

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| P2X purinoceptor 1 (Mus musculus) | BDBM50064800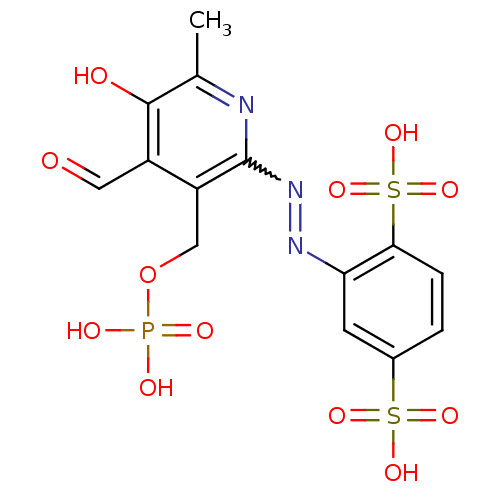 (2-(4-Formyl-5-hydroxy-6-methyl-3-phosphonooxymethy...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 31 | n/a | n/a | n/a | n/a | n/a | n/a |
Gwangju Institute of Science and Technology Curated by ChEMBL | Assay Description Antagonist activity at mouse recombinant P2X1 receptor expressed in Xenopus oocytes assessed as inhibition of ATP-induced ion current incubated for 2... | Bioorg Med Chem 21: 2643-50 (2013) Article DOI: 10.1016/j.bmc.2013.01.073 BindingDB Entry DOI: 10.7270/Q2DF6SK7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P2X purinoceptor 1 (RAT) | BDBM50064800 (2-(4-Formyl-5-hydroxy-6-methyl-3-phosphonooxymethy...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 43 | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Inhibition of inward ion current elicited by ATP at P2X1 receptor expressed in Xenopus oocytes | J Med Chem 44: 340-9 (2001) BindingDB Entry DOI: 10.7270/Q2FN15HC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P2X purinoceptor 1 (RAT) | BDBM50064800 (2-(4-Formyl-5-hydroxy-6-methyl-3-phosphonooxymethy...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.25E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Compound was tested in a functional ion channel assay of ATP-induced current at recombinant rat P2X1 receptor expressed in Xenopus oocytes. | J Med Chem 41: 2201-6 (1998) Article DOI: 10.1021/jm980183o BindingDB Entry DOI: 10.7270/Q2DB80ZC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P2X purinoceptor 1 (RAT) | BDBM50064800 (2-(4-Formyl-5-hydroxy-6-methyl-3-phosphonooxymethy...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 43 | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description The compound was evaluated for antagonist activity against recombinant rat P2X purinoceptor 1 (P2X1) | J Med Chem 45: 4057-93 (2002) BindingDB Entry DOI: 10.7270/Q2VX0H71 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||