

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| P-selectin (Homo sapiens (Human)) | BDBM50450369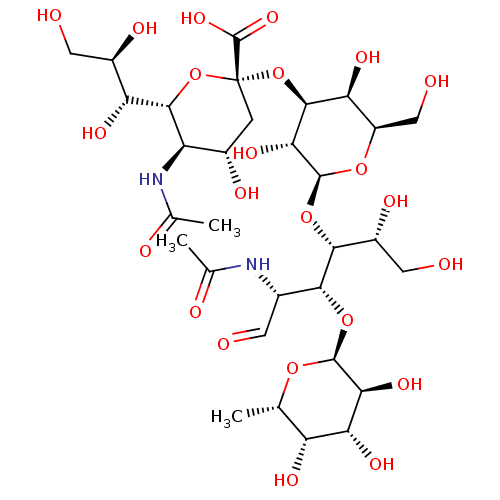 (SIALYL LEWIS X | Sialyl LeX | Sialyl lewis-x | sLe...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Similars | PubMed | n/a | n/a | >1.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Nippon Organon K.K. Curated by ChEMBL | Assay Description In vitro inhibitory activity of compound was determined against Selectin P binding by ELISA assay | Bioorg Med Chem Lett 10: 1827-30 (2000) BindingDB Entry DOI: 10.7270/Q2ZP46M8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P-selectin (Homo sapiens (Human)) | BDBM50450369 (SIALYL LEWIS X | Sialyl LeX | Sialyl lewis-x | sLe...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
New Drug Research Laboratory Curated by ChEMBL | Assay Description Inhibitory concentration against blocking of Selectin P during migration of inflammatory cells to inflammatory site | J Med Chem 39: 2055-9 (1996) Article DOI: 10.1021/jm950877m BindingDB Entry DOI: 10.7270/Q2KH0P0B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P-selectin (Homo sapiens (Human)) | BDBM50450369 (SIALYL LEWIS X | Sialyl LeX | Sialyl lewis-x | sLe...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Kanebo Curated by ChEMBL | Assay Description Inhibition of Selectin P binding | J Med Chem 41: 4279-87 (1998) Article DOI: 10.1021/jm980267x BindingDB Entry DOI: 10.7270/Q2474BHN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P-selectin (Mus musculus) | BDBM50450369 (SIALYL LEWIS X | Sialyl LeX | Sialyl lewis-x | sLe...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Kanebo Curated by ChEMBL | Assay Description Blocking activity using seletin-IgG chimeras in a Selectin P competitive binding assay. | J Med Chem 40: 3534-41 (1997) Article DOI: 10.1021/jm970262k BindingDB Entry DOI: 10.7270/Q2NP253K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P-selectin (Homo sapiens (Human)) | BDBM50450369 (SIALYL LEWIS X | Sialyl LeX | Sialyl lewis-x | sLe...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Cancer Research Laboratories Curated by ChEMBL | Assay Description Inhibitory activity against human chimeric Selectin P-Ig | J Med Chem 39: 1339-43 (1996) Article DOI: 10.1021/jm9506478 BindingDB Entry DOI: 10.7270/Q2513ZWW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P-selectin (Homo sapiens (Human)) | BDBM50450369 (SIALYL LEWIS X | Sialyl LeX | Sialyl lewis-x | sLe...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.30E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Scripps Research Institute Curated by ChEMBL | Assay Description Compound was tested in a cell-free SLe-polyacrylamide glycoconjugate binding assay (assay B) in P-selectin; value ranges from 1.3-2 | Bioorg Med Chem Lett 8: 2333-8 (1999) BindingDB Entry DOI: 10.7270/Q2NV9JR1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P-selectin (Homo sapiens (Human)) | BDBM50450369 (SIALYL LEWIS X | Sialyl LeX | Sialyl lewis-x | sLe...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 2.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Texas Biotechnology Corporation Curated by ChEMBL | Assay Description In vitro inhibitory activity against sialyl Lewis X expressing HL60 cell binding Selectin P | J Med Chem 38: 4976-84 (1996) BindingDB Entry DOI: 10.7270/Q2B56KCP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P-selectin (Homo sapiens (Human)) | BDBM50450369 (SIALYL LEWIS X | Sialyl LeX | Sialyl lewis-x | sLe...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Similars | PubMed | n/a | n/a | >3.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Scripps Research Institute Curated by ChEMBL | Assay Description Tested in a cell-free SLe-polyacrylamide glycoconjugate binding assay (assay A) in P-selectin | Bioorg Med Chem Lett 8: 2333-8 (1999) BindingDB Entry DOI: 10.7270/Q2NV9JR1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P-selectin (Homo sapiens (Human)) | BDBM50450369 (SIALYL LEWIS X | Sialyl LeX | Sialyl lewis-x | sLe...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.40E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Texas Biotechnology Corporation Curated by ChEMBL | Assay Description Tested in vitro for the concentration to inhibit sLex bearing HL-60 cells binding to Selectin P-IgG fusion proteins | J Med Chem 41: 1099-111 (1998) Article DOI: 10.1021/jm9704917 BindingDB Entry DOI: 10.7270/Q2BP03G4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P-selectin (Homo sapiens (Human)) | BDBM50450369 (SIALYL LEWIS X | Sialyl LeX | Sialyl lewis-x | sLe...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 5.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Montr£al Curated by ChEMBL | Assay Description Concentration required to inhibit the interaction of Selectin P and HL-60 cells | Bioorg Med Chem Lett 11: 1109-12 (2001) BindingDB Entry DOI: 10.7270/Q28C9WSC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P-selectin (Homo sapiens (Human)) | BDBM50450369 (SIALYL LEWIS X | Sialyl LeX | Sialyl lewis-x | sLe...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 5.50E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Montr£al Curated by ChEMBL | Assay Description Concentration required to inhibit the interaction of Selectin P and HL-60 cells | Bioorg Med Chem Lett 11: 1109-12 (2001) BindingDB Entry DOI: 10.7270/Q28C9WSC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P-selectin (Homo sapiens (Human)) | BDBM50450369 (SIALYL LEWIS X | Sialyl LeX | Sialyl lewis-x | sLe...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 5.50E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Montr£al Curated by ChEMBL | Assay Description Concentration required to inhibit the interaction of Selectin P and HL-60 cells | Bioorg Med Chem Lett 11: 1109-12 (2001) BindingDB Entry DOI: 10.7270/Q28C9WSC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||