 Found 10 hits Enz. Inhib. hit(s) with Target = 'Histone deacetylase 1' and Ligand = 'BDBM50048864'
Found 10 hits Enz. Inhib. hit(s) with Target = 'Histone deacetylase 1' and Ligand = 'BDBM50048864' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Histone deacetylase 1
(Homo sapiens (Human)) | BDBM50048864
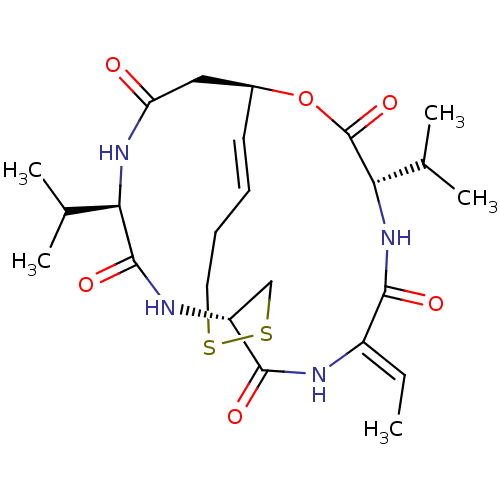
(CHEMBL3310505)Show SMILES [H][C@@]12CC(=O)N[C@H](C(C)C)C(=O)N[C@]([H])(CSSCC\C=C\1)C(=O)N\C(=C/C)C(=O)N[C@@H](C(C)C)C(=O)O2 |r,t:20| Show InChI InChI=1S/C24H36N4O6S2/c1-6-16-21(30)28-20(14(4)5)24(33)34-15-9-7-8-10-35-36-12-17(22(31)25-16)26-23(32)19(13(2)3)27-18(29)11-15/h6-7,9,13-15,17,19-20H,8,10-12H2,1-5H3,(H,25,31)(H,26,32)(H,27,29)(H,28,30)/b9-7+,16-6-/t15-,17+,19+,20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Rajshahi
Curated by ChEMBL
| Assay Description
Inhibition of human HDAC1 using fluorescent substrate Ac-KGLGK(Ac)-MCA after 30 mins by fluorescence plate reader in presence of 0.1 mM dithiothreito... |
Bioorg Med Chem 22: 3850-5 (2014)
Article DOI: 10.1016/j.bmc.2014.06.029
BindingDB Entry DOI: 10.7270/Q23F4R9P |
More data for this
Ligand-Target Pair | |
Histone deacetylase 1
(Homo sapiens (Human)) | BDBM50048864

(CHEMBL3310505)Show SMILES [H][C@@]12CC(=O)N[C@H](C(C)C)C(=O)N[C@]([H])(CSSCC\C=C\1)C(=O)N\C(=C/C)C(=O)N[C@@H](C(C)C)C(=O)O2 |r,t:20| Show InChI InChI=1S/C24H36N4O6S2/c1-6-16-21(30)28-20(14(4)5)24(33)34-15-9-7-8-10-35-36-12-17(22(31)25-16)26-23(32)19(13(2)3)27-18(29)11-15/h6-7,9,13-15,17,19-20H,8,10-12H2,1-5H3,(H,25,31)(H,26,32)(H,27,29)(H,28,30)/b9-7+,16-6-/t15-,17+,19+,20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of HDAC1 (unknown origin) |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00830
BindingDB Entry DOI: 10.7270/Q2F76H9K |
More data for this
Ligand-Target Pair | |
Histone deacetylase 1
(Homo sapiens (Human)) | BDBM50048864

(CHEMBL3310505)Show SMILES [H][C@@]12CC(=O)N[C@H](C(C)C)C(=O)N[C@]([H])(CSSCC\C=C\1)C(=O)N\C(=C/C)C(=O)N[C@@H](C(C)C)C(=O)O2 |r,t:20| Show InChI InChI=1S/C24H36N4O6S2/c1-6-16-21(30)28-20(14(4)5)24(33)34-15-9-7-8-10-35-36-12-17(22(31)25-16)26-23(32)19(13(2)3)27-18(29)11-15/h6-7,9,13-15,17,19-20H,8,10-12H2,1-5H3,(H,25,31)(H,26,32)(H,27,29)(H,28,30)/b9-7+,16-6-/t15-,17+,19+,20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of HDAC1 in human DU-145 cells assessed as increase in histone H3 acetylation after 24 hrs by immunofluorescence microscopy |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01928
BindingDB Entry DOI: 10.7270/Q2H41WB8 |
More data for this
Ligand-Target Pair | |
Histone deacetylase 1
(Homo sapiens (Human)) | BDBM50048864

(CHEMBL3310505)Show SMILES [H][C@@]12CC(=O)N[C@H](C(C)C)C(=O)N[C@]([H])(CSSCC\C=C\1)C(=O)N\C(=C/C)C(=O)N[C@@H](C(C)C)C(=O)O2 |r,t:20| Show InChI InChI=1S/C24H36N4O6S2/c1-6-16-21(30)28-20(14(4)5)24(33)34-15-9-7-8-10-35-36-12-17(22(31)25-16)26-23(32)19(13(2)3)27-18(29)11-15/h6-7,9,13-15,17,19-20H,8,10-12H2,1-5H3,(H,25,31)(H,26,32)(H,27,29)(H,28,30)/b9-7+,16-6-/t15-,17+,19+,20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02067
BindingDB Entry DOI: 10.7270/Q2XD15PJ |
More data for this
Ligand-Target Pair | |
Histone deacetylase 1
(Homo sapiens (Human)) | BDBM50048864

(CHEMBL3310505)Show SMILES [H][C@@]12CC(=O)N[C@H](C(C)C)C(=O)N[C@]([H])(CSSCC\C=C\1)C(=O)N\C(=C/C)C(=O)N[C@@H](C(C)C)C(=O)O2 |r,t:20| Show InChI InChI=1S/C24H36N4O6S2/c1-6-16-21(30)28-20(14(4)5)24(33)34-15-9-7-8-10-35-36-12-17(22(31)25-16)26-23(32)19(13(2)3)27-18(29)11-15/h6-7,9,13-15,17,19-20H,8,10-12H2,1-5H3,(H,25,31)(H,26,32)(H,27,29)(H,28,30)/b9-7+,16-6-/t15-,17+,19+,20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Naples Federico II
Curated by ChEMBL
| Assay Description
Inhibition of HADC1 (unknown origin) |
J Med Chem 63: 23-39 (2020)
Article DOI: 10.1021/acs.jmedchem.9b00924
BindingDB Entry DOI: 10.7270/Q2280BWS |
More data for this
Ligand-Target Pair | |
Histone deacetylase 1
(Homo sapiens (Human)) | BDBM50048864

(CHEMBL3310505)Show SMILES [H][C@@]12CC(=O)N[C@H](C(C)C)C(=O)N[C@]([H])(CSSCC\C=C\1)C(=O)N\C(=C/C)C(=O)N[C@@H](C(C)C)C(=O)O2 |r,t:20| Show InChI InChI=1S/C24H36N4O6S2/c1-6-16-21(30)28-20(14(4)5)24(33)34-15-9-7-8-10-35-36-12-17(22(31)25-16)26-23(32)19(13(2)3)27-18(29)11-15/h6-7,9,13-15,17,19-20H,8,10-12H2,1-5H3,(H,25,31)(H,26,32)(H,27,29)(H,28,30)/b9-7+,16-6-/t15-,17+,19+,20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human HDAC1 expressed in 293T cells using Ac-KGLGK(Ac)-MCA as substrate incubated for 30 mins in presence of 0.1 mM DTT by fluorescence... |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.031
BindingDB Entry DOI: 10.7270/Q2ZP49T6 |
More data for this
Ligand-Target Pair | |
Histone deacetylase 1
(Homo sapiens (Human)) | BDBM50048864

(CHEMBL3310505)Show SMILES [H][C@@]12CC(=O)N[C@H](C(C)C)C(=O)N[C@]([H])(CSSCC\C=C\1)C(=O)N\C(=C/C)C(=O)N[C@@H](C(C)C)C(=O)O2 |r,t:20| Show InChI InChI=1S/C24H36N4O6S2/c1-6-16-21(30)28-20(14(4)5)24(33)34-15-9-7-8-10-35-36-12-17(22(31)25-16)26-23(32)19(13(2)3)27-18(29)11-15/h6-7,9,13-15,17,19-20H,8,10-12H2,1-5H3,(H,25,31)(H,26,32)(H,27,29)(H,28,30)/b9-7+,16-6-/t15-,17+,19+,20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 36 | n/a | n/a | n/a | n/a | n/a | n/a |
Indian CSIR-Central Drug Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of HDAC1 (unknown origin) |
Eur J Med Chem 158: 620-706 (2018)
Article DOI: 10.1016/j.ejmech.2018.08.073
BindingDB Entry DOI: 10.7270/Q2HT2STK |
More data for this
Ligand-Target Pair | |
Histone deacetylase 1
(Homo sapiens (Human)) | BDBM50048864

(CHEMBL3310505)Show SMILES [H][C@@]12CC(=O)N[C@H](C(C)C)C(=O)N[C@]([H])(CSSCC\C=C\1)C(=O)N\C(=C/C)C(=O)N[C@@H](C(C)C)C(=O)O2 |r,t:20| Show InChI InChI=1S/C24H36N4O6S2/c1-6-16-21(30)28-20(14(4)5)24(33)34-15-9-7-8-10-35-36-12-17(22(31)25-16)26-23(32)19(13(2)3)27-18(29)11-15/h6-7,9,13-15,17,19-20H,8,10-12H2,1-5H3,(H,25,31)(H,26,32)(H,27,29)(H,28,30)/b9-7+,16-6-/t15-,17+,19+,20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 36 | n/a | n/a | n/a | n/a | n/a | n/a |
Hunan University of Science and Technology
Curated by ChEMBL
| Assay Description
Inhibition of HDAC1 (unknown origin) |
Eur J Med Chem 183: (2019)
Article DOI: 10.1016/j.ejmech.2019.111691
BindingDB Entry DOI: 10.7270/Q2BR8WGC |
More data for this
Ligand-Target Pair | |
Histone deacetylase 1
(Homo sapiens (Human)) | BDBM50048864

(CHEMBL3310505)Show SMILES [H][C@@]12CC(=O)N[C@H](C(C)C)C(=O)N[C@]([H])(CSSCC\C=C\1)C(=O)N\C(=C/C)C(=O)N[C@@H](C(C)C)C(=O)O2 |r,t:20| Show InChI InChI=1S/C24H36N4O6S2/c1-6-16-21(30)28-20(14(4)5)24(33)34-15-9-7-8-10-35-36-12-17(22(31)25-16)26-23(32)19(13(2)3)27-18(29)11-15/h6-7,9,13-15,17,19-20H,8,10-12H2,1-5H3,(H,25,31)(H,26,32)(H,27,29)(H,28,30)/b9-7+,16-6-/t15-,17+,19+,20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 36 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmc.2021.116510
BindingDB Entry DOI: 10.7270/Q25M69NW |
More data for this
Ligand-Target Pair | |
Histone deacetylase 1
(Homo sapiens (Human)) | BDBM50048864

(CHEMBL3310505)Show SMILES [H][C@@]12CC(=O)N[C@H](C(C)C)C(=O)N[C@]([H])(CSSCC\C=C\1)C(=O)N\C(=C/C)C(=O)N[C@@H](C(C)C)C(=O)O2 |r,t:20| Show InChI InChI=1S/C24H36N4O6S2/c1-6-16-21(30)28-20(14(4)5)24(33)34-15-9-7-8-10-35-36-12-17(22(31)25-16)26-23(32)19(13(2)3)27-18(29)11-15/h6-7,9,13-15,17,19-20H,8,10-12H2,1-5H3,(H,25,31)(H,26,32)(H,27,29)(H,28,30)/b9-7+,16-6-/t15-,17+,19+,20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human HDAC1 |
Bioorg Med Chem Lett 26: 375-9 (2016)
Article DOI: 10.1016/j.bmcl.2015.12.007
BindingDB Entry DOI: 10.7270/Q251413S |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data