| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Isocitrate dehydrogenase [NADP] cytoplasmic [R132H] |
|---|
| Ligand | BDBM389381 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Biochemical Assay |
|---|
| IC50 | 4.00±n/a nM |
|---|
| Citation |  Rehwinkel, H; Siebeneicher, H; Anlauf, S; Nguyen, D; Panknin, O; Ring, S; Schwede, W; Bauser, M; Zimmermann, K; Kaulfuss, S; Neuhaus, R Benzimidazol-2-amines as MIDH1 inhibitors US Patent US9951027 Publication Date 4/24/2018 Rehwinkel, H; Siebeneicher, H; Anlauf, S; Nguyen, D; Panknin, O; Ring, S; Schwede, W; Bauser, M; Zimmermann, K; Kaulfuss, S; Neuhaus, R Benzimidazol-2-amines as MIDH1 inhibitors US Patent US9951027 Publication Date 4/24/2018 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Isocitrate dehydrogenase [NADP] cytoplasmic [R132H] |
|---|
| Name: | Isocitrate dehydrogenase [NADP] cytoplasmic [R132H] |
|---|
| Synonyms: | Cytosolic NADP-isocitrate dehydrogenase (IDH1)(R132H) | IDH1 | IDH1 R132H | IDH1(R132H) | IDHC_HUMAN | Isocitrate dehydrogenase (IDH1)(R132H) | Isocitrate dehydrogenase 1 mutant (R132H) | Isocitrate dehydrogenase [NADP] cytoplasmic (IDH)(R132H) | Isocitrate dehydrogenase [NADP] cytoplasmic (IDH1)(R132H) | Isocitrate dehydrogenase [NADP] cytoplasmic (R132H) | PICD |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 46641.74 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Human IDH1 R132H (SEQ ID No. 2 in patent). First three are removed. Google patent parsed wrong. |
|---|
| Residue: | 414 |
|---|
| Sequence: | MSKKISGGSVVEMQGDEMTRIIWELIKEKLIFPYVELDLHSYDLGIENRDATNDQVTKDA
AEAIKKHNVGVKCATITPDEKRVEEFKLKQMWKSPNGTIRNILGGTVFREAIICKNIPRL
VSGWVKPIIIGHHAYGDQYRATDFVVPGPGKVEITYTPSDGTQKVTYLVHNFEEGGGVAM
GMYNQDKSIEDFAHSSFQMALSKGWPLYLSTKNTILKKYDGRFKDIFQEIYDKQYKSQFE
AQKIWYEHRLIDDMVAQAMKSEGGFIWACKNYDGDVQSDSVAQGYGSLGMMTSVLVCPDG
KTVEAEAAHGTVTRHYRMYQKGQETSTNPIASIFAWTRGLAHRAKLDNNKELAFFANALE
EVSIETIEAGFMTKDLAACIKGLPNVQRSDYLNTFEFMDKLGENLKIKLAQAKL
|
|
|
|---|
| BDBM389381 |
|---|
| n/a |
|---|
| Name | BDBM389381 |
|---|
| Synonyms: | (+-) 6-methoxy-2-{[4-(trifluoromethoxy)phenyl]amino}-1-[(cis)-3,3,5-trimethylcyclohexyl]-1H-benzimidazole-5-carboxylic acid | US9951027, 2-153 | US9951027, 2-153-2 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H28F3N3O4 |
|---|
| Mol. Mass. | 491.5027 |
|---|
| SMILES | COc1cc2n([C@H]3C[C@@H](C)CC(C)(C)C3)c(Nc3ccc(OC(F)(F)F)cc3)nc2cc1C(O)=O |r| |
|---|
| Structure | 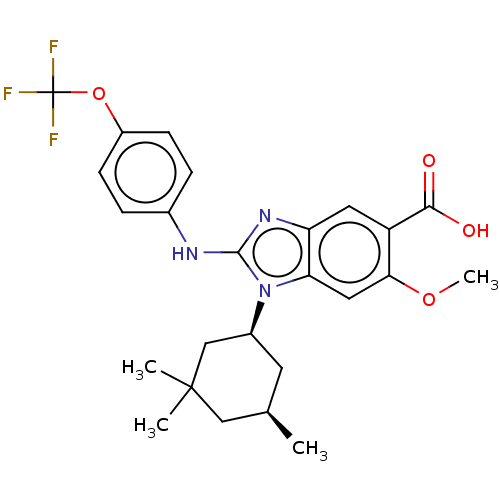
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Rehwinkel, H; Siebeneicher, H; Anlauf, S; Nguyen, D; Panknin, O; Ring, S; Schwede, W; Bauser, M; Zimmermann, K; Kaulfuss, S; Neuhaus, R Benzimidazol-2-amines as MIDH1 inhibitors US Patent US9951027 Publication Date 4/24/2018
Rehwinkel, H; Siebeneicher, H; Anlauf, S; Nguyen, D; Panknin, O; Ring, S; Schwede, W; Bauser, M; Zimmermann, K; Kaulfuss, S; Neuhaus, R Benzimidazol-2-amines as MIDH1 inhibitors US Patent US9951027 Publication Date 4/24/2018