

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Rhodopsin kinase GRK1 (Homo sapiens (Human)) | BDBM50173320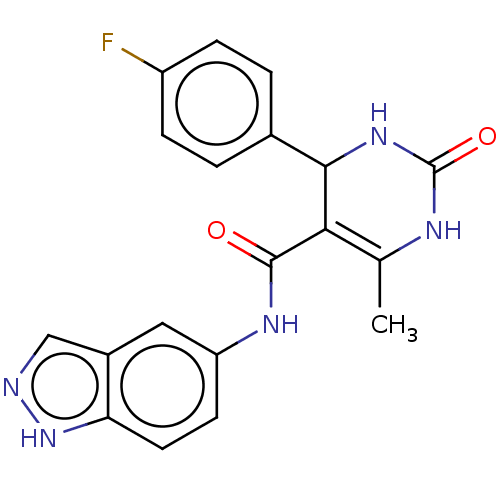 (GSK-180736A | GSK180736A | US10023564, Compound GS...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University Curated by ChEMBL | Assay Description Inhibition of GRK1 (unknown origin) using tubulin as substrate by SDS-PAGE method | J Med Chem 59: 3793-807 (2016) Article DOI: 10.1021/acs.jmedchem.5b02000 BindingDB Entry DOI: 10.7270/Q2JQ12X4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Rhodopsin kinase GRK1 (Bos taurus) | BDBM50173320 (GSK-180736A | GSK180736A | US10023564, Compound GS...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Medicinal Chemistry, College of Pharmacy,?Departments of Pharmacology and Biological Chemistry, Life Sciences Institute,�Ph.D. Program in Chemical Biology,?Vahlteich Medicinal Chemistry Curated by ChEMBL | Assay Description Binding affinity of compound towards dopamine transporter determined using [3H]WIN-35 428 as radioligand | J Med Chem 60: 3052-3069 (2017) Article DOI: 10.1021/acs.jmedchem.7b00112 BindingDB Entry DOI: 10.7270/Q2M047XQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Rhodopsin kinase GRK1 (Homo sapiens (Human)) | BDBM50173320 (GSK-180736A | GSK180736A | US10023564, Compound GS...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem Similars | US Patent | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | 7.0 | n/a |
THE REGENTS OF THE UNIVERSITY OF MICHIGAN US Patent | Assay Description GRK1, 2 and 5 kinetic assays were conducted in a buffer containing 20 mM HEPES pH 7.0, 5 μM ATP, 2 mM MgCl2, and 0.025% DDM with 50 nM GRK and e... | US Patent US10023564 (2018) BindingDB Entry DOI: 10.7270/Q2V126V5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Rhodopsin kinase GRK1 (Bos taurus) | BDBM50173320 (GSK-180736A | GSK180736A | US10023564, Compound GS...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem Similars | PubMed | n/a | n/a | >1.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Messina Curated by ChEMBL | Assay Description Inhibition of bovine GRK1 (1 to 535 residues) assessed as decrease in phosphorylation of tubulin after 5 mins by SDS-PAGE analysis | J Med Chem 59: 9277-9294 (2016) BindingDB Entry DOI: 10.7270/Q2348NB9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||