

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM4374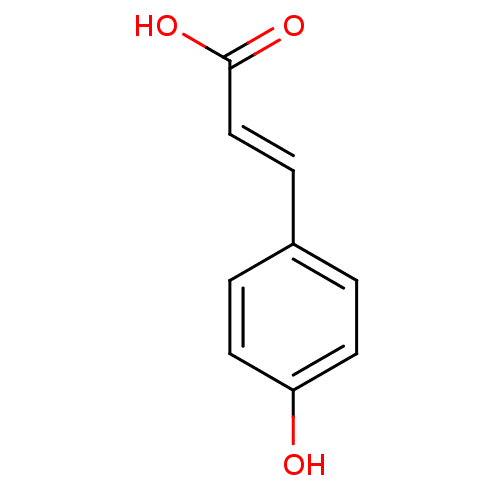 ((2E)-3-(4-hydroxyphenyl)prop-2-enoic acid | (2E)-3...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 1.00E+6 | n/a | 3.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Hebrew University of Jerusalem | Assay Description The activity of EGFR, preactivated with EGF, is measured by its ability to transfer terminal phosphate from [gamma-32P]ATP to poly(GAT) substrate. | J Med Chem 32: 2344-52 (1989) Article DOI: 10.1021/jm00130a020 BindingDB Entry DOI: 10.7270/Q2G44NHF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member B1 (Homo sapiens (Human)) | BDBM4374 ((2E)-3-(4-hydroxyphenyl)prop-2-enoic acid | (2E)-3...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article | n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of aldose reductase (unknown origin) | Citation and Details Article DOI: 10.1007/s00044-010-9412-4 BindingDB Entry DOI: 10.7270/Q26Q214P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Polyphenol oxidase 2 (Agaricus bisporus (Common mushroom)) | BDBM4374 ((2E)-3-(4-hydroxyphenyl)prop-2-enoic acid | (2E)-3...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | n/a | n/a | 296 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosinase (Homo sapiens (Human)) | BDBM4374 ((2E)-3-(4-hydroxyphenyl)prop-2-enoic acid | (2E)-3...) | KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human tyrosinase expressed in HEK293 cells | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00994 BindingDB Entry DOI: 10.7270/Q2PV6Q29 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosinase (Homo sapiens (Human)) | BDBM4374 ((2E)-3-(4-hydroxyphenyl)prop-2-enoic acid | (2E)-3...) | KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of tyrosinase in neonatal human epidermal melanocytes | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00994 BindingDB Entry DOI: 10.7270/Q2PV6Q29 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosinase (Homo sapiens (Human)) | BDBM4374 ((2E)-3-(4-hydroxyphenyl)prop-2-enoic acid | (2E)-3...) | KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn Curated by ChEMBL | Assay Description Inhibition of human tyrosinase after 120 mins | J Med Chem 61: 7395-7418 (2018) Article DOI: 10.1021/acs.jmedchem.7b00967 BindingDB Entry DOI: 10.7270/Q2CN77C2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosinase (Homo sapiens (Human)) | BDBM4374 ((2E)-3-(4-hydroxyphenyl)prop-2-enoic acid | (2E)-3...) | KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn Curated by ChEMBL | Assay Description Inhibition of human tyrosinase after 120 mins | J Med Chem 61: 7395-7418 (2018) Article DOI: 10.1021/acs.jmedchem.7b00967 BindingDB Entry DOI: 10.7270/Q2CN77C2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Xanthine dehydrogenase/oxidase (Bos taurus (Bovine)) | BDBM4374 ((2E)-3-(4-hydroxyphenyl)prop-2-enoic acid | (2E)-3...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 6.80E+3 | n/a | n/a | n/a | n/a | 7.6 | 25 |
SRTM University, Nanded, Maharashtra, 431606, India; Department of Molecular Biotechnology, College of Life and Environmental Sciences, Konkuk University, Seoul, 143-701, South Korea. | Assay Description Inhibition of xanthine oxidase (XO) by each isolated phenolics was measured by following the decrease in the uric acid formation at 293nm at 25°... | Chem Biol Drug Des 83: 119-25 (2014) Article DOI: 10.1111/cbdd.12205 BindingDB Entry DOI: 10.7270/Q2Z89B2H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member B10 (Homo sapiens (Human)) | BDBM4374 ((2E)-3-(4-hydroxyphenyl)prop-2-enoic acid | (2E)-3...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 6.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Gifu Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human recombinant N-terminus His6-tagged AKR1B10 expressed in Escherichia coli BL21 DE3 assessed as pyridine-3-aldehyde reduction by sp... | Eur J Med Chem 48: 321-9 (2012) Article DOI: 10.1016/j.ejmech.2011.12.034 BindingDB Entry DOI: 10.7270/Q2TT4RDB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family 1 member B1 (Homo sapiens (Human)) | BDBM4374 ((2E)-3-(4-hydroxyphenyl)prop-2-enoic acid | (2E)-3...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 7.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Gifu Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human recombinant N-terminus His6-tagged AKR1B1 expressed in Escherichia coli BL21 DE3 assessed as pyridine-3-aldehyde reduction by spe... | Eur J Med Chem 48: 321-9 (2012) Article DOI: 10.1016/j.ejmech.2011.12.034 BindingDB Entry DOI: 10.7270/Q2TT4RDB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Alpha-glucosidase MAL12 (Saccharomyces cerevisiae) | BDBM4374 ((2E)-3-(4-hydroxyphenyl)prop-2-enoic acid | (2E)-3...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 9.08E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
SRTM University, Nanded, Maharashtra, 431606, India; Department of Molecular Biotechnology, College of Life and Environmental Sciences, Konkuk University, Seoul, 143-701, South Korea. | Assay Description The assay was performed using isolated phenolics from maize, and inhibition was determined according to previously described method. | Chem Biol Drug Des 83: 119-25 (2014) Article DOI: 10.1111/cbdd.12205 BindingDB Entry DOI: 10.7270/Q2Z89B2H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Polyphenol oxidase 2 (Agaricus bisporus (Common mushroom)) | BDBM4374 ((2E)-3-(4-hydroxyphenyl)prop-2-enoic acid | (2E)-3...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 1.21E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Kinki University Curated by ChEMBL | Assay Description Inhibition of mushroom tyrosinase after 25 mins by spectrophotometry | Bioorg Med Chem Lett 21: 1983-6 (2011) Article DOI: 10.1016/j.bmcl.2011.02.028 BindingDB Entry DOI: 10.7270/Q2T43TCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein phosphatase non-receptor type 1 [1-321] (Homo sapiens (Human)) | BDBM4374 ((2E)-3-(4-hydroxyphenyl)prop-2-enoic acid | (2E)-3...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 1.68E+5 | n/a | n/a | n/a | n/a | 7.5 | 37 |
Chulalongkorn University | Assay Description PTP1B was diluted before the experiment to 1.2 μg/mL in Tris buffer, pH7.6 (10 mM Tris, 1.0 mM EDTA, 3.0 mM DTT, 0.01% w/v NaN3). The tested com... | J Enzyme Inhib Med Chem 28: 1067-72 (2013) Article DOI: 10.3109/14756366.2012.715286 BindingDB Entry DOI: 10.7270/Q2FN1530 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Alpha-glucosidase MAL62 (Saccharomyces cerevisiae) | BDBM4374 ((2E)-3-(4-hydroxyphenyl)prop-2-enoic acid | (2E)-3...) | Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article | n/a | n/a | >3.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of yeast alpha-glucosidase using p-nitrophenyl-alpha-D-glucopyranoside as substrate preincubated for 10 min before substrate addition and ... | Citation and Details Article DOI: 10.1007/s00044-011-9699-9 BindingDB Entry DOI: 10.7270/Q2930X2H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Alpha-glucosidase MAL62 (Saccharomyces cerevisiae) | BDBM4374 ((2E)-3-(4-hydroxyphenyl)prop-2-enoic acid | (2E)-3...) | Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article | n/a | n/a | 2.00E+11 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of baker's yeast alpha-glucosidase using p-nitrophenyl-alpha-D-glucopyranoside as substrate by spectrophotometry | Citation and Details Article DOI: 10.1007/s00044-011-9938-0 BindingDB Entry DOI: 10.7270/Q29W0JBG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||