine iodide as substrate after 5 mins by DTNB method |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM50387097
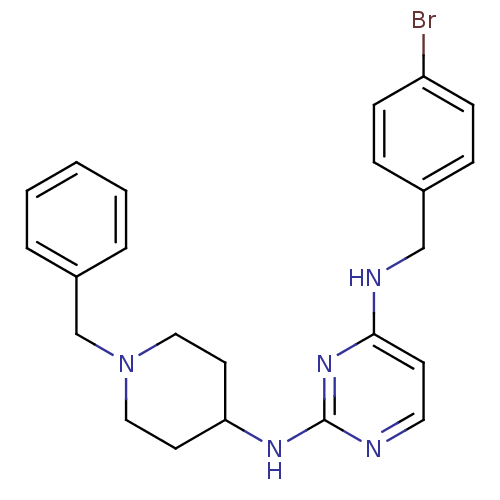
(CHEMBL2047226)Show SMILES Brc1ccc(CNc2ccnc(NC3CCN(Cc4ccccc4)CC3)n2)cc1 Show InChI InChI=1S/C23H26BrN5/c24-20-8-6-18(7-9-20)16-26-22-10-13-25-23(28-22)27-21-11-14-29(15-12-21)17-19-4-2-1-3-5-19/h1-10,13,21H,11-12,14-17H2,(H2,25,26,27,28) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of equine BuChE using butyrylthiocholine iodide as substrate after 5 mins by DTNB method |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM50387090

(CHEMBL2047229)Show SMILES COc1ccc(CNc2ccnc(NC3CCN(Cc4ccccc4)CC3)n2)cc1 Show InChI InChI=1S/C24H29N5O/c1-30-22-9-7-19(8-10-22)17-26-23-11-14-25-24(28-23)27-21-12-15-29(16-13-21)18-20-5-3-2-4-6-20/h2-11,14,21H,12-13,15-18H2,1H3,(H2,25,26,27,28) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of equine BuChE using butyrylthiocholine iodide as substrate after 5 mins by DTNB method |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM50387089
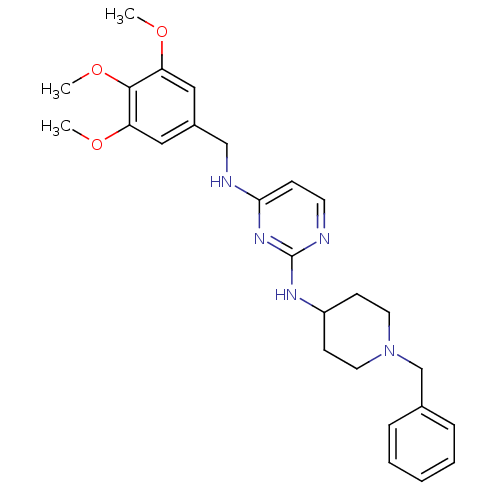
(CHEMBL2047231)Show SMILES COc1cc(CNc2ccnc(NC3CCN(Cc4ccccc4)CC3)n2)cc(OC)c1OC Show InChI InChI=1S/C26H33N5O3/c1-32-22-15-20(16-23(33-2)25(22)34-3)17-28-24-9-12-27-26(30-24)29-21-10-13-31(14-11-21)18-19-7-5-4-6-8-19/h4-9,12,15-16,21H,10-11,13-14,17-18H2,1-3H3,(H2,27,28,29,30) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of equine BuChE using butyrylthiocholine iodide as substrate after 5 mins by DTNB method |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50387095

(CHEMBL2047223)Show InChI InChI=1S/C23H26ClN5/c24-21-9-5-4-8-19(21)16-26-22-10-13-25-23(28-22)27-20-11-14-29(15-12-20)17-18-6-2-1-3-7-18/h1-10,13,20H,11-12,14-17H2,(H2,25,26,27,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of human AChE using acetylthiocholine iodide as substrate after 5 mins by DTNB method |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50387092

(CHEMBL2047227)Show SMILES Fc1ccc(CNc2ccnc(NC3CCN(Cc4ccccc4)CC3)n2)cc1 Show InChI InChI=1S/C23H26FN5/c24-20-8-6-18(7-9-20)16-26-22-10-13-25-23(28-22)27-21-11-14-29(15-12-21)17-19-4-2-1-3-5-19/h1-10,13,21H,11-12,14-17H2,(H2,25,26,27,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of human AChE using acetylthiocholine iodide as substrate after 5 mins by DTNB method |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50387094

(CHEMBL2047224)Show SMILES Clc1cccc(CNc2ccnc(NC3CCN(Cc4ccccc4)CC3)n2)c1 Show InChI InChI=1S/C23H26ClN5/c24-20-8-4-7-19(15-20)16-26-22-9-12-25-23(28-22)27-21-10-13-29(14-11-21)17-18-5-2-1-3-6-18/h1-9,12,15,21H,10-11,13-14,16-17H2,(H2,25,26,27,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of human AChE using acetylthiocholine iodide as substrate after 5 mins by DTNB method |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50387093

(CHEMBL2047225)Show SMILES Clc1ccc(CNc2ccnc(NC3CCN(Cc4ccccc4)CC3)n2)cc1 Show InChI InChI=1S/C23H26ClN5/c24-20-8-6-18(7-9-20)16-26-22-10-13-25-23(28-22)27-21-11-14-29(15-12-21)17-19-4-2-1-3-5-19/h1-10,13,21H,11-12,14-17H2,(H2,25,26,27,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of human AChE using acetylthiocholine iodide as substrate after 5 mins by DTNB method |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50387089

(CHEMBL2047231)Show SMILES COc1cc(CNc2ccnc(NC3CCN(Cc4ccccc4)CC3)n2)cc(OC)c1OC Show InChI InChI=1S/C26H33N5O3/c1-32-22-15-20(16-23(33-2)25(22)34-3)17-28-24-9-12-27-26(30-24)29-21-10-13-31(14-11-21)18-19-7-5-4-6-8-19/h4-9,12,15-16,21H,10-11,13-14,17-18H2,1-3H3,(H2,27,28,29,30) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50387090

(CHEMBL2047229)Show SMILES COc1ccc(CNc2ccnc(NC3CCN(Cc4ccccc4)CC3)n2)cc1 Show InChI InChI=1S/C24H29N5O/c1-30-22-9-7-19(8-10-22)17-26-23-11-14-25-24(28-23)27-21-12-15-29(16-13-21)18-20-5-3-2-4-6-20/h2-11,14,21H,12-13,15-18H2,1H3,(H2,25,26,27,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of human AChE using acetylthiocholine iodide as substrate after 5 mins by DTNB method |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50387097

(CHEMBL2047226)Show SMILES Brc1ccc(CNc2ccnc(NC3CCN(Cc4ccccc4)CC3)n2)cc1 Show InChI InChI=1S/C23H26BrN5/c24-20-8-6-18(7-9-20)16-26-22-10-13-25-23(28-22)27-21-11-14-29(15-12-21)17-19-4-2-1-3-5-19/h1-10,13,21H,11-12,14-17H2,(H2,25,26,27,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of human AChE using acetylthiocholine iodide as substrate after 5 mins by DTNB method |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50387096
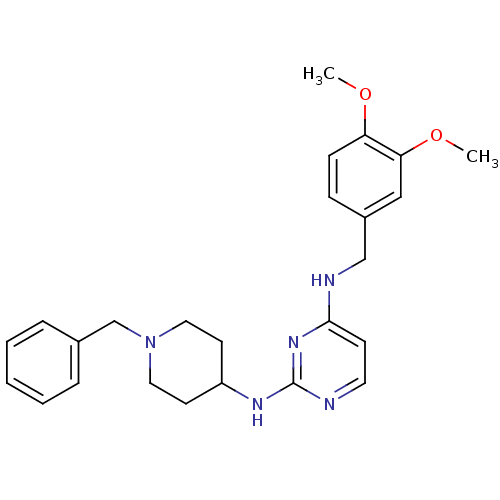
(CHEMBL2047230)Show SMILES COc1ccc(CNc2ccnc(NC3CCN(Cc4ccccc4)CC3)n2)cc1OC Show InChI InChI=1S/C25H31N5O2/c1-31-22-9-8-20(16-23(22)32-2)17-27-24-10-13-26-25(29-24)28-21-11-14-30(15-12-21)18-19-6-4-3-5-7-19/h3-10,13,16,21H,11-12,14-15,17-18H2,1-2H3,(H2,26,27,28,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of human AChE using acetylthiocholine iodide as substrate after 5 mins by DTNB method |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50387089

(CHEMBL2047231)Show SMILES COc1cc(CNc2ccnc(NC3CCN(Cc4ccccc4)CC3)n2)cc(OC)c1OC Show InChI InChI=1S/C26H33N5O3/c1-32-22-15-20(16-23(33-2)25(22)34-3)17-28-24-9-12-27-26(30-24)29-21-10-13-31(14-11-21)18-19-7-5-4-6-8-19/h4-9,12,15-16,21H,10-11,13-14,17-18H2,1-3H3,(H2,27,28,29,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.03E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of human AChE using acetylthiocholine iodide as substrate after 5 mins by DTNB method |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50387091

(CHEMBL2047228)Show SMILES Cc1ccc(CNc2ccnc(NC3CCN(Cc4ccccc4)CC3)n2)cc1 Show InChI InChI=1S/C24H29N5/c1-19-7-9-20(10-8-19)17-26-23-11-14-25-24(28-23)27-22-12-15-29(16-13-22)18-21-5-3-2-4-6-21/h2-11,14,22H,12-13,15-18H2,1H3,(H2,25,26,27,28) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.11E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM50387096

(CHEMBL2047230)Show SMILES COc1ccc(CNc2ccnc(NC3CCN(Cc4ccccc4)CC3)n2)cc1OC Show InChI InChI=1S/C25H31N5O2/c1-31-22-9-8-20(16-23(22)32-2)17-27-24-10-13-26-25(29-24)28-21-11-14-30(15-12-21)18-19-6-4-3-5-7-19/h3-10,13,16,21H,11-12,14-15,17-18H2,1-2H3,(H2,26,27,28,29) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.14E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of equine BuChE using butyrylthiocholine iodide as substrate after 5 mins by DTNB method |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM10404

((1S,12S,14R)-9-methoxy-4-methyl-11-oxa-4-azatetrac...)Show SMILES COc1ccc2CN(C)CC[C@@]34C=C[C@H](O)C[C@@H]3Oc1c24 |r,c:12| Show InChI InChI=1S/C17H21NO3/c1-18-8-7-17-6-5-12(19)9-14(17)21-16-13(20-2)4-3-11(10-18)15(16)17/h3-6,12,14,19H,7-10H2,1-2H3/t12-,14-,17-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.26E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of equine BuChE using butyrylthiocholine iodide as substrate after 5 mins by DTNB method |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50387088

(CHEMBL2047375)Show SMILES C(Nc1ccnc(NC2CCN(Cc3ccccc3)CC2)n1)c1ccc2OCOc2c1 Show InChI InChI=1S/C24H27N5O2/c1-2-4-18(5-3-1)16-29-12-9-20(10-13-29)27-24-25-11-8-23(28-24)26-15-19-6-7-21-22(14-19)31-17-30-21/h1-8,11,14,20H,9-10,12-13,15-17H2,(H2,25,26,27,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.26E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of human AChE using acetylthiocholine iodide as substrate after 5 mins by DTNB method |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50387091

(CHEMBL2047228)Show SMILES Cc1ccc(CNc2ccnc(NC3CCN(Cc4ccccc4)CC3)n2)cc1 Show InChI InChI=1S/C24H29N5/c1-19-7-9-20(10-8-19)17-26-23-11-14-25-24(28-23)27-22-12-15-29(16-13-22)18-21-5-3-2-4-6-21/h2-11,14,22H,12-13,15-18H2,1H3,(H2,25,26,27,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.29E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of human AChE using acetylthiocholine iodide as substrate after 5 mins by DTNB method |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50387087

(CHEMBL2047376)Show SMILES CC(C)C[C@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](NC(=O)OCc1ccccc1)C(C)C)C=O |r| Show InChI InChI=1S/C25H39N3O5/c1-16(2)12-20(14-29)26-23(30)21(13-17(3)4)27-24(31)22(18(5)6)28-25(32)33-15-19-10-8-7-9-11-19/h7-11,14,16-18,20-22H,12-13,15H2,1-6H3,(H,26,30)(H,27,31)(H,28,32)/t20-,21-,22-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Waterloo
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 |
Bioorg Med Chem Lett 22: 4707-12 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.077
BindingDB Entry DOI: 10.7270/Q27S7PSZ |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data