| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 11B2, mitochondrial |
|---|
| Ligand | BDBM351257 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Inhibition Assay |
|---|
| EC50 | 0.005±n/a nM |
|---|
| Citation |  Aebi, J; Amrein, K; Hornsperger, B; Kuhn, B; Liu, Y; Maerki, HP; Martin, RE; Mayweg, AV; Tan, X; Wang, L; Li, D; Wu, J Dihydroquinoline pyrazolyl compounds US Patent US9796702 Publication Date 10/24/2017 Aebi, J; Amrein, K; Hornsperger, B; Kuhn, B; Liu, Y; Maerki, HP; Martin, RE; Mayweg, AV; Tan, X; Wang, L; Li, D; Wu, J Dihydroquinoline pyrazolyl compounds US Patent US9796702 Publication Date 10/24/2017 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 11B2, mitochondrial |
|---|
| Name: | Cytochrome P450 11B2, mitochondrial |
|---|
| Synonyms: | Aldosterone Synthase (CYP11B2) | Aldosterone synthase | Aldosterone-synthesizing enzyme | C11B2_HUMAN | CYP11B2 | CYPXIB2 | Cytochrome P450 11B2 | Cytochrome P450 11B2 (CYP11B2) | Cytochrome P450 11B2, mitochondrial | P-450Aldo | P-450C18 | Steroid 18-hydroxylase |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 57582.15 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P19099 |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALRAKAEVCVAAPWLSLQRARALGTRAARAPRTVLPFEAMPQHPGNRWLRLLQIWREQG
YEHLHLEMHQTFQELGPIFRYNLGGPRMVCVMLPEDVEKLQQVDSLHPCRMILEPWVAYR
QHRGHKCGVFLLNGPEWRFNRLRLNPDVLSPKAVQRFLPMVDAVARDFSQALKKKVLQNA
RGSLTLDVQPSIFHYTIEASNLALFGERLGLVGHSPSSASLNFLHALEVMFKSTVQLMFM
PRSLSRWISPKVWKEHFEAWDCIFQYGDNCIQKIYQELAFNRPQHYTGIVAELLLKAELS
LEAIKANSMELTAGSVDTTAFPLLMTLFELARNPDVQQILRQESLAAAASISEHPQKATT
ELPLLRAALKETLRLYPVGLFLERVVSSDLVLQNYHIPAGTLVQVFLYSLGRNAALFPRP
ERYNPQRWLDIRGSGRNFHHVPFGFGMRQCLGRRLAEAEMLLLLHHVLKHFLVETLTQED
IKMVYSFILRPGTSPLLTFRAIN
|
|
|
|---|
| BDBM351257 |
|---|
| n/a |
|---|
| Name | BDBM351257 |
|---|
| Synonyms: | 6-[4-[(1-Ethylsulfonyl-4-piperidyl)methyl]-1H-pyrazol-3-yl]-1-methyl-3,4-dihydroquinolin-2-one | US9796702, 18 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H28N4O3S |
|---|
| Mol. Mass. | 416.537 |
|---|
| SMILES | CCS(=O)(=O)N1CCC(Cc2c[nH]nc2-c2ccc3N(C)C(=O)CCc3c2)CC1 |
|---|
| Structure | 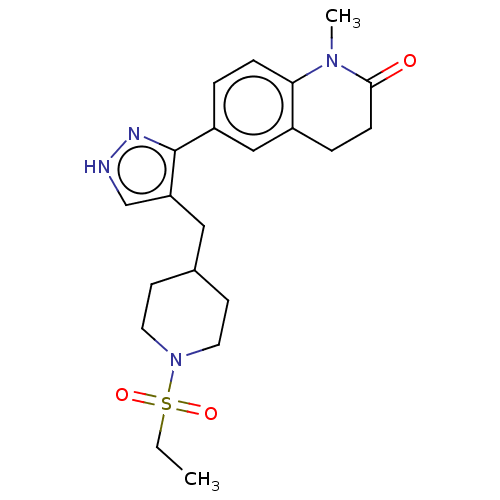
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Aebi, J; Amrein, K; Hornsperger, B; Kuhn, B; Liu, Y; Maerki, HP; Martin, RE; Mayweg, AV; Tan, X; Wang, L; Li, D; Wu, J Dihydroquinoline pyrazolyl compounds US Patent US9796702 Publication Date 10/24/2017
Aebi, J; Amrein, K; Hornsperger, B; Kuhn, B; Liu, Y; Maerki, HP; Martin, RE; Mayweg, AV; Tan, X; Wang, L; Li, D; Wu, J Dihydroquinoline pyrazolyl compounds US Patent US9796702 Publication Date 10/24/2017