

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50032221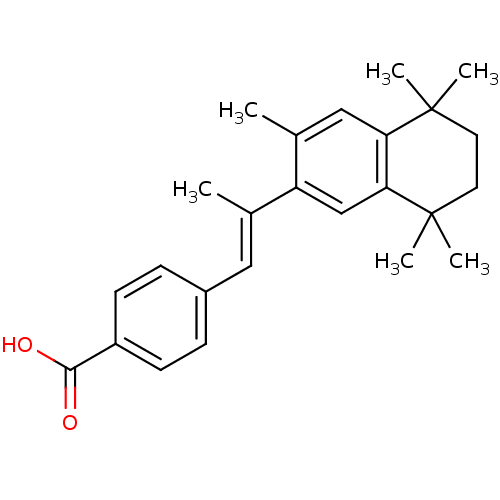 (4-[(E)-2-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a | n/a |
Ligand Pharmaceuticals, Inc Curated by ChEMBL | Assay Description Binding affinity against retinoic Acid X alpha receptor using [3H]- -9-cis-Retinoic Acid in competitive binding assay | J Med Chem 37: 408-14 (1994) BindingDB Entry DOI: 10.7270/Q20V8BT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50032221 (4-[(E)-2-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | n/a | n/a | 385 | n/a | n/a | n/a | n/a |
Allergan, Inc Curated by ChEMBL | Assay Description Transcriptional activation of Retinoid X receptor RXR alpha | J Med Chem 38: 2820-9 (1995) BindingDB Entry DOI: 10.7270/Q2FN157S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50032221 (4-[(E)-2-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article | n/a | n/a | n/a | n/a | 385 | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Transcriptional activation for RXR alpha receptor | Bioorg Med Chem Lett 4: 1447-1452 (1994) Article DOI: 10.1016/S0960-894X(01)80511-4 BindingDB Entry DOI: 10.7270/Q2B8582K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50032221 (4-[(E)-2-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a |
Universidad de Santiago de Compostela Curated by ChEMBL | Assay Description Dissociation constant for Retinoid X receptor alpha | J Med Chem 48: 6212-9 (2005) Article DOI: 10.1021/jm050285w BindingDB Entry DOI: 10.7270/Q26D5SJ6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50032221 (4-[(E)-2-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article | n/a | n/a | n/a | n/a | 1.20E+3 | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Agonist activity for retinoic acid receptor RXR alpha in transcriptional activation assay | Bioorg Med Chem Lett 5: 2729-2734 (1995) Article DOI: 10.1016/0960-894X(95)00455-3 BindingDB Entry DOI: 10.7270/Q22N527N | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50032221 (4-[(E)-2-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | n/a | n/a | 1.20E+3 | n/a | n/a | n/a | n/a |
Ligand Pharmaceuticals, Inc Curated by ChEMBL | Assay Description Binding affinity against retinoic Acid X alpha receptors cotransfected into CV-1 cells | J Med Chem 37: 408-14 (1994) BindingDB Entry DOI: 10.7270/Q20V8BT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||