 Found 148 hits of ic50 for UniProtKB: P07949
Found 148 hits of ic50 for UniProtKB: P07949 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM576988
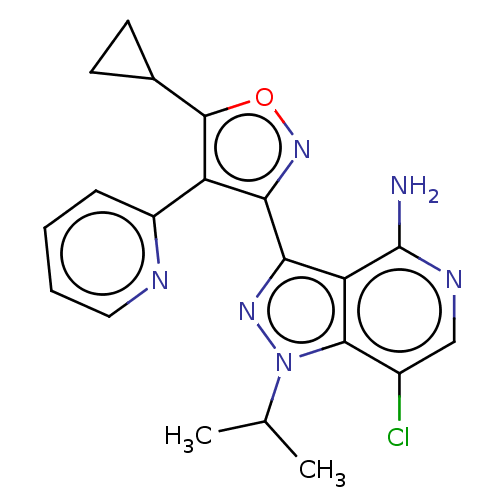
(US11472802, Example 8)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2ccccn2)c2c(N)ncc(Cl)c12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577053

(US11472802, Example 58)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2c[nH]cn2)c2c(N)nccc12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577049

(US11472802, Example 55)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2cn(C)cn2)c2c(N)ncc(Cl)c12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.900 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577050

(7-chloro-3-(5- cyclopropyl-4-(1H- imidazol-4- yl)i...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2c[nH]cn2)c2c(N)ncc(Cl)c12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577046
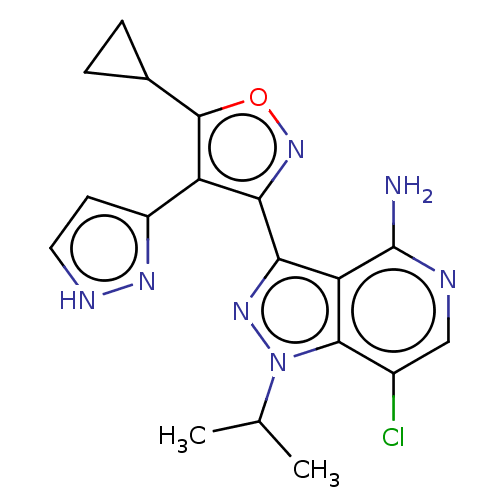
(7-chloro-3-(5-cyclopropyl- 4-(1H-pyrazol-3- yl)iso...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2cc[nH]n2)c2c(N)ncc(Cl)c12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577063
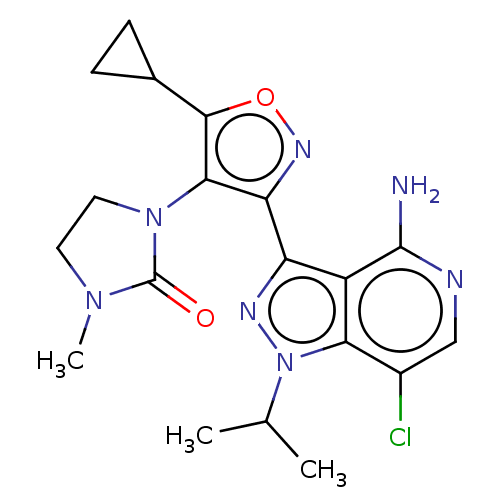
(1-(3-(4-amino-7-chloro-1- isopropyl-1H-pyrazolo[4,...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2N2CCN(C)C2=O)c2c(N)ncc(Cl)c12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577061

(US11472802, Example 65)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2N2CCC2=O)c2c(N)ncc(Cl)c12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM576996

(US11472802, Example 11)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2ccccn2)c2c(N)ncc(F)c12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577064
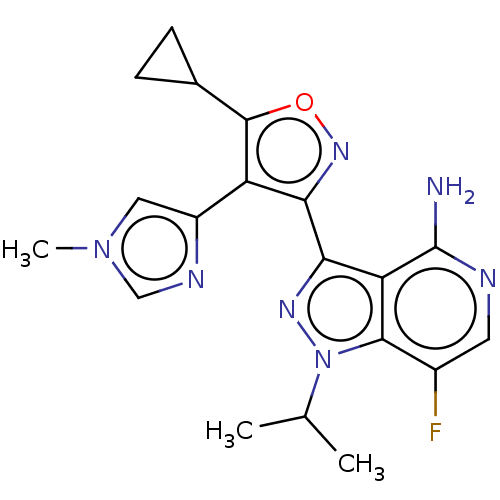
(US11472802, Example 68)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2cn(C)cn2)c2c(N)ncc(F)c12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM597662

(US11603374, Example 17) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The potency of compounds inhibiting several different RET kinase forms (Wild Type, V804M, M918T, G810R, & G810S) were determined using CisBio's H... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2Z323KZ |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577065

(US11472802, Example 69)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2ccn(C)n2)c2c(N)ncc(F)c12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM597647
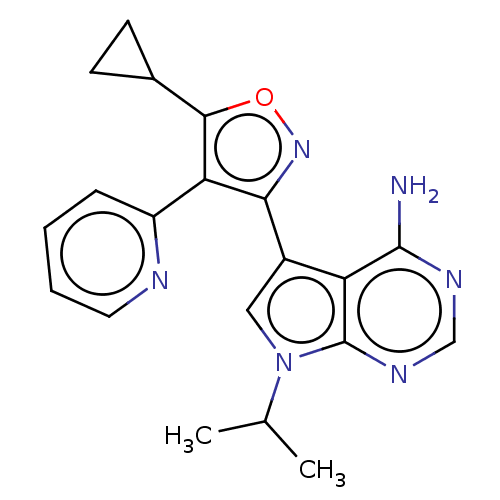
(US11603374, Example 2)Show SMILES CC(C)n1cc(-c2noc(C3CC3)c2-c2ccccn2)c2c(N)ncnc12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The potency of compounds inhibiting several different RET kinase forms (Wild Type, V804M, M918T, G810R, & G810S) were determined using CisBio's H... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2Z323KZ |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577066
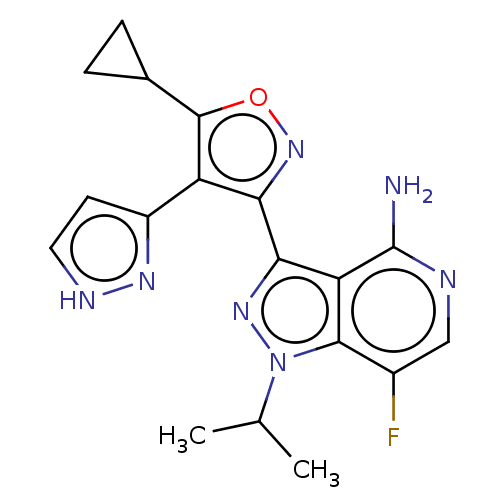
(3-(5-cyclopropyl- 4-(1H-pyrazol-3- yl)isoxazol-3- ...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2cc[nH]n2)c2c(N)ncc(F)c12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577045

(US11472802, Example 51)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2cc(C)[nH]n2)c2c(N)ncc(Cl)c12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577047

(7-chloro-3-(5-cyclopropyl- 4-(1-methyl-1H-pyrazol-...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2ccn(C)n2)c2c(N)ncc(Cl)c12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM597672

((3-(4-amino-5-(5- cyclopropylisoxazol-3-yl)- 7H-py...)Show SMILES Nc1ncnc2n(cc(-c3cc(on3)C3CC3)c12)C1CC(CO)C1 |(-2.78,4.69,;-3.17,3.2,;-4.68,2.88,;-5.16,1.42,;-4.13,.27,;-2.62,.59,;-1.37,-.31,;-.13,.59,;-.6,2.06,;.17,3.39,;1.71,3.39,;2.18,4.85,;.94,5.76,;-.31,4.85,;3.67,5.25,;5.16,4.85,;4.76,6.34,;-2.14,2.06,;-1.37,-1.85,;-2.46,-2.94,;-1.37,-4.03,;-1.37,-5.57,;-.04,-6.34,;-.29,-2.94,)| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The potency of compounds inhibiting several different RET kinase forms (Wild Type, V804M, M918T, G810R, & G810S) were determined using CisBio's H... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2Z323KZ |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM597674

(US11603374, Example 29)Show SMILES Nc1ncnc2n(cc(-c3cc(on3)C3CC3)c12)[C@H]1C[C@H](CO)C1 |r,wU:18.21,wD:20.24,(-2.78,4.69,;-3.17,3.2,;-4.68,2.88,;-5.16,1.42,;-4.13,.27,;-2.62,.59,;-1.37,-.31,;-.13,.59,;-.6,2.06,;.17,3.39,;1.71,3.39,;2.18,4.85,;.94,5.76,;-.31,4.85,;3.67,5.25,;5.16,4.85,;4.76,6.34,;-2.14,2.06,;-1.37,-1.85,;-.29,-2.94,;-1.37,-4.03,;-1.37,-5.57,;-.04,-6.34,;-2.46,-2.94,)| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 2.10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The potency of compounds inhibiting several different RET kinase forms (Wild Type, V804M, M918T, G810R, & G810S) were determined using CisBio's H... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2Z323KZ |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577067
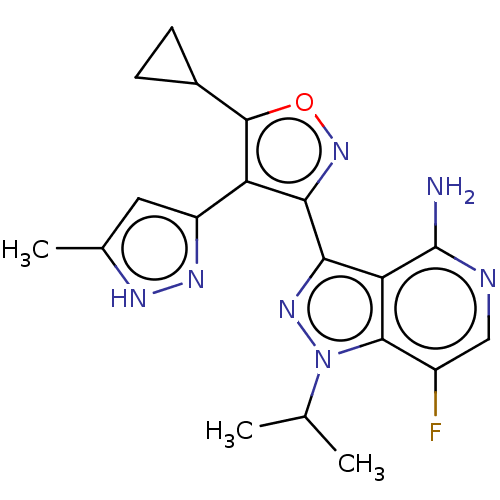
(3-(5-cyclopropyl- 4-(5-methyl-1H- pyrazol-3-yl) is...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2cc(C)[nH]n2)c2c(N)ncc(F)c12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 2.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577062
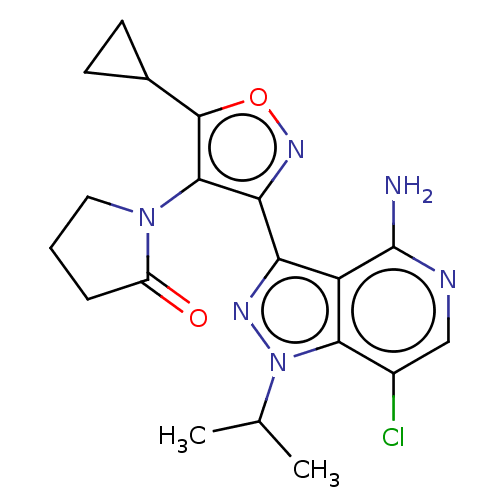
(1-(3-(4-amino-7-chloro-1- isopropyl-1H-pyrazolo[4,...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2N2CCCC2=O)c2c(N)ncc(Cl)c12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM576985

(US11472802, Example 6)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2ccccn2)c2c(N)nccc12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 2.45 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM597648
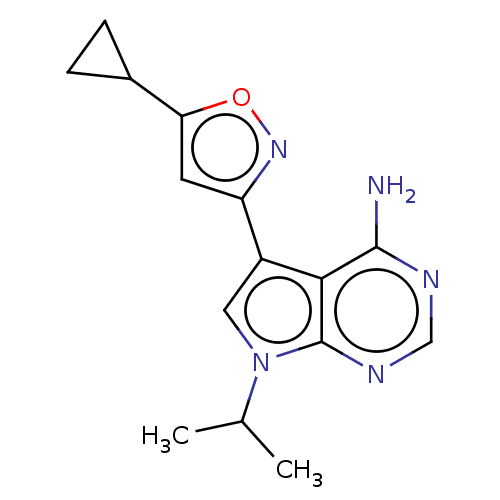
(US11603374, Example 3) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The potency of compounds inhibiting several different RET kinase forms (Wild Type, V804M, M918T, G810R, & G810S) were determined using CisBio's H... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2Z323KZ |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM597646
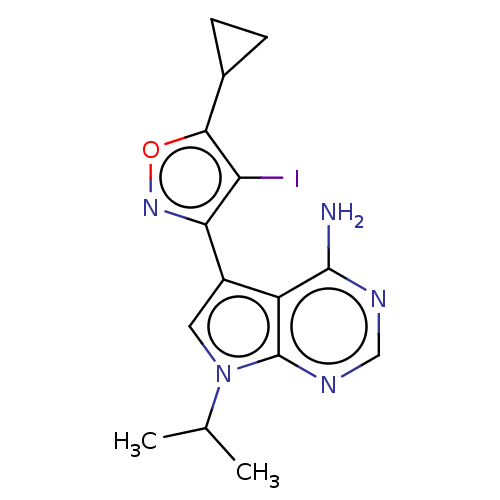
(US11603374, Example 1) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The potency of compounds inhibiting several different RET kinase forms (Wild Type, V804M, M918T, G810R, & G810S) were determined using CisBio's H... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2Z323KZ |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577022
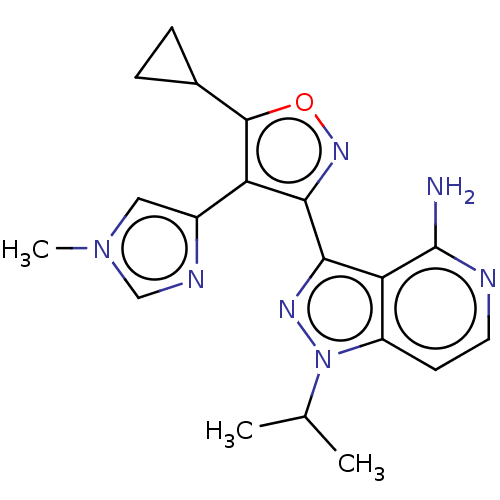
(3-(5-cyclopropyl-4-(1-methyl- 1H-imidazol-4-yl)iso...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2cn(C)cn2)c2c(N)nccc12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577026

(7-chloro-3-(5-cyclopropyl-4-(4- methylpyridin-2-yl...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2cc(C)ccn2)c2c(N)ncc(Cl)c12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 2.70 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM597673
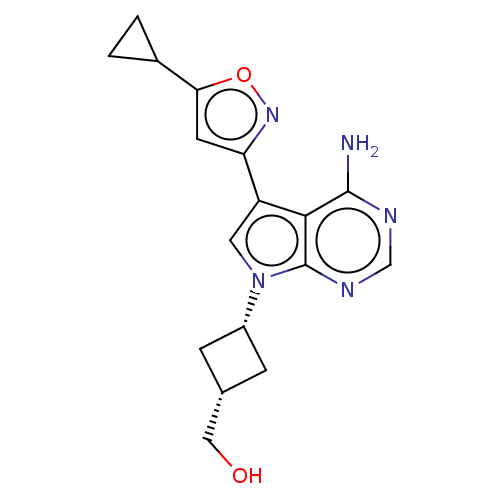
(US11603374, Example 28)Show SMILES Nc1ncnc2n(cc(-c3cc(on3)C3CC3)c12)[C@@H]1C[C@H](CO)C1 |r,wU:18.21,20.24,(-2.78,4.69,;-3.17,3.2,;-4.68,2.88,;-5.16,1.42,;-4.13,.27,;-2.62,.59,;-1.37,-.31,;-.13,.59,;-.6,2.06,;.17,3.39,;1.71,3.39,;2.18,4.85,;.94,5.76,;-.31,4.85,;3.67,5.25,;5.16,4.85,;4.76,6.34,;-2.14,2.06,;-1.37,-1.85,;-2.46,-2.94,;-1.37,-4.03,;-1.37,-5.57,;-.04,-6.34,;-.29,-2.94,)| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 2.90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The potency of compounds inhibiting several different RET kinase forms (Wild Type, V804M, M918T, G810R, & G810S) were determined using CisBio's H... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2Z323KZ |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577025
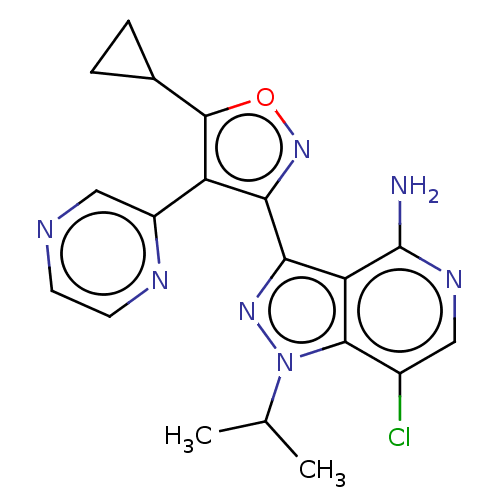
(7-chloro-3-(5-cyclopropyl-4- (pyrazin-2-yl)isoxazo...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2cnccn2)c2c(N)ncc(Cl)c12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 2.90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM597669

(5-(5-cyclopropylisoxazol-3- yl)-7-(1,1,1-trifluoro...)Show SMILES CC(n1cc(-c2cc(on2)C2CC2)c2c(N)ncnc12)C(F)(F)F | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 3.10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The potency of compounds inhibiting several different RET kinase forms (Wild Type, V804M, M918T, G810R, & G810S) were determined using CisBio's H... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2Z323KZ |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577015
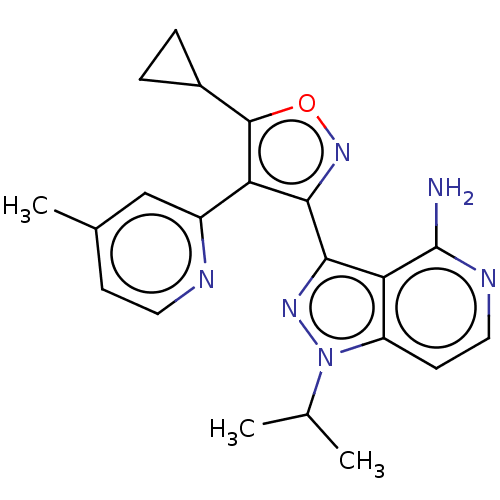
(3-(5-cyclopropyl-4-(4- methylpyridin-2-yl)isoxazol...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2cc(C)ccn2)c2c(N)nccc12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 3.10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577029

(7-chloro-3-(5-cyclopropyl-4-(5- methylpyridin-2-yl...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2ccc(C)cn2)c2c(N)ncc(Cl)c12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM597663
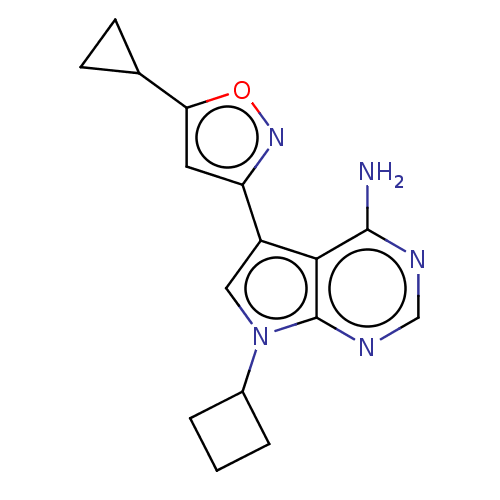
(7-cyclobutyl-5-(5- cyclopropylisoxazol-3-yl)- 7H-p...) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The potency of compounds inhibiting several different RET kinase forms (Wild Type, V804M, M918T, G810R, & G810S) were determined using CisBio's H... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2Z323KZ |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM597666
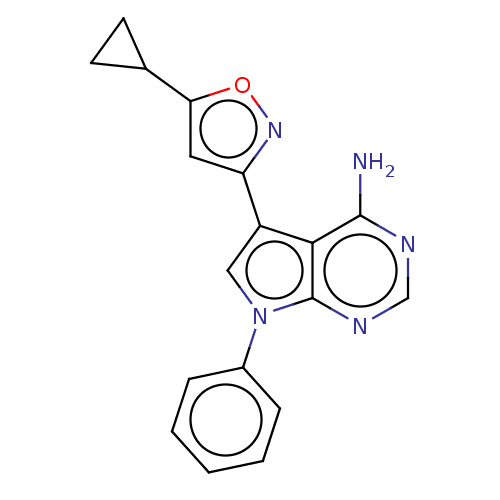
(US11603374, Example 21) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 3.70 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The potency of compounds inhibiting several different RET kinase forms (Wild Type, V804M, M918T, G810R, & G810S) were determined using CisBio's H... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2Z323KZ |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577048

(7-chloro-3-(5-cyclopropyl- 4-(1-ethyl-1H-pyrazol-3...)Show SMILES CCn1ccc(n1)-c1c(noc1C1CC1)-c1nn(C(C)C)c2c(Cl)cnc(N)c12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577007

(US11472802, Example 18)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2ccn(C)n2)c2c(N)nccc12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 4.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577019
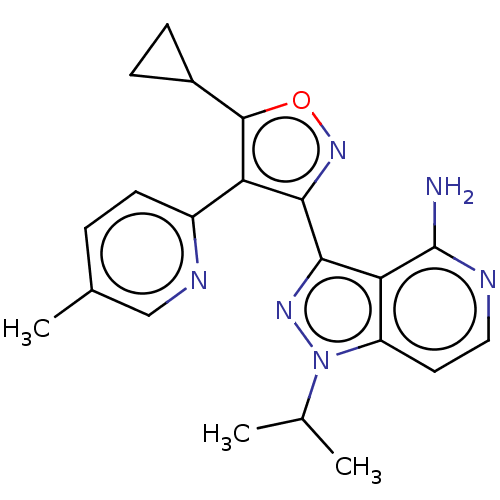
(3-(5-cyclopropyl-4-(5- methylpyridin-2-yl)isoxazol...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2ccc(C)cn2)c2c(N)nccc12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 4.30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577071
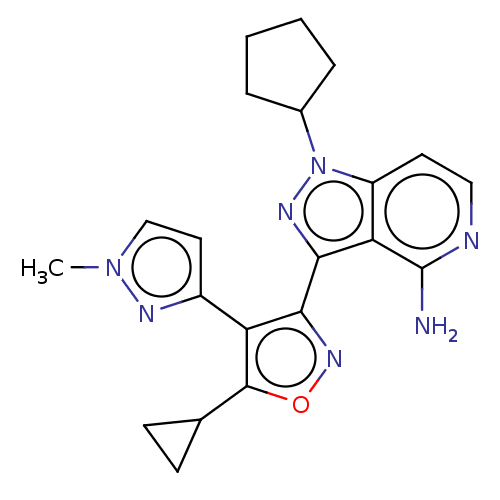
(US11472802, Example 75)Show SMILES Cn1ccc(n1)-c1c(noc1C1CC1)-c1nn(C2CCCC2)c2ccnc(N)c12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 4.60 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM597675
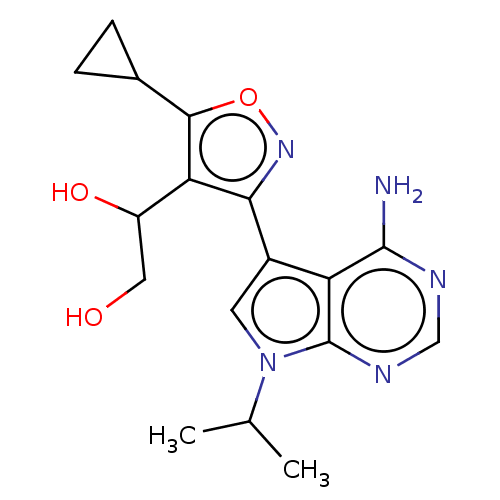
(US11603374, Example 30)Show SMILES CC(C)n1cc(-c2noc(C3CC3)c2C(O)CO)c2c(N)ncnc12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 4.80 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The potency of compounds inhibiting several different RET kinase forms (Wild Type, V804M, M918T, G810R, & G810S) were determined using CisBio's H... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2Z323KZ |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577008
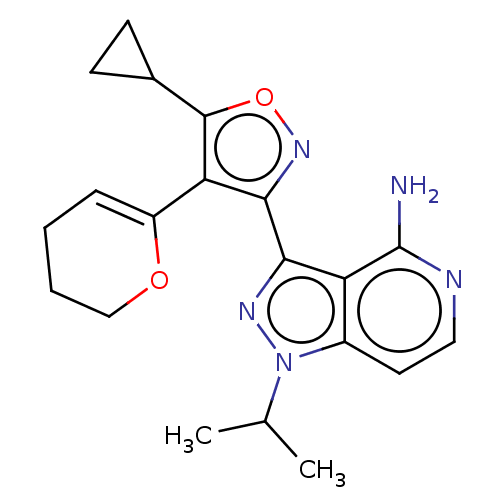
(US11472802, Example 19)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2C2=CCCCO2)c2c(N)nccc12 |t:16| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 5.30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM576971
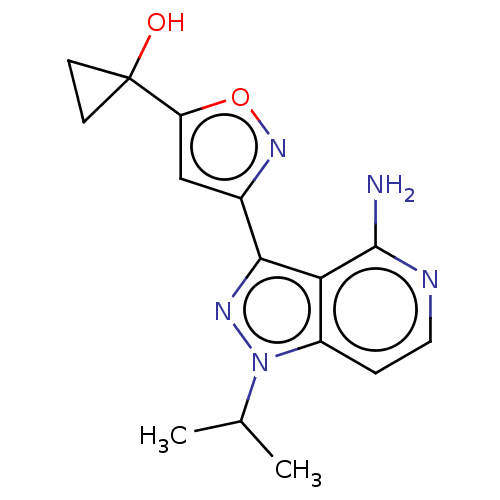
(US11472802, Example 1) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 5.70 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577006
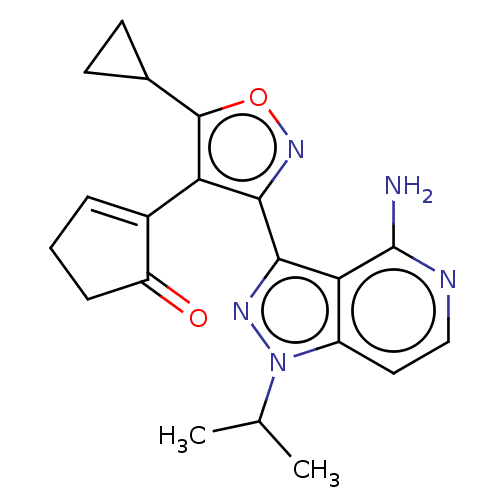
(US11472802, Example 17)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2C2=CCCC2=O)c2c(N)nccc12 |t:16| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577028
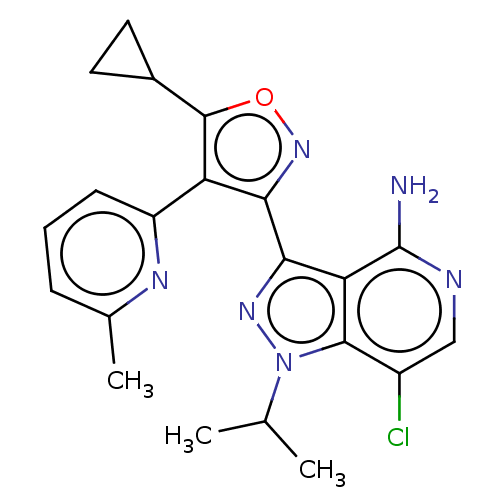
(7-chloro-3-(5-cyclopropyl-4-(6- methylpyridin-2-yl...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2cccc(C)n2)c2c(N)ncc(Cl)c12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577060

(1-(3-(4-amino-1-isopropyl- 1H-pyrazolo[4,3-c]pyrid...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2N2CCN(C)C2=O)c2c(N)nccc12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 6.30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577032
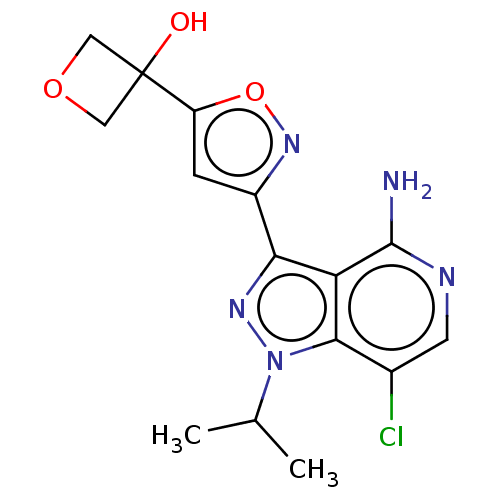
(US11472802, Example 39)Show SMILES CC(C)n1nc(-c2cc(on2)C2(O)COC2)c2c(N)ncc(Cl)c12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 6.40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577013

(3-(5-cyclopropyl-4-(pyrazin-2- yl)isoxazol-3-yl)-1...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2cnccn2)c2c(N)nccc12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 6.90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577042
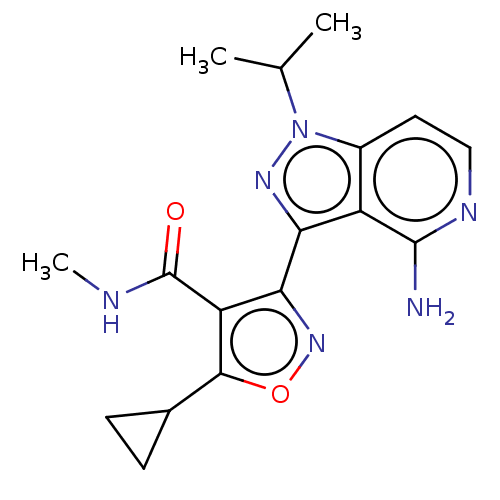
(US11472802, Example 48)Show SMILES CNC(=O)c1c(noc1C1CC1)-c1nn(C(C)C)c2ccnc(N)c12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 7.60 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577018
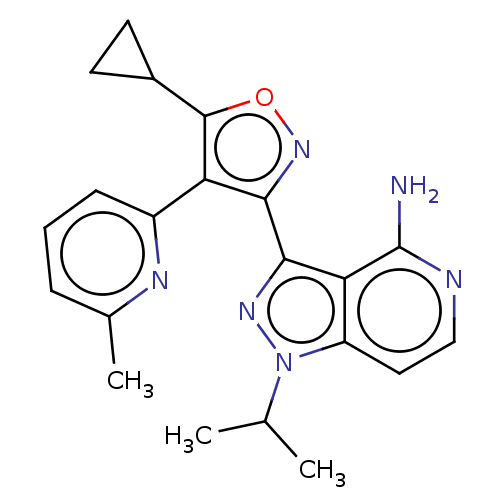
(3-(5-cyclopropyl-4-(6- methylpyridin-2-yl)isoxazol...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2cccc(C)n2)c2c(N)nccc12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 7.60 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577057
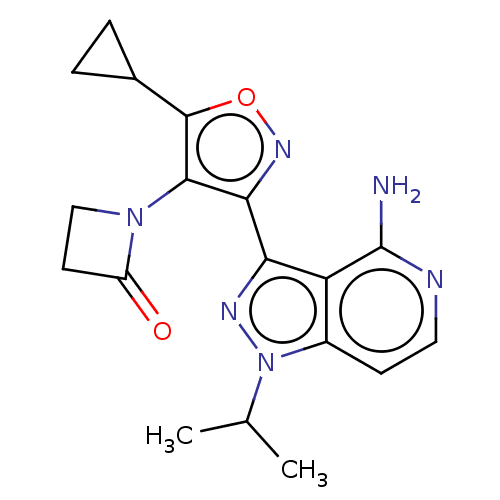
(1-(3-(4-amino-1-isopropyl- 1H-pyrazolo[4,3-c]pyrid...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2N2CCC2=O)c2c(N)nccc12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 8.10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM597668
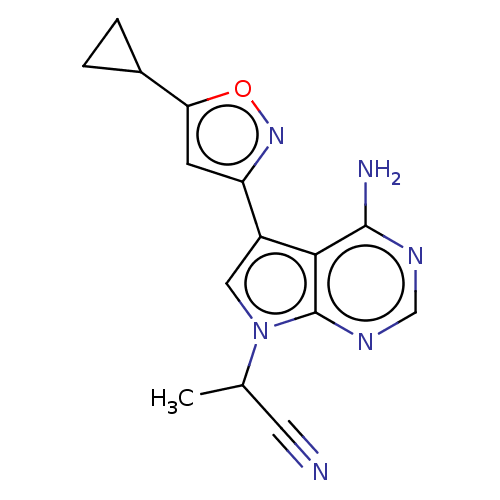
(2-(4-amino-5-(5- cyclopropylisoxazol-3-yl)- 7H-pyr...) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 8.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The potency of compounds inhibiting several different RET kinase forms (Wild Type, V804M, M918T, G810R, & G810S) were determined using CisBio's H... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2Z323KZ |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM597665
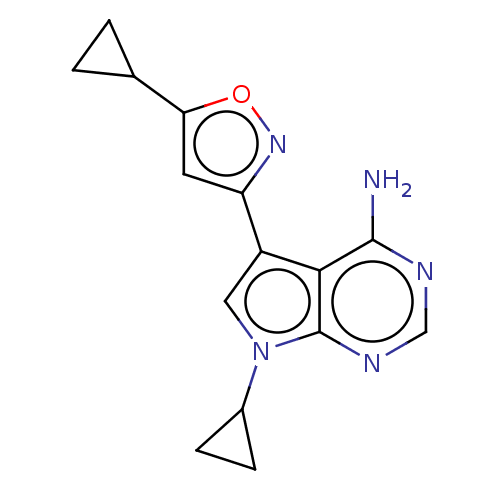
(US11603374, Example 20) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 8.60 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The potency of compounds inhibiting several different RET kinase forms (Wild Type, V804M, M918T, G810R, & G810S) were determined using CisBio's H... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2Z323KZ |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577069
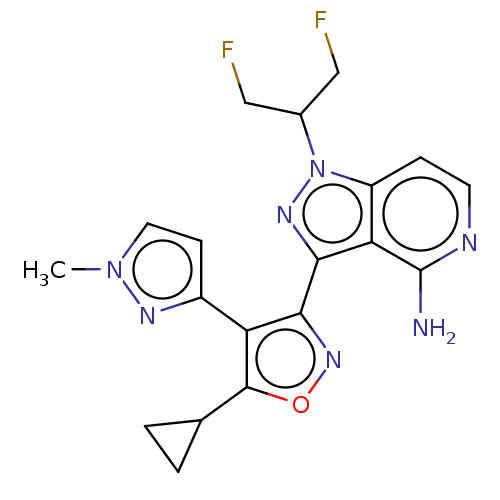
(US11472802, Example 73)Show SMILES Cn1ccc(n1)-c1c(noc1C1CC1)-c1nn(C(CF)CF)c2ccnc(N)c12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 9.10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM577043
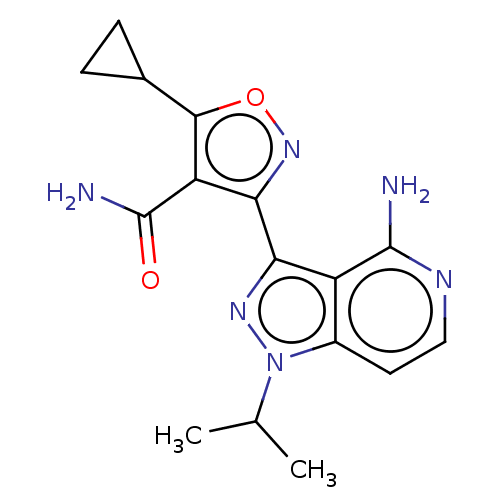
(3-(4-amino-1-isopropyl-1H- pyrazolo[4,3-c]pyridin-...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2C(N)=O)c2c(N)nccc12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 9.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data