

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Reverse transcriptase (Human immunodeficiency virus 1) | BDBM50075214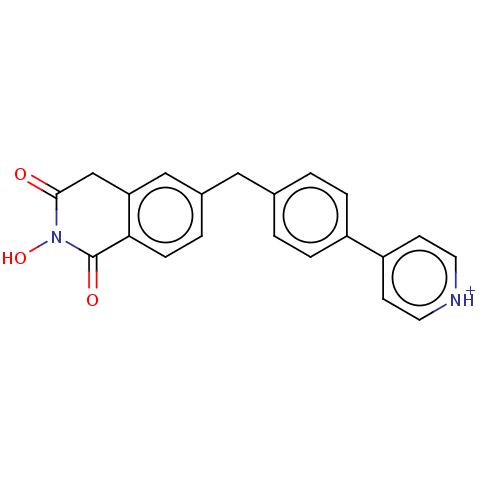 (CHEMBL3414876) | PDB MMDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 900 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Minnesota Curated by ChEMBL | Assay Description Inhibition of HIV1 recombinant reverse transcriptase polymerase activity using [3H]TTP and poly(rA)-oligo(dT)16 substrate incubated for 20 mins by li... | J Med Chem 58: 651-64 (2015) Article DOI: 10.1021/jm501132s BindingDB Entry DOI: 10.7270/Q2W95BWG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase (Human immunodeficiency virus 1) | BDBM50075214 (CHEMBL3414876) | PDB MMDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Minnesota Curated by ChEMBL | Assay Description Inhibition of Moloney murine leukemia virus reverse transcriptase polymerase activity using Td100/Pd18 DNA-DNA substrate incubated for 30 mins by flu... | J Med Chem 58: 651-64 (2015) Article DOI: 10.1021/jm501132s BindingDB Entry DOI: 10.7270/Q2W95BWG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||