 Found 1752 hits with Last Name = 'skotnicki' and Initial = 'j'
Found 1752 hits with Last Name = 'skotnicki' and Initial = 'j' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50070256
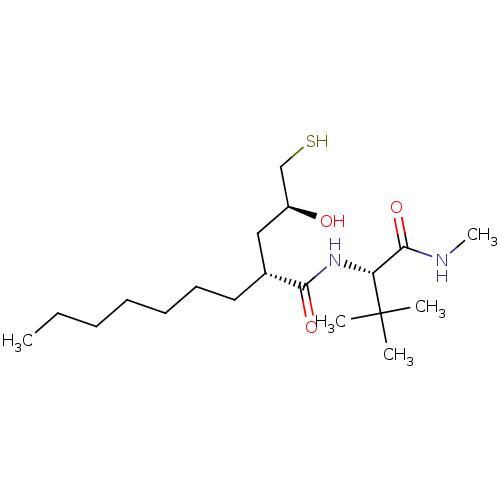
((R)-2-((S)-2-Hydroxy-3-mercapto-propyl)-nonanoic a...)Show SMILES CCCCCCC[C@H](C[C@H](O)CS)C(=O)N[C@H](C(=O)NC)C(C)(C)C Show InChI InChI=1S/C19H38N2O3S/c1-6-7-8-9-10-11-14(12-15(22)13-25)17(23)21-16(18(24)20-5)19(2,3)4/h14-16,22,25H,6-13H2,1-5H3,(H,20,24)(H,21,23)/t14-,15+,16-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 0.140 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth-Ayerst Research
Curated by ChEMBL
| Assay Description
Inhibition of matrix metalloproteinase-9, gelatinase-B |
Bioorg Med Chem Lett 8: 1163-8 (1999)
BindingDB Entry DOI: 10.7270/Q2348JH9 |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50070257
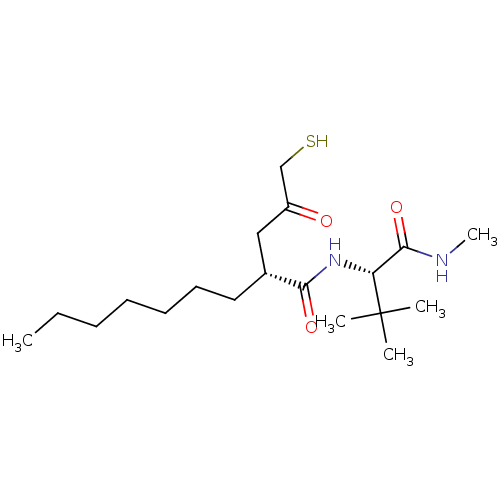
((R)-2-(3-Mercapto-2-oxo-propyl)-nonanoic acid ((S)...)Show SMILES CCCCCCC[C@H](CC(=O)CS)C(=O)N[C@H](C(=O)NC)C(C)(C)C Show InChI InChI=1S/C19H36N2O3S/c1-6-7-8-9-10-11-14(12-15(22)13-25)17(23)21-16(18(24)20-5)19(2,3)4/h14,16,25H,6-13H2,1-5H3,(H,20,24)(H,21,23)/t14-,16-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 0.140 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth-Ayerst Research
Curated by ChEMBL
| Assay Description
Inhibition of matrix metalloproteinase-9, gelatinase-B |
Bioorg Med Chem Lett 8: 1163-8 (1999)
BindingDB Entry DOI: 10.7270/Q2348JH9 |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50070228

(2-(2-Mercapto-acetyl)-nonanoic acid ((S)-2,2-dimet...)Show SMILES CCCCCCCC(C(=O)CS)C(=O)N[C@H](C(=O)NC)C(C)(C)C Show InChI InChI=1S/C18H34N2O3S/c1-6-7-8-9-10-11-13(14(21)12-24)16(22)20-15(17(23)19-5)18(2,3)4/h13,15,24H,6-12H2,1-5H3,(H,19,23)(H,20,22)/t13?,15-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Affymax Research Institute
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against gelatinase-B(MMP-9). |
Bioorg Med Chem Lett 8: 1157-62 (1999)
BindingDB Entry DOI: 10.7270/Q26W997Q |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128610

(2-(4-Methoxy-benzenesulfonyl)-4-methylene-2-(2-met...)Show SMILES [#6]-[#8]-c1ccc(cc1)S(=O)(=O)C([#6]\[#6]=[#6](\[#6])-[#6])([#6]\[#6]=[#6](/[#6])-[#6])[#6](=O)-[#7]-[#8] Show InChI InChI=1S/C19H27NO5S/c1-14(2)10-12-19(18(21)20-22,13-11-15(3)4)26(23,24)17-8-6-16(25-5)7-9-17/h6-11,22H,12-13H2,1-5H3,(H,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibition of matrix metalloprotease-13 |
J Med Chem 46: 2361-75 (2003)
Article DOI: 10.1021/jm0205548
BindingDB Entry DOI: 10.7270/Q2RR1ZZJ |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50173235

((S)-2-{4'-[(5-Bromo-4-methoxy-benzofuran-2-carbony...)Show SMILES COc1c(Br)ccc2oc(cc12)C(=O)Nc1ccc(cc1)-c1ccc(cc1)S(=O)(=O)N[C@@H](C(C)C)C(O)=O Show InChI InChI=1S/C27H25BrN2O7S/c1-15(2)24(27(32)33)30-38(34,35)19-10-6-17(7-11-19)16-4-8-18(9-5-16)29-26(31)23-14-20-22(37-23)13-12-21(28)25(20)36-3/h4-15,24,30H,1-3H3,(H,29,31)(H,32,33)/t24-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibitory concentration against MMP-13 |
Bioorg Med Chem Lett 15: 4961-6 (2005)
Article DOI: 10.1016/j.bmcl.2005.08.001
BindingDB Entry DOI: 10.7270/Q20Z72TH |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50118985
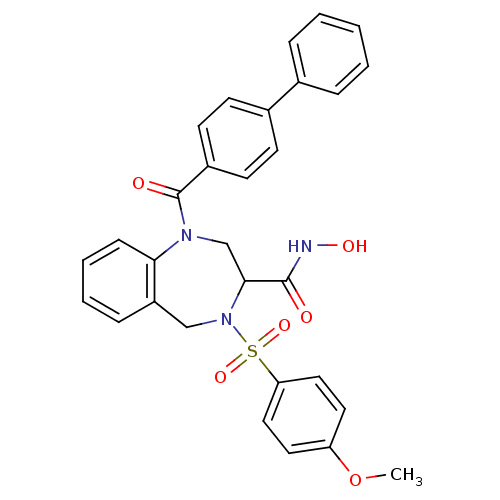
(1-(Biphenyl-4-carbonyl)-4-(4-methoxy-benzenesulfon...)Show SMILES COc1ccc(cc1)S(=O)(=O)N1Cc2ccccc2N(CC1C(=O)NO)C(=O)c1ccc(cc1)-c1ccccc1 Show InChI InChI=1S/C30H27N3O6S/c1-39-25-15-17-26(18-16-25)40(37,38)33-19-24-9-5-6-10-27(24)32(20-28(33)29(34)31-36)30(35)23-13-11-22(12-14-23)21-7-3-2-4-8-21/h2-18,28,36H,19-20H2,1H3,(H,31,34) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Concentration of the compound required in vitro to inhibit Matrix metalloproteinase-13 |
Bioorg Med Chem Lett 12: 2867-70 (2002)
BindingDB Entry DOI: 10.7270/Q2JS9PSH |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50118983
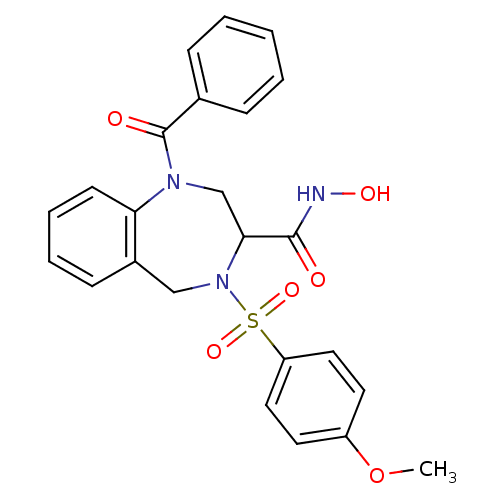
(1-Benzoyl-4-(4-methoxy-benzenesulfonyl)-2,3,4,5-te...)Show SMILES COc1ccc(cc1)S(=O)(=O)N1Cc2ccccc2N(CC1C(=O)NO)C(=O)c1ccccc1 Show InChI InChI=1S/C24H23N3O6S/c1-33-19-11-13-20(14-12-19)34(31,32)27-15-18-9-5-6-10-21(18)26(16-22(27)23(28)25-30)24(29)17-7-3-2-4-8-17/h2-14,22,30H,15-16H2,1H3,(H,25,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Concentration of the compound required in vitro to inhibit Matrix metalloproteinase-13 |
Bioorg Med Chem Lett 12: 2867-70 (2002)
BindingDB Entry DOI: 10.7270/Q2JS9PSH |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Bos taurus) | BDBM50170292
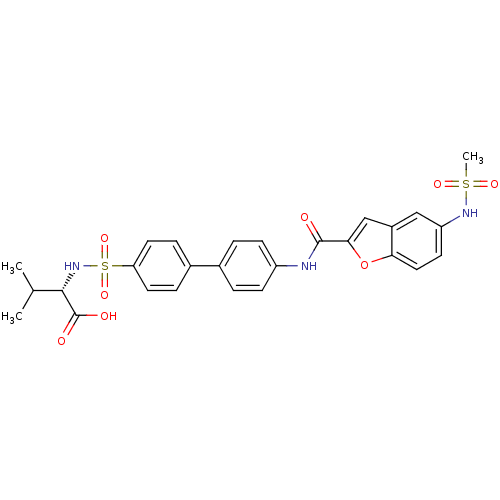
((S)-2-{4'-[(5-Methanesulfonylamino-benzofuran-2-ca...)Show SMILES CC(C)[C@H](NS(=O)(=O)c1ccc(cc1)-c1ccc(NC(=O)c2cc3cc(NS(C)(=O)=O)ccc3o2)cc1)C(O)=O Show InChI InChI=1S/C27H27N3O8S2/c1-16(2)25(27(32)33)30-40(36,37)22-11-6-18(7-12-22)17-4-8-20(9-5-17)28-26(31)24-15-19-14-21(29-39(3,34)35)10-13-23(19)38-24/h4-16,25,29-30H,1-3H3,(H,28,31)(H,32,33)/t25-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.430 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibitory concentration against MMP-13 of bovine articular cartilage explants |
Bioorg Med Chem Lett 15: 4105-9 (2005)
Article DOI: 10.1016/j.bmcl.2005.06.019
BindingDB Entry DOI: 10.7270/Q2TX3DXK |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50128610

(2-(4-Methoxy-benzenesulfonyl)-4-methylene-2-(2-met...)Show SMILES [#6]-[#8]-c1ccc(cc1)S(=O)(=O)C([#6]\[#6]=[#6](\[#6])-[#6])([#6]\[#6]=[#6](/[#6])-[#6])[#6](=O)-[#7]-[#8] Show InChI InChI=1S/C19H27NO5S/c1-14(2)10-12-19(18(21)20-22,13-11-15(3)4)26(23,24)17-8-6-16(25-5)7-9-17/h6-11,22H,12-13H2,1-5H3,(H,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibition of matrix metalloprotease-9 |
J Med Chem 46: 2361-75 (2003)
Article DOI: 10.1021/jm0205548
BindingDB Entry DOI: 10.7270/Q2RR1ZZJ |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50106133
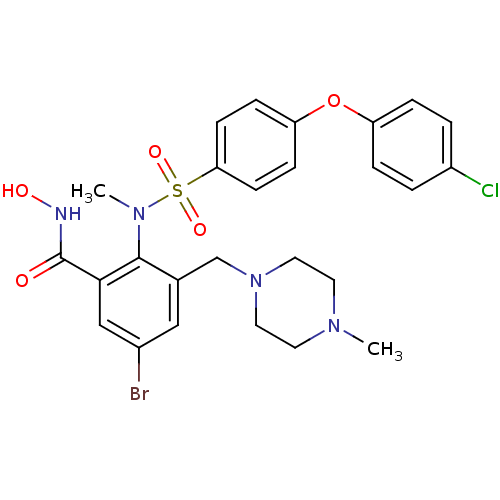
(5-Bromo-2-{[4-(4-chloro-phenoxy)-benzenesulfonyl]-...)Show SMILES CN(c1c(CN2CCN(C)CC2)cc(Br)cc1C(=O)NO)S(=O)(=O)c1ccc(Oc2ccc(Cl)cc2)cc1 Show InChI InChI=1S/C26H28BrClN4O5S/c1-30-11-13-32(14-12-30)17-18-15-19(27)16-24(26(33)29-34)25(18)31(2)38(35,36)23-9-7-22(8-10-23)37-21-5-3-20(28)4-6-21/h3-10,15-16,34H,11-14,17H2,1-2H3,(H,29,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth-Ayerst Research
Curated by ChEMBL
| Assay Description
In vitro inhibition of Matrix metalloprotease-9. |
Bioorg Med Chem Lett 11: 2975-8 (2001)
BindingDB Entry DOI: 10.7270/Q2125T64 |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Bos taurus) | BDBM50170302
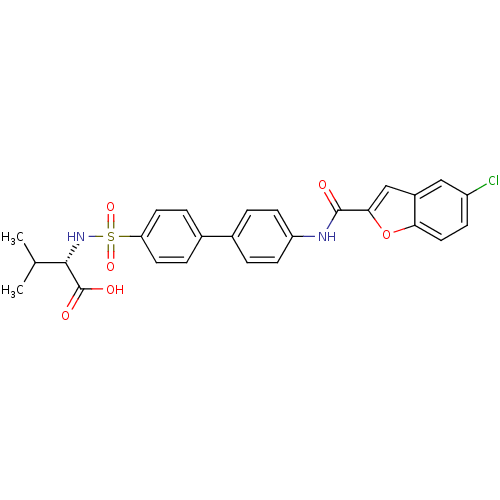
((S)-2-{4'-[(5-Chloro-benzofuran-2-carbonyl)-amino]...)Show SMILES CC(C)[C@H](NS(=O)(=O)c1ccc(cc1)-c1ccc(NC(=O)c2cc3cc(Cl)ccc3o2)cc1)C(O)=O Show InChI InChI=1S/C26H23ClN2O6S/c1-15(2)24(26(31)32)29-36(33,34)21-10-5-17(6-11-21)16-3-8-20(9-4-16)28-25(30)23-14-18-13-19(27)7-12-22(18)35-23/h3-15,24,29H,1-2H3,(H,28,30)(H,31,32)/t24-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibitory concentration against MMP-13 of bovine articular cartilage explants |
Bioorg Med Chem Lett 15: 4105-9 (2005)
Article DOI: 10.1016/j.bmcl.2005.06.019
BindingDB Entry DOI: 10.7270/Q2TX3DXK |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50118983

(1-Benzoyl-4-(4-methoxy-benzenesulfonyl)-2,3,4,5-te...)Show SMILES COc1ccc(cc1)S(=O)(=O)N1Cc2ccccc2N(CC1C(=O)NO)C(=O)c1ccccc1 Show InChI InChI=1S/C24H23N3O6S/c1-33-19-11-13-20(14-12-19)34(31,32)27-15-18-9-5-6-10-21(18)26(16-22(27)23(28)25-30)24(29)17-7-3-2-4-8-17/h2-14,22,30H,15-16H2,1H3,(H,25,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Concentration required in vitro to inhibit Matrix metalloproteinase-9 |
Bioorg Med Chem Lett 12: 2867-70 (2002)
BindingDB Entry DOI: 10.7270/Q2JS9PSH |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50173236

((S)-2-{4'-[(5-Bromo-benzofuran-2-carbonyl)-amino]-...)Show SMILES CC(C)[C@H](NS(=O)(=O)c1ccc(cc1)-c1ccc(NC(=O)c2cc3cc(Br)ccc3o2)cc1)C(O)=O Show InChI InChI=1S/C26H23BrN2O6S/c1-15(2)24(26(31)32)29-36(33,34)21-10-5-17(6-11-21)16-3-8-20(9-4-16)28-25(30)23-14-18-13-19(27)7-12-22(18)35-23/h3-15,24,29H,1-2H3,(H,28,30)(H,31,32)/t24-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.610 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibitory concentration against MMP-13 |
Bioorg Med Chem Lett 15: 4961-6 (2005)
Article DOI: 10.1016/j.bmcl.2005.08.001
BindingDB Entry DOI: 10.7270/Q20Z72TH |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Bos taurus) | BDBM50170287
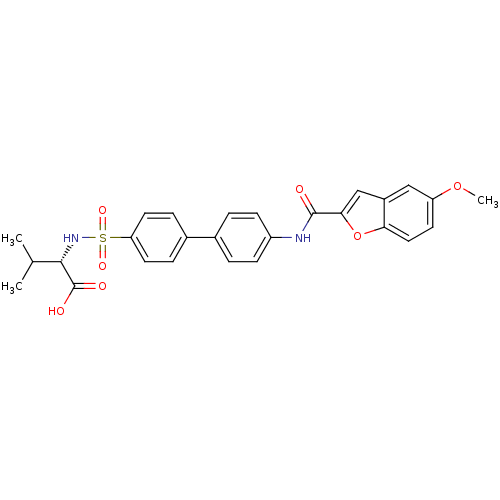
((S)-2-{4'-[(5-Methoxy-benzofuran-2-carbonyl)-amino...)Show SMILES COc1ccc2oc(cc2c1)C(=O)Nc1ccc(cc1)-c1ccc(cc1)S(=O)(=O)N[C@@H](C(C)C)C(O)=O Show InChI InChI=1S/C27H26N2O7S/c1-16(2)25(27(31)32)29-37(33,34)22-11-6-18(7-12-22)17-4-8-20(9-5-17)28-26(30)24-15-19-14-21(35-3)10-13-23(19)36-24/h4-16,25,29H,1-3H3,(H,28,30)(H,31,32)/t25-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.660 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibitory concentration against MMP-13 of bovine articular cartilage explants |
Bioorg Med Chem Lett 15: 4105-9 (2005)
Article DOI: 10.1016/j.bmcl.2005.06.019
BindingDB Entry DOI: 10.7270/Q2TX3DXK |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50118985

(1-(Biphenyl-4-carbonyl)-4-(4-methoxy-benzenesulfon...)Show SMILES COc1ccc(cc1)S(=O)(=O)N1Cc2ccccc2N(CC1C(=O)NO)C(=O)c1ccc(cc1)-c1ccccc1 Show InChI InChI=1S/C30H27N3O6S/c1-39-25-15-17-26(18-16-25)40(37,38)33-19-24-9-5-6-10-27(24)32(20-28(33)29(34)31-36)30(35)23-13-11-22(12-14-23)21-7-3-2-4-8-21/h2-18,28,36H,19-20H2,1H3,(H,31,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Concentration required in vitro to inhibit Matrix metalloproteinase-9 |
Bioorg Med Chem Lett 12: 2867-70 (2002)
BindingDB Entry DOI: 10.7270/Q2JS9PSH |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50106133

(5-Bromo-2-{[4-(4-chloro-phenoxy)-benzenesulfonyl]-...)Show SMILES CN(c1c(CN2CCN(C)CC2)cc(Br)cc1C(=O)NO)S(=O)(=O)c1ccc(Oc2ccc(Cl)cc2)cc1 Show InChI InChI=1S/C26H28BrClN4O5S/c1-30-11-13-32(14-12-30)17-18-15-19(27)16-24(26(33)29-34)25(18)31(2)38(35,36)23-9-7-22(8-10-23)37-21-5-3-20(28)4-6-21/h3-10,15-16,34H,11-14,17H2,1-2H3,(H,29,33) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth-Ayerst Research
Curated by ChEMBL
| Assay Description
Inhibition of Matrix metalloprotease-13 |
Bioorg Med Chem Lett 11: 2975-8 (2001)
BindingDB Entry DOI: 10.7270/Q2125T64 |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Bos taurus) | BDBM50170300
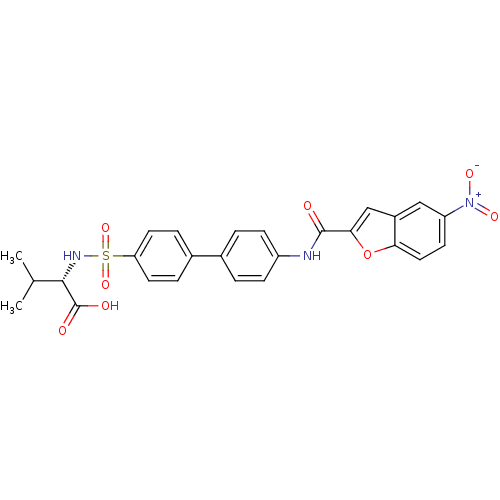
((S)-3-Methyl-2-{4'-[(5-nitro-benzofuran-2-carbonyl...)Show SMILES CC(C)[C@H](NS(=O)(=O)c1ccc(cc1)-c1ccc(NC(=O)c2cc3cc(ccc3o2)[N+]([O-])=O)cc1)C(O)=O Show InChI InChI=1S/C26H23N3O8S/c1-15(2)24(26(31)32)28-38(35,36)21-10-5-17(6-11-21)16-3-7-19(8-4-16)27-25(30)23-14-18-13-20(29(33)34)9-12-22(18)37-23/h3-15,24,28H,1-2H3,(H,27,30)(H,31,32)/t24-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibitory concentration against MMP-13 of bovine articular cartilage explants |
Bioorg Med Chem Lett 15: 4105-9 (2005)
Article DOI: 10.1016/j.bmcl.2005.06.019
BindingDB Entry DOI: 10.7270/Q2TX3DXK |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50106136
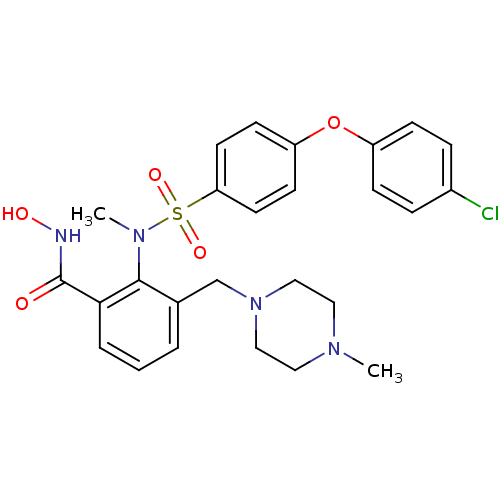
(2-{[4-(4-Chloro-phenoxy)-benzenesulfonyl]-methyl-a...)Show SMILES CN(c1c(CN2CCN(C)CC2)cccc1C(=O)NO)S(=O)(=O)c1ccc(Oc2ccc(Cl)cc2)cc1 Show InChI InChI=1S/C26H29ClN4O5S/c1-29-14-16-31(17-15-29)18-19-4-3-5-24(26(32)28-33)25(19)30(2)37(34,35)23-12-10-22(11-13-23)36-21-8-6-20(27)7-9-21/h3-13,33H,14-18H2,1-2H3,(H,28,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth-Ayerst Research
Curated by ChEMBL
| Assay Description
In vitro inhibition of Matrix metalloprotease-13. |
Bioorg Med Chem Lett 11: 2975-8 (2001)
BindingDB Entry DOI: 10.7270/Q2125T64 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50173236

((S)-2-{4'-[(5-Bromo-benzofuran-2-carbonyl)-amino]-...)Show SMILES CC(C)[C@H](NS(=O)(=O)c1ccc(cc1)-c1ccc(NC(=O)c2cc3cc(Br)ccc3o2)cc1)C(O)=O Show InChI InChI=1S/C26H23BrN2O6S/c1-15(2)24(26(31)32)29-36(33,34)21-10-5-17(6-11-21)16-3-8-20(9-4-16)28-25(30)23-14-18-13-19(27)7-12-22(18)35-23/h3-15,24,29H,1-2H3,(H,28,30)(H,31,32)/t24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.910 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibitory concentration against MMP-2 |
Bioorg Med Chem Lett 15: 4961-6 (2005)
Article DOI: 10.1016/j.bmcl.2005.08.001
BindingDB Entry DOI: 10.7270/Q20Z72TH |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Bos taurus) | BDBM50170294
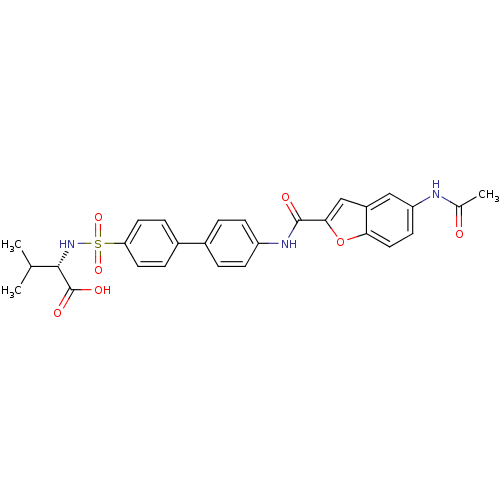
((S)-2-{4'-[(5-Acetylamino-benzofuran-2-carbonyl)-a...)Show SMILES CC(C)[C@H](NS(=O)(=O)c1ccc(cc1)-c1ccc(NC(=O)c2cc3cc(NC(C)=O)ccc3o2)cc1)C(O)=O Show InChI InChI=1S/C28H27N3O7S/c1-16(2)26(28(34)35)31-39(36,37)23-11-6-19(7-12-23)18-4-8-21(9-5-18)30-27(33)25-15-20-14-22(29-17(3)32)10-13-24(20)38-25/h4-16,26,31H,1-3H3,(H,29,32)(H,30,33)(H,34,35)/t26-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.980 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibitory concentration against MMP-13 of bovine articular cartilage explants |
Bioorg Med Chem Lett 15: 4105-9 (2005)
Article DOI: 10.1016/j.bmcl.2005.06.019
BindingDB Entry DOI: 10.7270/Q2TX3DXK |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50126612
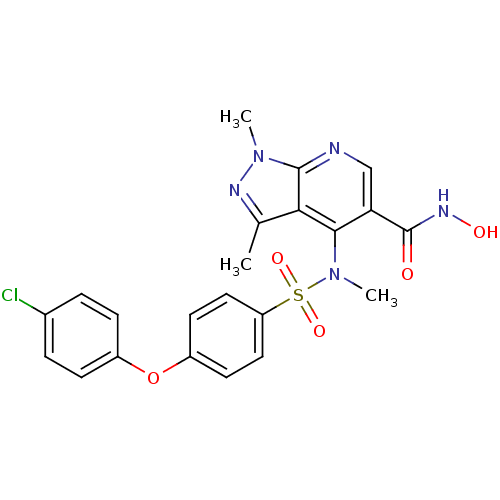
(4-{[4-(4-Chloro-phenoxy)-benzenesulfonyl]-methyl-a...)Show SMILES CN(c1c(cnc2n(C)nc(C)c12)C(=O)NO)S(=O)(=O)c1ccc(Oc2ccc(Cl)cc2)cc1 Show InChI InChI=1S/C22H20ClN5O5S/c1-13-19-20(18(22(29)26-30)12-24-21(19)27(2)25-13)28(3)34(31,32)17-10-8-16(9-11-17)33-15-6-4-14(23)5-7-15/h4-12,30H,1-3H3,(H,26,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth-Ayerst Research
Curated by ChEMBL
| Assay Description
Inhibitory activity against matrix metalloproteinase 13 (MMP-13). |
Bioorg Med Chem Lett 13: 1487-90 (2003)
BindingDB Entry DOI: 10.7270/Q29W0DW0 |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50126626
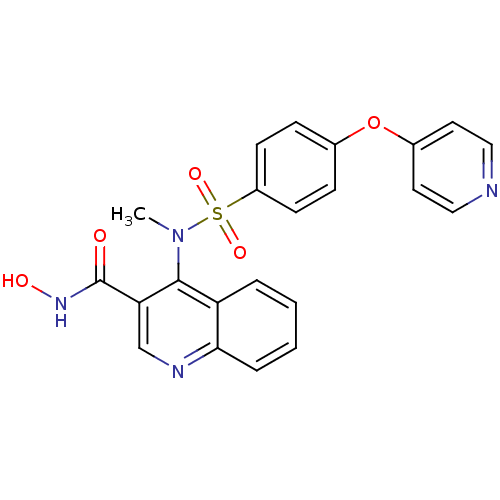
(4-{Methyl-[4-(pyridin-4-yloxy)-benzenesulfonyl]-am...)Show SMILES CN(c1c(cnc2ccccc12)C(=O)NO)S(=O)(=O)c1ccc(Oc2ccncc2)cc1 Show InChI InChI=1S/C22H18N4O5S/c1-26(21-18-4-2-3-5-20(18)24-14-19(21)22(27)25-28)32(29,30)17-8-6-15(7-9-17)31-16-10-12-23-13-11-16/h2-14,28H,1H3,(H,25,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth-Ayerst Research
Curated by ChEMBL
| Assay Description
Inhibitory activity against matrix metalloproteinase 13 (MMP-13). |
Bioorg Med Chem Lett 13: 1487-90 (2003)
BindingDB Entry DOI: 10.7270/Q29W0DW0 |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128668
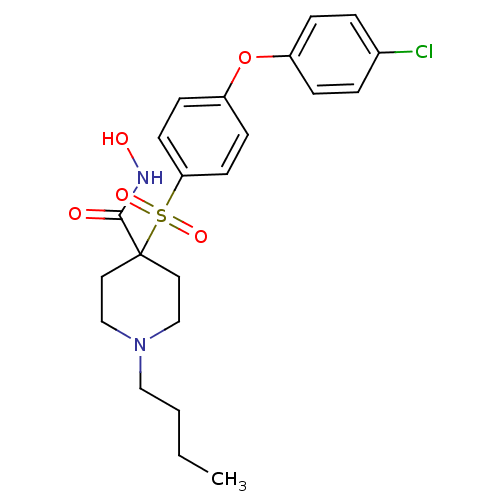
(1-Butyl-4-[4-(4-chloro-phenoxy)-benzenesulfonyl]-p...)Show SMILES CCCCN1CCC(CC1)(C(=O)NO)S(=O)(=O)c1ccc(Oc2ccc(Cl)cc2)cc1 Show InChI InChI=1S/C22H27ClN2O5S/c1-2-3-14-25-15-12-22(13-16-25,21(26)24-27)31(28,29)20-10-8-19(9-11-20)30-18-6-4-17(23)5-7-18/h4-11,27H,2-3,12-16H2,1H3,(H,24,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128631
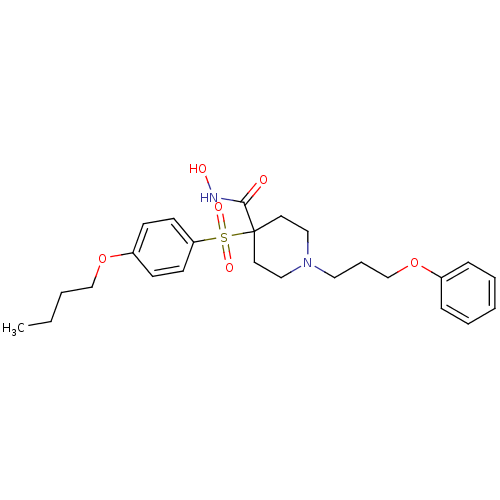
(4-(4-Butoxy-benzenesulfonyl)-1-(3-phenoxy-propyl)-...)Show SMILES CCCCOc1ccc(cc1)S(=O)(=O)C1(CCN(CCCOc2ccccc2)CC1)C(=O)NO Show InChI InChI=1S/C25H34N2O6S/c1-2-3-19-32-22-10-12-23(13-11-22)34(30,31)25(24(28)26-29)14-17-27(18-15-25)16-7-20-33-21-8-5-4-6-9-21/h4-6,8-13,29H,2-3,7,14-20H2,1H3,(H,26,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128645
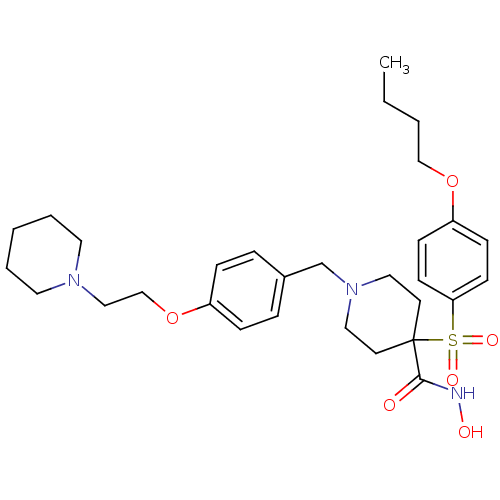
(1-(4-(2-(piperidin-1-yl)ethoxy)benzyl)-4-(4-butoxy...)Show SMILES CCCCOc1ccc(cc1)S(=O)(=O)C1(CCN(Cc2ccc(OCCN3CCCCC3)cc2)CC1)C(=O)NO Show InChI InChI=1S/C30H43N3O6S/c1-2-3-22-38-27-11-13-28(14-12-27)40(36,37)30(29(34)31-35)15-19-33(20-16-30)24-25-7-9-26(10-8-25)39-23-21-32-17-5-4-6-18-32/h7-14,35H,2-6,15-24H2,1H3,(H,31,34) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128643
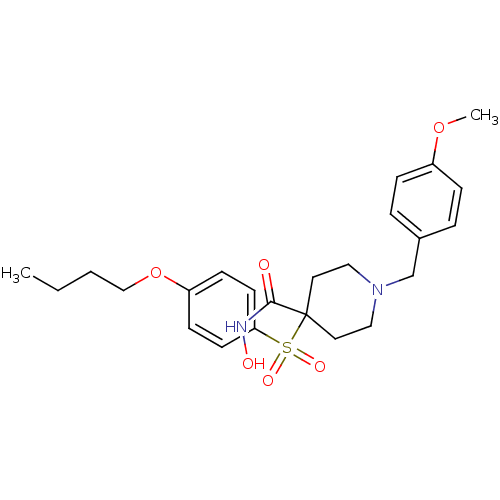
(1-(4-methoxybenzyl)-4-(4-butoxyphenylsulfonyl)-N-h...)Show SMILES CCCCOc1ccc(cc1)S(=O)(=O)C1(CCN(Cc2ccc(OC)cc2)CC1)C(=O)NO Show InChI InChI=1S/C24H32N2O6S/c1-3-4-17-32-21-9-11-22(12-10-21)33(29,30)24(23(27)25-28)13-15-26(16-14-24)18-19-5-7-20(31-2)8-6-19/h5-12,28H,3-4,13-18H2,1-2H3,(H,25,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50128649
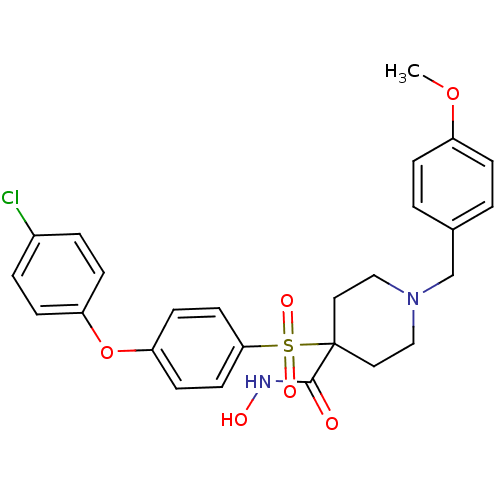
(1-(4-methoxybenzyl)-4-(4-(4-chlorophenoxy)phenylsu...)Show SMILES COc1ccc(CN2CCC(CC2)(C(=O)NO)S(=O)(=O)c2ccc(Oc3ccc(Cl)cc3)cc2)cc1 Show InChI InChI=1S/C26H27ClN2O6S/c1-34-21-6-2-19(3-7-21)18-29-16-14-26(15-17-29,25(30)28-31)36(32,33)24-12-10-23(11-13-24)35-22-8-4-20(27)5-9-22/h2-13,31H,14-18H2,1H3,(H,28,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-9. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128651
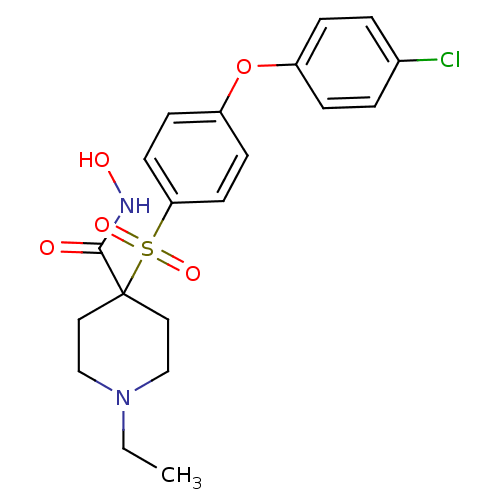
(4-(4-(4-chlorophenoxy)phenylsulfonyl)-1-ethyl-N-hy...)Show SMILES CCN1CCC(CC1)(C(=O)NO)S(=O)(=O)c1ccc(Oc2ccc(Cl)cc2)cc1 Show InChI InChI=1S/C20H23ClN2O5S/c1-2-23-13-11-20(12-14-23,19(24)22-25)29(26,27)18-9-7-17(8-10-18)28-16-5-3-15(21)4-6-16/h3-10,25H,2,11-14H2,1H3,(H,22,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128597
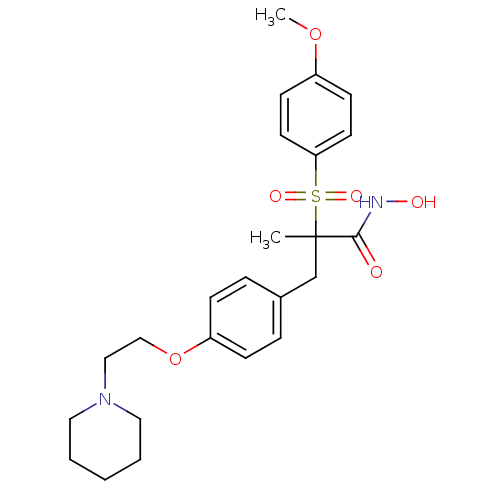
((R)-N-hydroxy-2-(4-methoxyphenylsulfonyl)-2-methyl...)Show SMILES COc1ccc(cc1)S(=O)(=O)C(C)(Cc1ccc(OCCN2CCCCC2)cc1)C(=O)NO Show InChI InChI=1S/C24H32N2O6S/c1-24(23(27)25-28,33(29,30)22-12-10-20(31-2)11-13-22)18-19-6-8-21(9-7-19)32-17-16-26-14-4-3-5-15-26/h6-13,28H,3-5,14-18H2,1-2H3,(H,25,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50128651

(4-(4-(4-chlorophenoxy)phenylsulfonyl)-1-ethyl-N-hy...)Show SMILES CCN1CCC(CC1)(C(=O)NO)S(=O)(=O)c1ccc(Oc2ccc(Cl)cc2)cc1 Show InChI InChI=1S/C20H23ClN2O5S/c1-2-23-13-11-20(12-14-23,19(24)22-25)29(26,27)18-9-7-17(8-10-18)28-16-5-3-15(21)4-6-16/h3-10,25H,2,11-14H2,1H3,(H,22,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-9. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128658
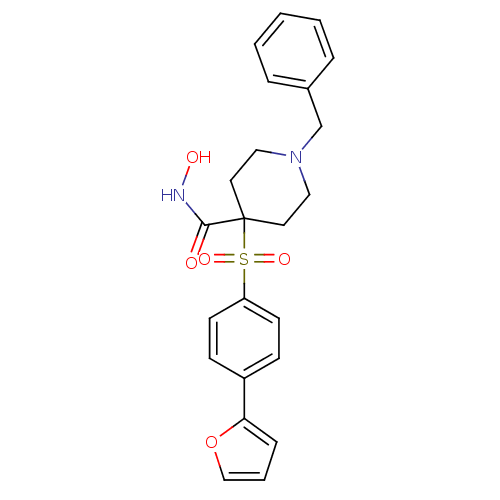
(1-Benzyl-4-(4-furan-2-yl-benzenesulfonyl)-piperidi...)Show SMILES ONC(=O)C1(CCN(Cc2ccccc2)CC1)S(=O)(=O)c1ccc(cc1)-c1ccco1 Show InChI InChI=1S/C23H24N2O5S/c26-22(24-27)23(12-14-25(15-13-23)17-18-5-2-1-3-6-18)31(28,29)20-10-8-19(9-11-20)21-7-4-16-30-21/h1-11,16,27H,12-15,17H2,(H,24,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128654
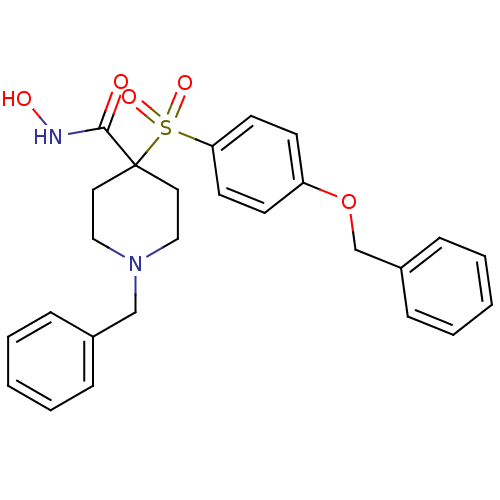
(1-Benzyl-4-(4-benzyloxy-benzenesulfonyl)-piperidin...)Show SMILES ONC(=O)C1(CCN(Cc2ccccc2)CC1)S(=O)(=O)c1ccc(OCc2ccccc2)cc1 Show InChI InChI=1S/C26H28N2O5S/c29-25(27-30)26(15-17-28(18-16-26)19-21-7-3-1-4-8-21)34(31,32)24-13-11-23(12-14-24)33-20-22-9-5-2-6-10-22/h1-14,30H,15-20H2,(H,27,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50118975
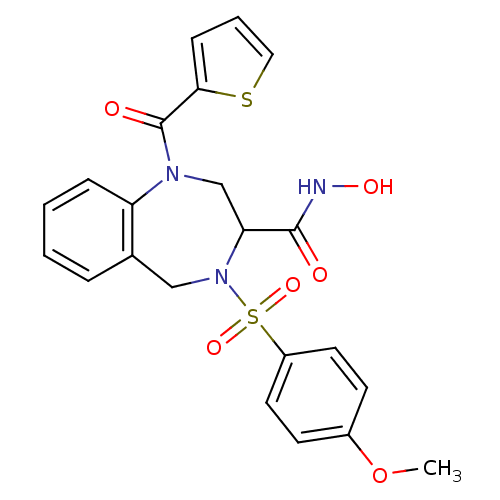
(4-(4-Methoxy-benzenesulfonyl)-1-(thiophene-2-carbo...)Show SMILES COc1ccc(cc1)S(=O)(=O)N1Cc2ccccc2N(CC1C(=O)NO)C(=O)c1cccs1 Show InChI InChI=1S/C22H21N3O6S2/c1-31-16-8-10-17(11-9-16)33(29,30)25-13-15-5-2-3-6-18(15)24(14-19(25)21(26)23-28)22(27)20-7-4-12-32-20/h2-12,19,28H,13-14H2,1H3,(H,23,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Concentration required in vitro to inhibit Matrix metalloproteinase-9 |
Bioorg Med Chem Lett 12: 2867-70 (2002)
BindingDB Entry DOI: 10.7270/Q2JS9PSH |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50128648
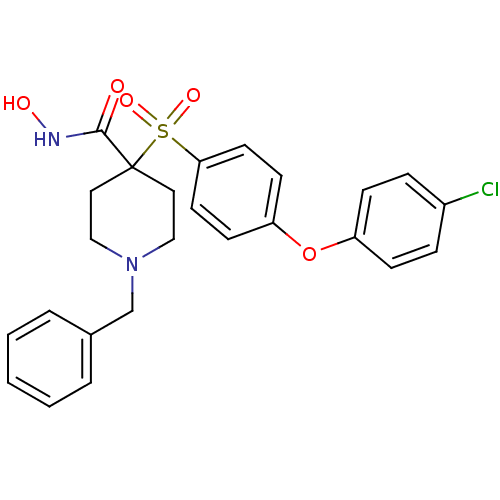
(1-Benzyl-4-[4-(4-chloro-phenoxy)-benzenesulfonyl]-...)Show SMILES ONC(=O)C1(CCN(Cc2ccccc2)CC1)S(=O)(=O)c1ccc(Oc2ccc(Cl)cc2)cc1 Show InChI InChI=1S/C25H25ClN2O5S/c26-20-6-8-21(9-7-20)33-22-10-12-23(13-11-22)34(31,32)25(24(29)27-30)14-16-28(17-15-25)18-19-4-2-1-3-5-19/h1-13,30H,14-18H2,(H,27,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-9. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50128668

(1-Butyl-4-[4-(4-chloro-phenoxy)-benzenesulfonyl]-p...)Show SMILES CCCCN1CCC(CC1)(C(=O)NO)S(=O)(=O)c1ccc(Oc2ccc(Cl)cc2)cc1 Show InChI InChI=1S/C22H27ClN2O5S/c1-2-3-14-25-15-12-22(13-16-25,21(26)24-27)31(28,29)20-10-8-19(9-11-20)30-18-6-4-17(23)5-7-18/h4-11,27H,2-3,12-16H2,1H3,(H,24,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-9. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50128648

(1-Benzyl-4-[4-(4-chloro-phenoxy)-benzenesulfonyl]-...)Show SMILES ONC(=O)C1(CCN(Cc2ccccc2)CC1)S(=O)(=O)c1ccc(Oc2ccc(Cl)cc2)cc1 Show InChI InChI=1S/C25H25ClN2O5S/c26-20-6-8-21(9-7-20)33-22-10-12-23(13-11-22)34(31,32)25(24(29)27-30)14-16-28(17-15-25)18-19-4-2-1-3-5-19/h1-13,30H,14-18H2,(H,27,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of Matrix metalloprotease-2 |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128667
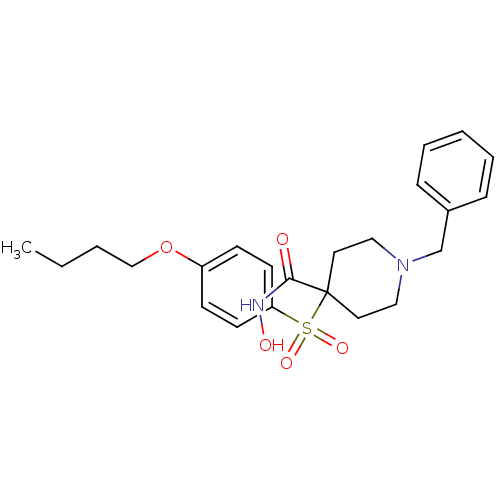
(1-Benzyl-4-(4-butoxy-benzenesulfonyl)-piperidine-4...)Show SMILES CCCCOc1ccc(cc1)S(=O)(=O)C1(CCN(Cc2ccccc2)CC1)C(=O)NO Show InChI InChI=1S/C23H30N2O5S/c1-2-3-17-30-20-9-11-21(12-10-20)31(28,29)23(22(26)24-27)13-15-25(16-14-23)18-19-7-5-4-6-8-19/h4-12,27H,2-3,13-18H2,1H3,(H,24,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128654

(1-Benzyl-4-(4-benzyloxy-benzenesulfonyl)-piperidin...)Show SMILES ONC(=O)C1(CCN(Cc2ccccc2)CC1)S(=O)(=O)c1ccc(OCc2ccccc2)cc1 Show InChI InChI=1S/C26H28N2O5S/c29-25(27-30)26(15-17-28(18-16-26)19-21-7-3-1-4-8-21)34(31,32)24-13-11-23(12-14-24)33-20-22-9-5-2-6-10-22/h1-14,30H,15-20H2,(H,27,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Neutrophil collagenase
(Homo sapiens (Human)) | BDBM50128648

(1-Benzyl-4-[4-(4-chloro-phenoxy)-benzenesulfonyl]-...)Show SMILES ONC(=O)C1(CCN(Cc2ccccc2)CC1)S(=O)(=O)c1ccc(Oc2ccc(Cl)cc2)cc1 Show InChI InChI=1S/C25H25ClN2O5S/c26-20-6-8-21(9-7-20)33-22-10-12-23(13-11-22)34(31,32)25(24(29)27-30)14-16-28(17-15-25)18-19-4-2-1-3-5-19/h1-13,30H,14-18H2,(H,27,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of Matrix metalloprotease-8 |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128648

(1-Benzyl-4-[4-(4-chloro-phenoxy)-benzenesulfonyl]-...)Show SMILES ONC(=O)C1(CCN(Cc2ccccc2)CC1)S(=O)(=O)c1ccc(Oc2ccc(Cl)cc2)cc1 Show InChI InChI=1S/C25H25ClN2O5S/c26-20-6-8-21(9-7-20)33-22-10-12-23(13-11-22)34(31,32)25(24(29)27-30)14-16-28(17-15-25)18-19-4-2-1-3-5-19/h1-13,30H,14-18H2,(H,27,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128597

((R)-N-hydroxy-2-(4-methoxyphenylsulfonyl)-2-methyl...)Show SMILES COc1ccc(cc1)S(=O)(=O)C(C)(Cc1ccc(OCCN2CCCCC2)cc1)C(=O)NO Show InChI InChI=1S/C24H32N2O6S/c1-24(23(27)25-28,33(29,30)22-12-10-20(31-2)11-13-22)18-19-6-8-21(9-7-19)32-17-16-26-14-4-3-5-15-26/h6-13,28H,3-5,14-18H2,1-2H3,(H,25,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibition of matrix metalloprotease-13 |
J Med Chem 46: 2361-75 (2003)
Article DOI: 10.1021/jm0205548
BindingDB Entry DOI: 10.7270/Q2RR1ZZJ |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128673
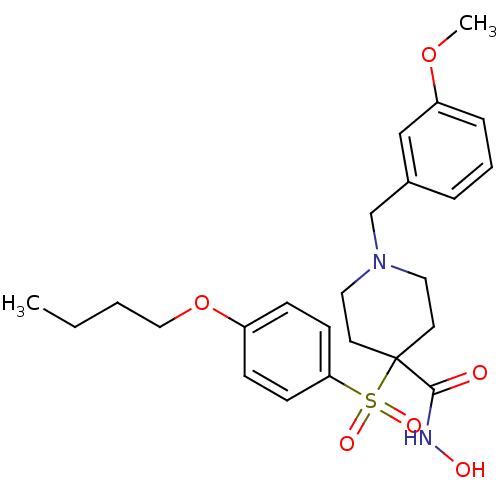
(1-(3-methoxybenzyl)-4-(4-butoxyphenylsulfonyl)-N-h...)Show SMILES CCCCOc1ccc(cc1)S(=O)(=O)C1(CCN(Cc2cccc(OC)c2)CC1)C(=O)NO Show InChI InChI=1S/C24H32N2O6S/c1-3-4-16-32-20-8-10-22(11-9-20)33(29,30)24(23(27)25-28)12-14-26(15-13-24)18-19-6-5-7-21(17-19)31-2/h5-11,17,28H,3-4,12-16,18H2,1-2H3,(H,25,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50126622
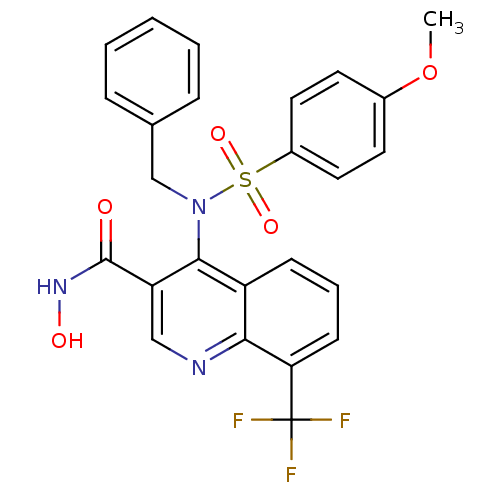
(4-(N-benzyl-4-methoxyphenylsulfonamido)-N-hydroxy-...)Show SMILES COc1ccc(cc1)S(=O)(=O)N(Cc1ccccc1)c1c(cnc2c(cccc12)C(F)(F)F)C(=O)NO Show InChI InChI=1S/C25H20F3N3O5S/c1-36-17-10-12-18(13-11-17)37(34,35)31(15-16-6-3-2-4-7-16)23-19-8-5-9-21(25(26,27)28)22(19)29-14-20(23)24(32)30-33/h2-14,33H,15H2,1H3,(H,30,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth-Ayerst Research
Curated by ChEMBL
| Assay Description
Inhibitory activity against matrix metalloproteinase 13 (MMP-13). |
Bioorg Med Chem Lett 13: 1487-90 (2003)
BindingDB Entry DOI: 10.7270/Q29W0DW0 |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50126623
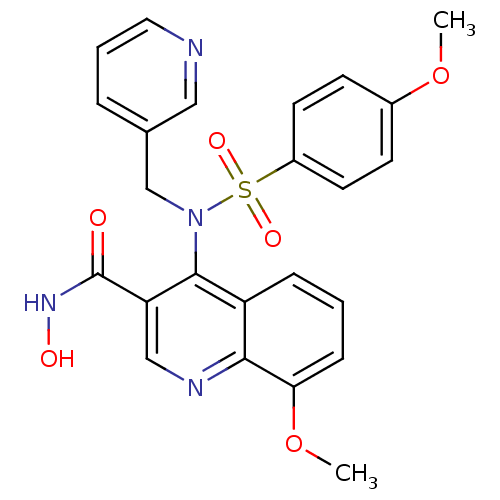
(8-Methoxy-4-[(4-methoxy-benzenesulfonyl)-pyridin-3...)Show SMILES COc1ccc(cc1)S(=O)(=O)N(Cc1cccnc1)c1c(cnc2c(OC)cccc12)C(=O)NO Show InChI InChI=1S/C24H22N4O6S/c1-33-17-8-10-18(11-9-17)35(31,32)28(15-16-5-4-12-25-13-16)23-19-6-3-7-21(34-2)22(19)26-14-20(23)24(29)27-30/h3-14,30H,15H2,1-2H3,(H,27,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth-Ayerst Research
Curated by ChEMBL
| Assay Description
Inhibitory activity against matrix metalloproteinase 13 (MMP-13). |
Bioorg Med Chem Lett 13: 1487-90 (2003)
BindingDB Entry DOI: 10.7270/Q29W0DW0 |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50096472
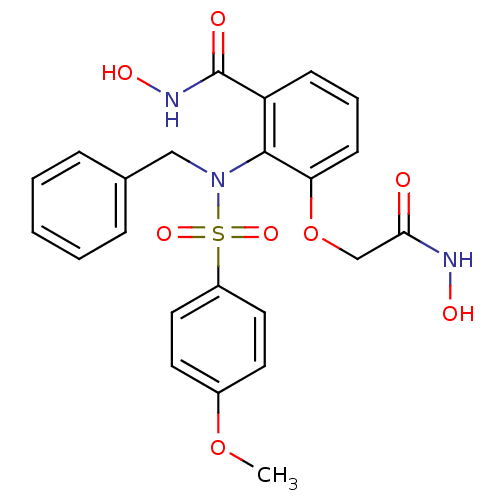
(2-(N-benzyl-4-methoxyphenylsulfonamido)-N-hydroxy-...)Show SMILES COc1ccc(cc1)S(=O)(=O)N(Cc1ccccc1)c1c(OCC(=O)NO)cccc1C(=O)NO Show InChI InChI=1S/C23H23N3O8S/c1-33-17-10-12-18(13-11-17)35(31,32)26(14-16-6-3-2-4-7-16)22-19(23(28)25-30)8-5-9-20(22)34-15-21(27)24-29/h2-13,29-30H,14-15H2,1H3,(H,24,27)(H,25,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth-Ayerst Research
Curated by ChEMBL
| Assay Description
Inhibitory activity against Matrix metalloprotease-13 |
Bioorg Med Chem Lett 11: 235-8 (2001)
BindingDB Entry DOI: 10.7270/Q2TH8N78 |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50106130
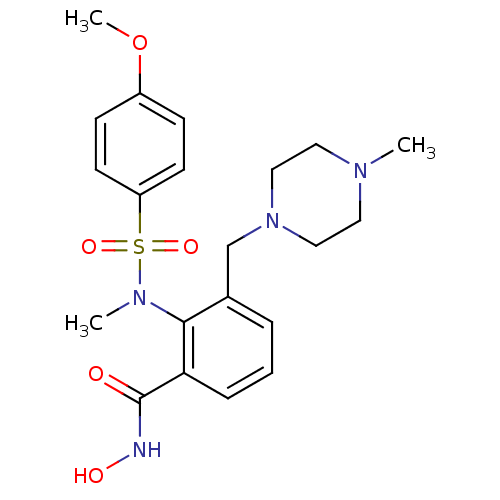
(CHEMBL100512 | N-Hydroxy-2-[(4-methoxy-benzenesulf...)Show SMILES COc1ccc(cc1)S(=O)(=O)N(C)c1c(CN2CCN(C)CC2)cccc1C(=O)NO Show InChI InChI=1S/C21H28N4O5S/c1-23-11-13-25(14-12-23)15-16-5-4-6-19(21(26)22-27)20(16)24(2)31(28,29)18-9-7-17(30-3)8-10-18/h4-10,27H,11-15H2,1-3H3,(H,22,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth-Ayerst Research
Curated by ChEMBL
| Assay Description
In vitro inhibition of Matrix metalloprotease-9. |
Bioorg Med Chem Lett 11: 2975-8 (2001)
BindingDB Entry DOI: 10.7270/Q2125T64 |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50106136

(2-{[4-(4-Chloro-phenoxy)-benzenesulfonyl]-methyl-a...)Show SMILES CN(c1c(CN2CCN(C)CC2)cccc1C(=O)NO)S(=O)(=O)c1ccc(Oc2ccc(Cl)cc2)cc1 Show InChI InChI=1S/C26H29ClN4O5S/c1-29-14-16-31(17-15-29)18-19-4-3-5-24(26(32)28-33)25(19)30(2)37(34,35)23-12-10-22(11-13-23)36-21-8-6-20(27)7-9-21/h3-13,33H,14-18H2,1-2H3,(H,28,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth-Ayerst Research
Curated by ChEMBL
| Assay Description
In vitro inhibition of Matrix metalloprotease-9. |
Bioorg Med Chem Lett 11: 2975-8 (2001)
BindingDB Entry DOI: 10.7270/Q2125T64 |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50126626

(4-{Methyl-[4-(pyridin-4-yloxy)-benzenesulfonyl]-am...)Show SMILES CN(c1c(cnc2ccccc12)C(=O)NO)S(=O)(=O)c1ccc(Oc2ccncc2)cc1 Show InChI InChI=1S/C22H18N4O5S/c1-26(21-18-4-2-3-5-20(18)24-14-19(21)22(27)25-28)32(29,30)17-8-6-15(7-9-17)31-16-10-12-23-13-11-16/h2-14,28H,1H3,(H,25,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth-Ayerst Research
Curated by ChEMBL
| Assay Description
Inhibitory activity against matrix metalloproteinase 9 (MMP-9). |
Bioorg Med Chem Lett 13: 1487-90 (2003)
BindingDB Entry DOI: 10.7270/Q29W0DW0 |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50103161

(3-Furan-2-yl-N-hydroxy-2-[(4-methoxy-benzenesulfon...)Show SMILES COc1ccc(cc1)S(=O)(=O)N(Cc1cccnc1)c1c(cc(C)cc1-c1ccco1)C(=O)NO Show InChI InChI=1S/C25H23N3O6S/c1-17-13-21(23-6-4-12-34-23)24(22(14-17)25(29)27-30)28(16-18-5-3-11-26-15-18)35(31,32)20-9-7-19(33-2)8-10-20/h3-15,30H,16H2,1-2H3,(H,27,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth-Ayerst Research
Curated by ChEMBL
| Assay Description
Inhibition of matrix metalloprotease-13 |
Bioorg Med Chem Lett 11: 2189-92 (2001)
BindingDB Entry DOI: 10.7270/Q2154HK0 |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50103171

(4-[(4-Methoxy-benzenesulfonyl)-pyridin-3-ylmethyl-...)Show SMILES COc1ccc(cc1)S(=O)(=O)N(Cc1cccnc1)c1c(C)cc(cc1C(=O)NO)-c1cccc(c1)C(F)(F)F Show InChI InChI=1S/C28H24F3N3O5S/c1-18-13-21(20-6-3-7-22(14-20)28(29,30)31)15-25(27(35)33-36)26(18)34(17-19-5-4-12-32-16-19)40(37,38)24-10-8-23(39-2)9-11-24/h3-16,36H,17H2,1-2H3,(H,33,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth-Ayerst Research
Curated by ChEMBL
| Assay Description
In vitro ability to inhibit matrix metalloprotease-9. |
Bioorg Med Chem Lett 11: 2189-92 (2001)
BindingDB Entry DOI: 10.7270/Q2154HK0 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data