 Found 3 hits Enzyme Inhibition Constant Data
Found 3 hits Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Glycogen phosphorylase, muscle form
(819/841 = 97%)†
(Homo sapiens (Human)) | BDBM34103
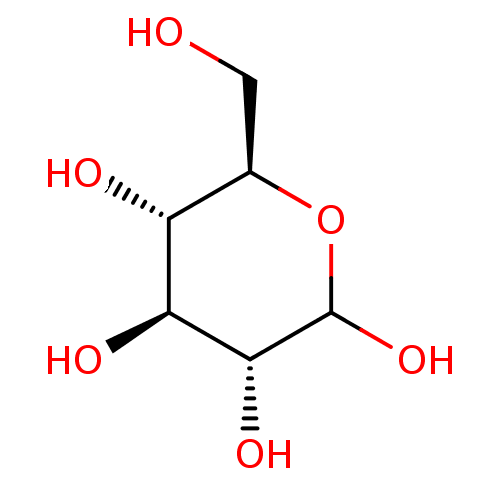
(D-glucose | dextrose | glucose)Show InChI InChI=1S/C6H12O6/c7-1-2-3(8)4(9)5(10)6(11)12-2/h2-11H,1H2/t2-,3-,4+,5-,6?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 2.77E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Alcala
Curated by ChEMBL
| Assay Description
Inhibitory activity against rabbit muscle glycogen phosphorylase |
J Med Chem 40: 4089-102 (1998)
Checked by Author
Article DOI: 10.1021/jm970273d
BindingDB Entry DOI: 10.7270/Q2S46TGM |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Glycogen phosphorylase, muscle form
(842/842 = 100%)†
(Oryctolagus cuniculus (rabbit)) | BDBM5655

(2-(2-chlorophenyl)-5,7-dihydroxy-8-[(3S,4R)-3-hydr...)Show SMILES CN1CC[C@@H]([C@H](O)C1)c1c(O)cc(O)c2c1oc(cc2=O)-c1ccccc1Cl |r| Show InChI InChI=1S/C21H20ClNO5/c1-23-7-6-12(17(27)10-23)19-14(24)8-15(25)20-16(26)9-18(28-21(19)20)11-4-2-3-5-13(11)22/h2-5,8-9,12,17,24-25,27H,6-7,10H2,1H3/t12-,17+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lyon
Curated by ChEMBL
| Assay Description
Binding affinity to rabbit muscular GPb by NMR binding assay |
J Med Chem 55: 1287-95 (2012)
Article DOI: 10.1021/jm201439b
BindingDB Entry DOI: 10.7270/Q21J9BS9 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Glycogen phosphorylase, muscle form
(819/841 = 97%)†
(Homo sapiens (Human)) | BDBM5655

(2-(2-chlorophenyl)-5,7-dihydroxy-8-[(3S,4R)-3-hydr...)Show SMILES CN1CC[C@@H]([C@H](O)C1)c1c(O)cc(O)c2c1oc(cc2=O)-c1ccccc1Cl |r| Show InChI InChI=1S/C21H20ClNO5/c1-23-7-6-12(17(27)10-23)19-14(24)8-15(25)20-16(26)9-18(28-21(19)20)11-4-2-3-5-13(11)22/h2-5,8-9,12,17,24-25,27H,6-7,10H2,1H3/t12-,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Novo Nordisk A/S
Curated by ChEMBL
| Assay Description
Inhibition of Rabbit muscle glycogen phosphorylase B. |
J Med Chem 47: 3537-45 (2004)
Article DOI: 10.1021/jm031121n
BindingDB Entry DOI: 10.7270/Q2HM5977 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data