

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50150998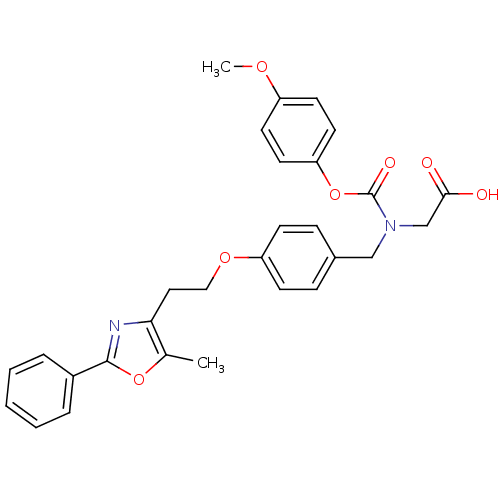 (((4-Methoxy-phenoxycarbonyl)-{4-[2-(5-methyl-2-phe...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 190 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Inhibition of human Peroxisome proliferator activated receptor gamma | J Med Chem 48: 2248-50 (2005) Article DOI: 10.1021/jm0496436 BindingDB Entry DOI: 10.7270/Q2SX6CRR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor alpha (Homo sapiens (Human)) | BDBM50150998 (((4-Methoxy-phenoxycarbonyl)-{4-[2-(5-methyl-2-phe...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 250 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Inhibition of human Peroxisome proliferator activated receptor alpha | J Med Chem 48: 2248-50 (2005) Article DOI: 10.1021/jm0496436 BindingDB Entry DOI: 10.7270/Q2SX6CRR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM28681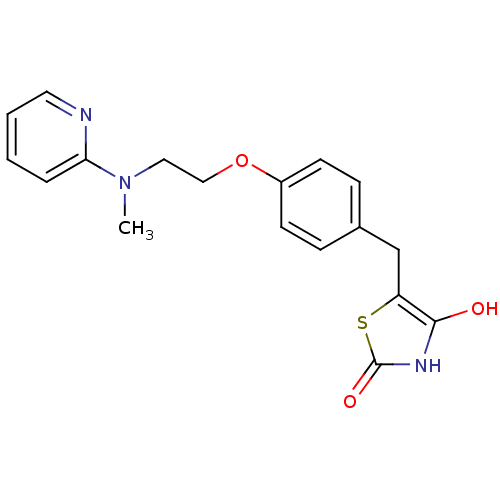 (5-[(4-{2-[methyl(pyridin-2-yl)amino]ethoxy}phenyl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 250 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Inhibition of human Peroxisome proliferator activated receptor gamma | J Med Chem 48: 2248-50 (2005) Article DOI: 10.1021/jm0496436 BindingDB Entry DOI: 10.7270/Q2SX6CRR | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM28680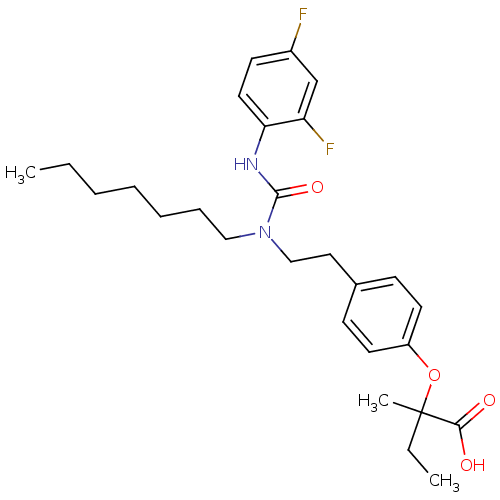 (2-[4-(2-{[(2,4-difluorophenyl)carbamoyl](heptyl)am...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 316 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Inhibition of human Peroxisome proliferator activated receptor gamma | J Med Chem 48: 2248-50 (2005) Article DOI: 10.1021/jm0496436 BindingDB Entry DOI: 10.7270/Q2SX6CRR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor alpha (Homo sapiens (Human)) | BDBM28680 (2-[4-(2-{[(2,4-difluorophenyl)carbamoyl](heptyl)am...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 538 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Inhibition of human Peroxisome proliferator activated receptor alpha | J Med Chem 48: 2248-50 (2005) Article DOI: 10.1021/jm0496436 BindingDB Entry DOI: 10.7270/Q2SX6CRR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor alpha (Homo sapiens (Human)) | BDBM50163728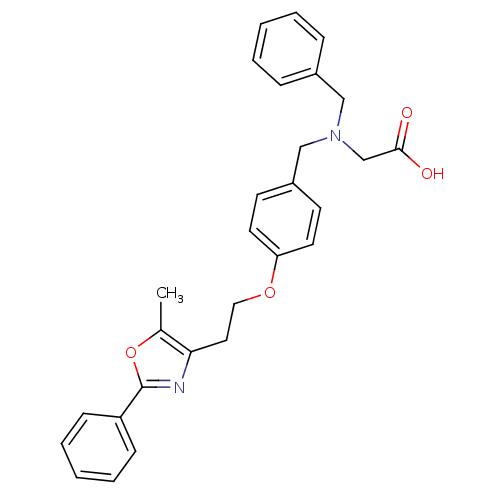 ((Benzyl-{4-[2-(5-methyl-2-phenyl-oxazol-4-yl)-etho...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Inhibition of human Peroxisome proliferator activated receptor alpha | J Med Chem 48: 2248-50 (2005) Article DOI: 10.1021/jm0496436 BindingDB Entry DOI: 10.7270/Q2SX6CRR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50163728 ((Benzyl-{4-[2-(5-methyl-2-phenyl-oxazol-4-yl)-etho...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 4.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Inhibition of human Peroxisome proliferator activated receptor gamma | J Med Chem 48: 2248-50 (2005) Article DOI: 10.1021/jm0496436 BindingDB Entry DOI: 10.7270/Q2SX6CRR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor alpha (Homo sapiens (Human)) | BDBM28700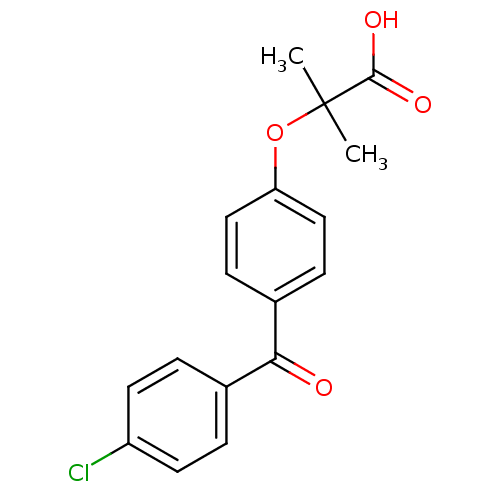 (2-(4-(4-Chlorobenzoyl)phenoxy)-2-methylpropionic a...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Inhibition of human Peroxisome proliferator activated receptor alpha | J Med Chem 48: 2248-50 (2005) Article DOI: 10.1021/jm0496436 BindingDB Entry DOI: 10.7270/Q2SX6CRR | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM28700 (2-(4-(4-Chlorobenzoyl)phenoxy)-2-methylpropionic a...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Inhibition of human Peroxisome proliferator activated receptor gamma | J Med Chem 48: 2248-50 (2005) Article DOI: 10.1021/jm0496436 BindingDB Entry DOI: 10.7270/Q2SX6CRR | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Peroxisome proliferator-activated receptor alpha (Homo sapiens (Human)) | BDBM28681 (5-[(4-{2-[methyl(pyridin-2-yl)amino]ethoxy}phenyl)...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Inhibition of human Peroxisome proliferator activated receptor alpha | J Med Chem 48: 2248-50 (2005) Article DOI: 10.1021/jm0496436 BindingDB Entry DOI: 10.7270/Q2SX6CRR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM28680 (2-[4-(2-{[(2,4-difluorophenyl)carbamoyl](heptyl)am...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 425 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Effective concentration against human Peroxisome proliferator activated receptor gamma | J Med Chem 48: 2248-50 (2005) Article DOI: 10.1021/jm0496436 BindingDB Entry DOI: 10.7270/Q2SX6CRR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor alpha (Homo sapiens (Human)) | BDBM28700 (2-(4-(4-Chlorobenzoyl)phenoxy)-2-methylpropionic a...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Effective concentration against human Peroxisome proliferator activated receptor alpha | J Med Chem 48: 2248-50 (2005) Article DOI: 10.1021/jm0496436 BindingDB Entry DOI: 10.7270/Q2SX6CRR | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50150998 (((4-Methoxy-phenoxycarbonyl)-{4-[2-(5-methyl-2-phe...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 110 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Effective concentration against human Peroxisome proliferator activated receptor gamma | J Med Chem 48: 2248-50 (2005) Article DOI: 10.1021/jm0496436 BindingDB Entry DOI: 10.7270/Q2SX6CRR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor alpha (Homo sapiens (Human)) | BDBM28681 (5-[(4-{2-[methyl(pyridin-2-yl)amino]ethoxy}phenyl)...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | >3.20E+4 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Effective concentration against human Peroxisome proliferator activated receptor alpha | J Med Chem 48: 2248-50 (2005) Article DOI: 10.1021/jm0496436 BindingDB Entry DOI: 10.7270/Q2SX6CRR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50163728 ((Benzyl-{4-[2-(5-methyl-2-phenyl-oxazol-4-yl)-etho...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Effective concentration against human Peroxisome proliferator activated receptor gamma | J Med Chem 48: 2248-50 (2005) Article DOI: 10.1021/jm0496436 BindingDB Entry DOI: 10.7270/Q2SX6CRR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor alpha (Homo sapiens (Human)) | BDBM50163728 ((Benzyl-{4-[2-(5-methyl-2-phenyl-oxazol-4-yl)-etho...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 20 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Effective concentration against human Peroxisome proliferator activated receptor alpha | J Med Chem 48: 2248-50 (2005) Article DOI: 10.1021/jm0496436 BindingDB Entry DOI: 10.7270/Q2SX6CRR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM28700 (2-(4-(4-Chlorobenzoyl)phenoxy)-2-methylpropionic a...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Effective concentration against human Peroxisome proliferator activated receptor gamma | J Med Chem 48: 2248-50 (2005) Article DOI: 10.1021/jm0496436 BindingDB Entry DOI: 10.7270/Q2SX6CRR | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM28681 (5-[(4-{2-[methyl(pyridin-2-yl)amino]ethoxy}phenyl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | n/a | n/a | 140 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Effective concentration against human Peroxisome proliferator activated receptor gamma | J Med Chem 48: 2248-50 (2005) Article DOI: 10.1021/jm0496436 BindingDB Entry DOI: 10.7270/Q2SX6CRR | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Peroxisome proliferator-activated receptor alpha (Homo sapiens (Human)) | BDBM50150998 (((4-Methoxy-phenoxycarbonyl)-{4-[2-(5-methyl-2-phe...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 320 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Effective concentration against human Peroxisome proliferator activated receptor alpha | J Med Chem 48: 2248-50 (2005) Article DOI: 10.1021/jm0496436 BindingDB Entry DOI: 10.7270/Q2SX6CRR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||