| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Matrilysin |
|---|
| Ligand | BDBM23498 |
|---|
| Substrate/Competitor | BDBM23499 |
|---|
| Meas. Tech. | MMP Inhibition Assay |
|---|
| pH | 7.4±n/a |
|---|
| Temperature | 295.15±n/a K |
|---|
| IC50 | 26±n/a nM |
|---|
| Citation |  Zhang, Y; Xu, J; Levin, J; Hegen, M; Li, G; Robertshaw, H; Brennan, F; Cummons, T; Clarke, D; Vansell, N; Nickerson-Nutter, C; Barone, D; Mohler, K; Black, R; Skotnicki, J; Gibbons, J; Feldmann, M; Frost, P; Larsen, G; Lin, LL Identification and characterization of 4-[[4-(2-butynyloxy)phenyl]sulfonyl]-N-hydroxy-2,2-dimethyl-(3S)thiomorpholinecarboxamide (TMI-1), a novel dual tumor necrosis factor-alpha-converting enzyme/matrix metalloprotease inhibitor for the treatment of rheumatoid arthritis. J Pharmacol Exp Ther309:348-55 (2004) [PubMed] Article Zhang, Y; Xu, J; Levin, J; Hegen, M; Li, G; Robertshaw, H; Brennan, F; Cummons, T; Clarke, D; Vansell, N; Nickerson-Nutter, C; Barone, D; Mohler, K; Black, R; Skotnicki, J; Gibbons, J; Feldmann, M; Frost, P; Larsen, G; Lin, LL Identification and characterization of 4-[[4-(2-butynyloxy)phenyl]sulfonyl]-N-hydroxy-2,2-dimethyl-(3S)thiomorpholinecarboxamide (TMI-1), a novel dual tumor necrosis factor-alpha-converting enzyme/matrix metalloprotease inhibitor for the treatment of rheumatoid arthritis. J Pharmacol Exp Ther309:348-55 (2004) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Inhibition_Run data, Solution Info, Assay Method |
|---|
| |
| Matrilysin |
|---|
| Name: | Matrilysin |
|---|
| Synonyms: | MMP7 | MMP7_HUMAN | MPSL1 | Matrix metalloproteinase 7 | Matrix metalloproteinase-7 (MMP-7) | Matrix metalloproteinase-7 (MMP7) | PUMP1 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 29681.54 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P09237 |
|---|
| Residue: | 267 |
|---|
| Sequence: | MRLTVLCAVCLLPGSLALPLPQEAGGMSELQWEQAQDYLKRFYLYDSETKNANSLEAKLK
EMQKFFGLPITGMLNSRVIEIMQKPRCGVPDVAEYSLFPNSPKWTSKVVTYRIVSYTRDL
PHITVDRLVSKALNMWGKEIPLHFRKVVWGTADIMIGFARGAHGDSYPFDGPGNTLAHAF
APGTGLGGDAHFDEDERWTDGSSLGINFLYAATHELGHSLGMGHSSDPNAVMYPTYGNGD
PQNFKLSQDDIKGIQKLYGKRSNSRKK
|
|
|
|---|
| BDBM23498 |
|---|
| BDBM23499 |
|---|
| Name | BDBM23498 |
|---|
| Synonyms: | (3S)-4-{[4-(but-2-yn-1-yloxy)benzene]sulfonyl}-N-hydroxy-2,2-dimethylthiomorpholine-3-carboxamide | CHEMBL207729 | TMI-1 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C17H22N2O5S2 |
|---|
| Mol. Mass. | 398.497 |
|---|
| SMILES | CC#CCOc1ccc(cc1)S(=O)(=O)N1CCSC(C)(C)[C@@H]1C(=O)NO |r| |
|---|
| Structure | 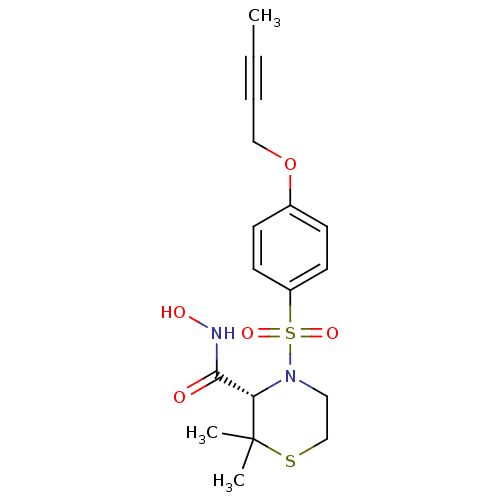
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zhang, Y; Xu, J; Levin, J; Hegen, M; Li, G; Robertshaw, H; Brennan, F; Cummons, T; Clarke, D; Vansell, N; Nickerson-Nutter, C; Barone, D; Mohler, K; Black, R; Skotnicki, J; Gibbons, J; Feldmann, M; Frost, P; Larsen, G; Lin, LL Identification and characterization of 4-[[4-(2-butynyloxy)phenyl]sulfonyl]-N-hydroxy-2,2-dimethyl-(3S)thiomorpholinecarboxamide (TMI-1), a novel dual tumor necrosis factor-alpha-converting enzyme/matrix metalloprotease inhibitor for the treatment of rheumatoid arthritis. J Pharmacol Exp Ther309:348-55 (2004) [PubMed] Article
Zhang, Y; Xu, J; Levin, J; Hegen, M; Li, G; Robertshaw, H; Brennan, F; Cummons, T; Clarke, D; Vansell, N; Nickerson-Nutter, C; Barone, D; Mohler, K; Black, R; Skotnicki, J; Gibbons, J; Feldmann, M; Frost, P; Larsen, G; Lin, LL Identification and characterization of 4-[[4-(2-butynyloxy)phenyl]sulfonyl]-N-hydroxy-2,2-dimethyl-(3S)thiomorpholinecarboxamide (TMI-1), a novel dual tumor necrosis factor-alpha-converting enzyme/matrix metalloprotease inhibitor for the treatment of rheumatoid arthritis. J Pharmacol Exp Ther309:348-55 (2004) [PubMed] Article