

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1-deoxy-D-xylulose 5-phosphate reductoisomerase (Escherichia coli) | BDBM59100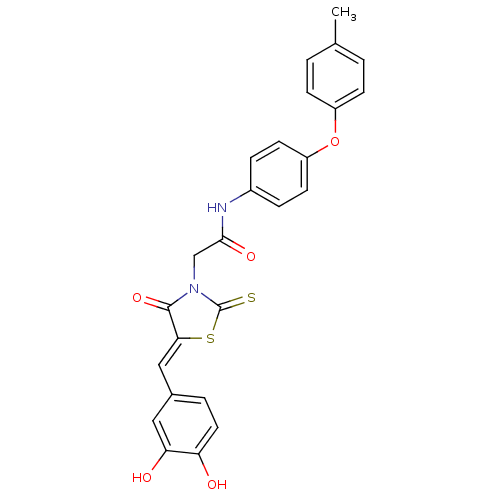 (Bi-ligand, 3) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 202 | n/a | n/a | n/a | n/a | n/a | n/a | 7.4 | n/a |
Triad Therapeutics, Inc | Assay Description All reactions were monitored spectrophotometrically at 340 nm by using initial rates from the first 5% of reaction. | Chem Biol 11: 185-94 (2004) Article DOI: 10.1016/j.chembiol.2004.02.012 BindingDB Entry DOI: 10.7270/Q2K9360M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 1-deoxy-D-xylulose 5-phosphate reductoisomerase (Escherichia coli) | BDBM59100 (Bi-ligand, 3) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Strasbourg/CNRS Curated by ChEMBL | Assay Description Inhibition of His-tagged Escherichia coli DXR pre-incubated for 2 mins before reaction initiation in presence of 160 uM NADPH in absence of 0.01% Tri... | Bioorg Med Chem 22: 3713-9 (2014) Article DOI: 10.1016/j.bmc.2014.05.004 BindingDB Entry DOI: 10.7270/Q2057HKT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||