

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 3-hydroxy-3-methylglutaryl-coenzyme A reductase (Homo sapiens (Human)) | BDBM34168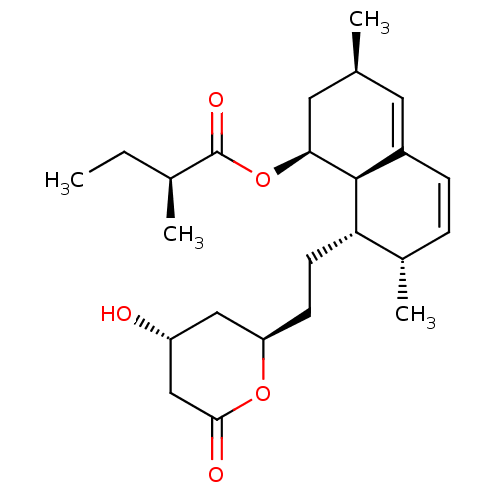 (LOVASTATIN | MLS000069585 | SMR000058779 | US91151...) | PDB UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 2.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Pharmaceutical Sciences, College of Pharmacy and Health Sciences , St. John's University , Queens , New York 11439 , United States. Curated by ChEMBL | Assay Description Inhibition of HMG-CoA reductase (unknown origin) using [14C]-HMG-CoA as substrate after 5 mins in presence of NADPH | J Med Chem 61: 2166-2210 (2018) Article DOI: 10.1021/acs.jmedchem.7b00315 BindingDB Entry DOI: 10.7270/Q2B56N68 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 3-hydroxy-3-methylglutaryl-coenzyme A reductase (Rattus norvegicus (rat)) | BDBM34168 (LOVASTATIN | MLS000069585 | SMR000058779 | US91151...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | n/a | n/a | 2.20 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 3-hydroxy-3-methylglutaryl-coenzyme A reductase (Rattus norvegicus (rat)) | BDBM34168 (LOVASTATIN | MLS000069585 | SMR000058779 | US91151...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Hoechst AG Curated by ChEMBL | Assay Description In vitro inhibition of rat liver HMG-CoA reductase | J Med Chem 33: 52-60 (1990) BindingDB Entry DOI: 10.7270/Q22N5177 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 3-hydroxy-3-methylglutaryl-coenzyme A reductase (Rattus norvegicus (rat)) | BDBM34168 (LOVASTATIN | MLS000069585 | SMR000058779 | US91151...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Hoechst AG Curated by ChEMBL | Assay Description In vitro inhibition of HMG-CoA reductase in solubilized rat liver. | J Med Chem 34: 2962-83 (1991) BindingDB Entry DOI: 10.7270/Q2C829WX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 3-hydroxy-3-methylglutaryl-coenzyme A reductase (Homo sapiens (Human)) | BDBM34168 (LOVASTATIN | MLS000069585 | SMR000058779 | US91151...) | PDB UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company Curated by ChEMBL | Assay Description Tested in vitro for the inhibition of HMG-CoA reductase from partially purified microsomal preparations. | J Med Chem 35: 2095-103 (1992) BindingDB Entry DOI: 10.7270/Q2HH6KN8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 3-hydroxy-3-methylglutaryl-coenzyme A reductase (Rattus norvegicus (rat)) | BDBM34168 (LOVASTATIN | MLS000069585 | SMR000058779 | US91151...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company Curated by ChEMBL | Assay Description In vitro inhibitory activity was measured against rat liver HMG-CoA reductase | J Med Chem 33: 2982-99 (1990) BindingDB Entry DOI: 10.7270/Q26T0N7R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 3-hydroxy-3-methylglutaryl-coenzyme A reductase (Homo sapiens (Human)) | BDBM34168 (LOVASTATIN | MLS000069585 | SMR000058779 | US91151...) | PDB UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | US Patent | n/a | n/a | 29.5 | n/a | n/a | n/a | n/a | 7.4 | n/a |
Academia Sinica; National Taiwan University US Patent | Assay Description The HMGR activity was performed using HMG-CoA reductase assay kit from Sigma-Aldrich with the human recombinant protein or 100 μg total cell lys... | US Patent US9115116 (2015) BindingDB Entry DOI: 10.7270/Q2X34W7F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 3-hydroxy-3-methylglutaryl-coenzyme A reductase (Homo sapiens (Human)) | BDBM34168 (LOVASTATIN | MLS000069585 | SMR000058779 | US91151...) | PDB UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | US Patent | n/a | n/a | 29.5 | n/a | n/a | n/a | n/a | 7.4 | n/a |
ACADEMIA SINICA; NATIONAL TAIWAN UNIVERSITY US Patent | Assay Description The HMGR activity was performed using HMG-CoA reductase assay kit from Sigma-Aldrich with the human recombinant protein or 100 μg total cell lys... | US Patent US9353061 (2016) BindingDB Entry DOI: 10.7270/Q2C24V9M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 3-hydroxy-3-methylglutaryl-coenzyme A reductase (Homo sapiens (Human)) | BDBM34168 (LOVASTATIN | MLS000069585 | SMR000058779 | US91151...) | PDB UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
National Taiwan University Curated by ChEMBL | Assay Description Inhibition of recombinant HMG-CoA reductase (unknown origin) after 10 mins by spectrophotometric analysis | J Med Chem 56: 3645-55 (2013) Article DOI: 10.1021/jm400179b BindingDB Entry DOI: 10.7270/Q2542PXP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 3-hydroxy-3-methylglutaryl-coenzyme A reductase (Rattus norvegicus (rat)) | BDBM34168 (LOVASTATIN | MLS000069585 | SMR000058779 | US91151...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company Curated by ChEMBL | Assay Description In vitro inhibitory activity was measured against rat liver HMG-CoA reductase using [2-14C]-acetate incorporation | J Med Chem 33: 2982-99 (1990) BindingDB Entry DOI: 10.7270/Q26T0N7R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 3-hydroxy-3-methylglutaryl-coenzyme A reductase (Homo sapiens (Human)) | BDBM34168 (LOVASTATIN | MLS000069585 | SMR000058779 | US91151...) | PDB UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | 37 | n/a | n/a | n/a | n/a | n/a | n/a |
British Bio-technology Limited Curated by ChEMBL | Assay Description Concentration required to inhibit HMG-CoA reductase by 50% was determined in Hep G2 cell line | J Med Chem 35: 3388-93 (1992) BindingDB Entry DOI: 10.7270/Q2HD7W9V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 3-hydroxy-3-methylglutaryl-coenzyme A reductase (Homo sapiens (Human)) | BDBM34168 (LOVASTATIN | MLS000069585 | SMR000058779 | US91151...) | PDB UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
Hoechst AG Curated by ChEMBL | Assay Description Inhibition of cellular HMG-CoA reductase in cultures of human HEP G2 cells, determined by decreased incorporation of sodium [14C]-acetate into choles... | J Med Chem 33: 52-60 (1990) BindingDB Entry DOI: 10.7270/Q22N5177 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 3-hydroxy-3-methylglutaryl-coenzyme A reductase (Homo sapiens (Human)) | BDBM34168 (LOVASTATIN | MLS000069585 | SMR000058779 | US91151...) | PDB UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
Hoechst AG Curated by ChEMBL | Assay Description Inhibition of the incorporation of sodium [14C]acetate into cholesterol in HEP G2 cells. | J Med Chem 34: 2962-83 (1991) BindingDB Entry DOI: 10.7270/Q2C829WX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 3-hydroxy-3-methylglutaryl-coenzyme A reductase (Rattus norvegicus (rat)) | BDBM34168 (LOVASTATIN | MLS000069585 | SMR000058779 | US91151...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | 400 | n/a | n/a | n/a | n/a | n/a | n/a |
R. W. Johnson Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Inhibitory activity against partially purified rat liver HMG-CoA reductase in vitro; 0.23-0.71 | J Med Chem 36: 3674-85 (1994) BindingDB Entry DOI: 10.7270/Q2R78FVG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 3-hydroxy-3-methylglutaryl-coenzyme A reductase (Homo sapiens (Human)) | BDBM34168 (LOVASTATIN | MLS000069585 | SMR000058779 | US91151...) | PDB UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 1.98E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
National Taiwan University Curated by ChEMBL | Assay Description Inhibition of HMG-CoA reductase in human A549 cells after 5 mins by spectrophotometric analysis | J Med Chem 56: 3645-55 (2013) Article DOI: 10.1021/jm400179b BindingDB Entry DOI: 10.7270/Q2542PXP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 3-hydroxy-3-methylglutaryl-coenzyme A reductase (Homo sapiens (Human)) | BDBM34168 (LOVASTATIN | MLS000069585 | SMR000058779 | US91151...) | PDB UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 2.01E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Central Drug Research Institute (CSIR) Curated by ChEMBL | Assay Description Inhibition of HMG-CoA reductase using HMG-CoA as substrate by spectrophotometry in presence of NADPH | Eur J Med Chem 46: 5206-11 (2011) Article DOI: 10.1016/j.ejmech.2011.08.012 BindingDB Entry DOI: 10.7270/Q2M32W50 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||