

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50055280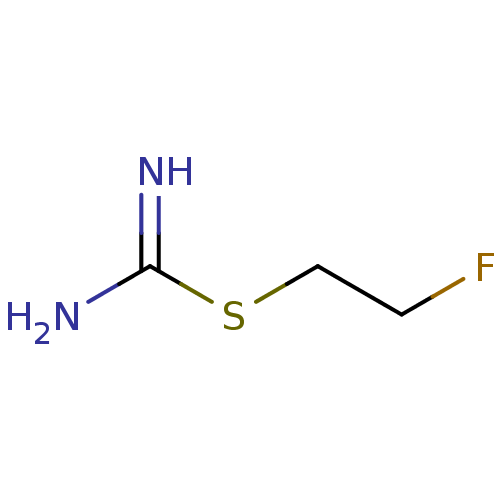 (2-(2-Fluoro-ethyl)-isothiourea | CHEMBL148951) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a | n/a |
Washington University Medical School Curated by ChEMBL | Assay Description The compound was evaluated for the inhibitory potencies using human Inducible nitric oxide synthase (hiNOS) | J Med Chem 39: 5110-8 (1997) Article DOI: 10.1021/jm960481q BindingDB Entry DOI: 10.7270/Q29W0DKZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50055281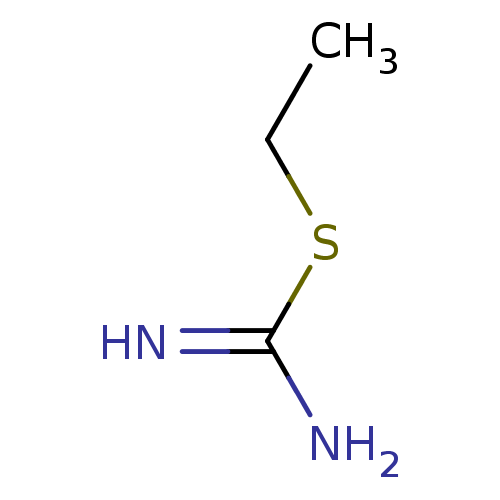 (2-Ethyl-isothiourea | CHEMBL321691 | ETHYLISOTHIOU...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 160 | n/a | n/a | n/a | n/a | n/a | n/a |
Washington University Medical School Curated by ChEMBL | Assay Description The compound was evaluated for the inhibitory potencies using human Inducible nitric oxide synthase (hiNOS) | J Med Chem 39: 5110-8 (1997) Article DOI: 10.1021/jm960481q BindingDB Entry DOI: 10.7270/Q29W0DKZ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50055282 (2-Methyl-isothiourea | CHEMBL356703) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Washington University Medical School Curated by ChEMBL | Assay Description The compound was evaluated for the inhibitory potencies using human Inducible nitric oxide synthase (hiNOS) | J Med Chem 39: 5110-8 (1997) Article DOI: 10.1021/jm960481q BindingDB Entry DOI: 10.7270/Q29W0DKZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50230993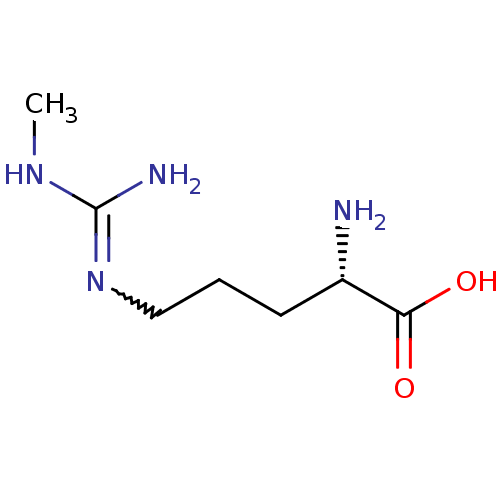 ((2S)-2-amino-5-[(N-methylcarbamimidoyl)amino]penta...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Washington University Medical School Curated by ChEMBL | Assay Description The compound was evaluated for the inhibitory potencies using human Inducible nitric oxide synthase (hiNOS) | J Med Chem 39: 5110-8 (1997) Article DOI: 10.1021/jm960481q BindingDB Entry DOI: 10.7270/Q29W0DKZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||