| Reaction Details |
|---|
 | Report a problem with these data |
| Target | C-C chemokine receptor type 5 |
|---|
| Ligand | BDBM314021 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Calcium Flux Inhibition Experiment |
|---|
| IC50 | 8.28±n/a nM |
|---|
| Citation |  Liu, H; Wu, B; Zheng, Y; Xie, X; Jiang, H; Peng, P; Luo, R; Li, J; Li, J; Zhu, Y; Chen, Y; Zhang, H; Yang, L; Zhou, Y; Chen, K 1-(3-aminopropyl) substituted cyclic amine compounds, preparation method therefor, and pharmaceutical compositions and uses thereof US Patent US10167299 Publication Date 1/1/2019 Liu, H; Wu, B; Zheng, Y; Xie, X; Jiang, H; Peng, P; Luo, R; Li, J; Li, J; Zhu, Y; Chen, Y; Zhang, H; Yang, L; Zhou, Y; Chen, K 1-(3-aminopropyl) substituted cyclic amine compounds, preparation method therefor, and pharmaceutical compositions and uses thereof US Patent US10167299 Publication Date 1/1/2019 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| C-C chemokine receptor type 5 |
|---|
| Name: | C-C chemokine receptor type 5 |
|---|
| Synonyms: | C-C CKR-5 | C-C chemokine receptor type 5 | CC-CKR-5 | CCR-5 | CCR5 | CCR5/mu opioid receptor complex | CCR5_HUMAN | CD_antigen=CD195 | CHEMR13 | CMKBR5 | HIV-1 fusion coreceptor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 40540.21 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P51681 |
|---|
| Residue: | 352 |
|---|
| Sequence: | MDYQVSSPIYDINYYTSEPCQKINVKQIAARLLPPLYSLVFIFGFVGNMLVILILINCKR
LKSMTDIYLLNLAISDLFFLLTVPFWAHYAAAQWDFGNTMCQLLTGLYFIGFFSGIFFII
LLTIDRYLAVVHAVFALKARTVTFGVVTSVITWVVAVFASLPGIIFTRSQKEGLHYTCSS
HFPYSQYQFWKNFQTLKIVILGLVLPLLVMVICYSGILKTLLRCRNEKKRHRAVRLIFTI
MIVYFLFWAPYNIVLLLNTFQEFFGLNNCSSSNRLDQAMQVTETLGMTHCCINPIIYAFV
GEKFRNYLLVFFQKHIAKRFCKCCSIFQQEAPERASSVYTRSTGEQEISVGL
|
|
|
|---|
| BDBM314021 |
|---|
| n/a |
|---|
| Name | BDBM314021 |
|---|
| Synonyms: | US10167299, Example 135 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H38FN5O2S |
|---|
| Mol. Mass. | 515.686 |
|---|
| SMILES | CC(C)C(=O)N1CCc2c(C1)nc(C)n2C1CC2CCC(C1)N2CC[C@H](NC(C)=O)c1ccc(F)s1 |r,TLB:23:22:15.21.16:18.19,14:15:22:18.19| |
|---|
| Structure | 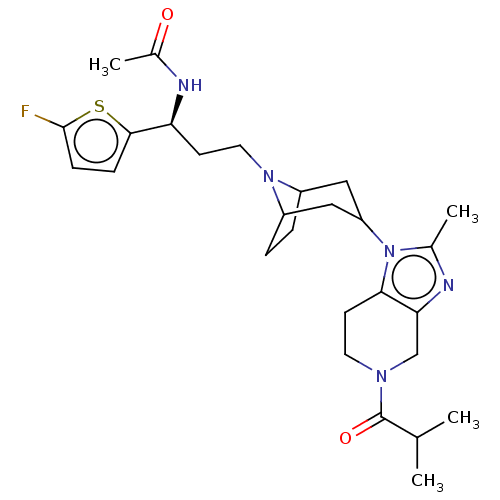
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Liu, H; Wu, B; Zheng, Y; Xie, X; Jiang, H; Peng, P; Luo, R; Li, J; Li, J; Zhu, Y; Chen, Y; Zhang, H; Yang, L; Zhou, Y; Chen, K 1-(3-aminopropyl) substituted cyclic amine compounds, preparation method therefor, and pharmaceutical compositions and uses thereof US Patent US10167299 Publication Date 1/1/2019
Liu, H; Wu, B; Zheng, Y; Xie, X; Jiang, H; Peng, P; Luo, R; Li, J; Li, J; Zhu, Y; Chen, Y; Zhang, H; Yang, L; Zhou, Y; Chen, K 1-(3-aminopropyl) substituted cyclic amine compounds, preparation method therefor, and pharmaceutical compositions and uses thereof US Patent US10167299 Publication Date 1/1/2019