

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Serine/threonine-protein kinase PLK3 (Homo sapiens (Human)) | BDBM25121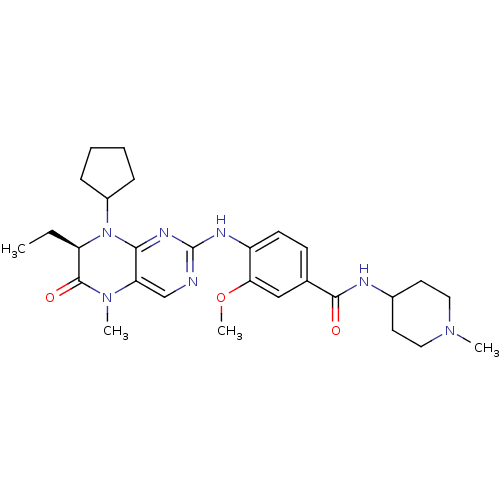 (4-{[(7R)-8-cyclopentyl-7-ethyl-5-methyl-6-oxo-5,6,...) | PDB NCI pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | <1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories | Assay Description Kinase was assayed in 384-well polypropylene plate format. The compound was mixed with kinase and biotinylated peptide substrate and incubated. After... | Biochemistry 46: 9551-63 (2007) Article DOI: 10.1021/bi7008745 BindingDB Entry DOI: 10.7270/Q28G8J11 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine/threonine-protein kinase PLK3 (Homo sapiens (Human)) | BDBM25121 (4-{[(7R)-8-cyclopentyl-7-ethyl-5-methyl-6-oxo-5,6,...) | PDB NCI pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 13.6 | n/a | n/a | n/a | n/a | 7.5 | 22 |
Pfizer | Assay Description Percent inhibition and IC50 values were determined with ATP concentrations at apparent Km using the Invitrogen SelectScreen Kinase Profiling Service ... | Chem Biol Drug Des 70: 540-6 (2007) Article DOI: 10.1111/j.1747-0285.2007.00594.x BindingDB Entry DOI: 10.7270/Q2T151Z8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine/threonine-protein kinase PLK3 (Homo sapiens (Human)) | BDBM25121 (4-{[(7R)-8-cyclopentyl-7-ethyl-5-methyl-6-oxo-5,6,...) | PDB NCI pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
Hefei University of Technology Curated by ChEMBL | Assay Description Inhibition of recombinant human GST-tagged PLK3 (58 to 340 residues) expressed in baculovirus expression system using ser/thr 16 as substrate incubat... | Eur J Med Chem 184: (2019) Article DOI: 10.1016/j.ejmech.2019.111769 BindingDB Entry DOI: 10.7270/Q290278N | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine/threonine-protein kinase PLK3 (Homo sapiens (Human)) | BDBM25121 (4-{[(7R)-8-cyclopentyl-7-ethyl-5-methyl-6-oxo-5,6,...) | PDB NCI pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
Hefei University of Technology Curated by ChEMBL | Assay Description Inhibition human recombinant GST-tagged Plk3 catalytic domain (58 to 340 residues) expressed in baculovirus expression system after 1 hr by FRET-base... | Eur J Med Chem 143: 724-731 (2018) Article DOI: 10.1016/j.ejmech.2017.11.058 BindingDB Entry DOI: 10.7270/Q27D2XPT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine/threonine-protein kinase PLK3 (Homo sapiens (Human)) | BDBM25121 (4-{[(7R)-8-cyclopentyl-7-ethyl-5-methyl-6-oxo-5,6,...) | PDB NCI pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | US Patent | n/a | n/a | 90 | n/a | n/a | n/a | n/a | 7.9 | n/a |
Nerviano Medical Sciences S.r.l. US Patent | Assay Description The inhibitory activity of putative kinase inhibitors and the potency of selected compounds were determined using a trans-phosphorylation assay. | US Patent US8598172 (2013) BindingDB Entry DOI: 10.7270/Q2SJ1J7D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine/threonine-protein kinase PLK3 (Homo sapiens (Human)) | BDBM25121 (4-{[(7R)-8-cyclopentyl-7-ethyl-5-methyl-6-oxo-5,6,...) | PDB NCI pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a |
Ambit Biosciences Curated by ChEMBL | Assay Description Binding constant for PLK3 kinase domain | Nat Biotechnol 29: 1046-51 (2011) Article DOI: 10.1038/nbt.1990 BindingDB Entry DOI: 10.7270/Q25D8S70 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||