| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prostaglandin G/H synthase 1 |
|---|
| Ligand | BDBM50462392 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1774824 (CHEMBL4231816) |
|---|
| IC50 | >100000±n/a nM |
|---|
| Citation |  Pippione, AC; Carnovale, IM; Bonanni, D; Sini, M; Goyal, P; Marini, E; Pors, K; Adinolfi, S; Zonari, D; Festuccia, C; Wahlgren, WY; Friemann, R; Bagnati, R; Boschi, D; Oliaro-Bosso, S; Lolli, ML Potent and selective aldo-keto reductase 1C3 (AKR1C3) inhibitors based on the benzoisoxazole moiety: application of a bioisosteric scaffold hopping approach to flufenamic acid. Eur J Med Chem150:930-945 (2018) [PubMed] Article Pippione, AC; Carnovale, IM; Bonanni, D; Sini, M; Goyal, P; Marini, E; Pors, K; Adinolfi, S; Zonari, D; Festuccia, C; Wahlgren, WY; Friemann, R; Bagnati, R; Boschi, D; Oliaro-Bosso, S; Lolli, ML Potent and selective aldo-keto reductase 1C3 (AKR1C3) inhibitors based on the benzoisoxazole moiety: application of a bioisosteric scaffold hopping approach to flufenamic acid. Eur J Med Chem150:930-945 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prostaglandin G/H synthase 1 |
|---|
| Name: | Prostaglandin G/H synthase 1 |
|---|
| Synonyms: | COX1 | Cyclooxygenase-1 | Cyclooxygenase-1 (COX-1) | PGH1_SHEEP | PTGS1 | Prostaglandin G/H synthase (cyclooxygenase) |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 68868.60 |
|---|
| Organism: | Ovis aries (Sheep) |
|---|
| Description: | n/a |
|---|
| Residue: | 600 |
|---|
| Sequence: | MSRQSISLRFPLLLLLLSPSPVFSADPGAPAPVNPCCYYPCQHQGICVRFGLDRYQCDCT
RTGYSGPNCTIPEIWTWLRTTLRPSPSFIHFLLTHGRWLWDFVNATFIRDTLMRLVLTVR
SNLIPSPPTYNIAHDYISWESFSNVSYYTRILPSVPRDCPTPMDTKGKKQLPDAEFLSRR
FLLRRKFIPDPQSTNLMFAFFAQHFTHQFFKTSGKMGPGFTKALGHGVDLGHIYGDNLER
QYQLRLFKDGKLKYQMLNGEVYPPSVEEAPVLMHYPRGIPPQSQMAVGQEVFGLLPGLML
YATIWLREHNRVCDLLKAEHPTWGDEQLFQTARLILIGETIKIVIEEYVQQLSGYFLQLK
FDPELLFGAQFQYRNRIAMEFNQLYHWHPLMPDSFRVGPQDYSYEQFLFNTSMLVDYGVE
ALVDAFSRQPAGRIGGGRNIDHHILHVAVDVIKESRVLRLQPFNEYRKRFGMKPYTSFQE
LTGEKEMAAELEELYGDIDALEFYPGLLLEKCHPNSIFGESMIEMGAPFSLKGLLGNPIC
SPEYWKASTFGGEVGFNLVKTATLKKLVCLNTKTCPYVSFHVPDPRQEDRPGVERPPTEL
|
|
|
|---|
| BDBM50462392 |
|---|
| n/a |
|---|
| Name | BDBM50462392 |
|---|
| Synonyms: | CHEMBL4248154 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H12F6N4O3 |
|---|
| Mol. Mass. | 446.3033 |
|---|
| SMILES | Oc1nnn(Cc2ccc(OC(F)(F)F)cc2)c1C(=O)Nc1cccc(c1)C(F)(F)F |
|---|
| Structure | 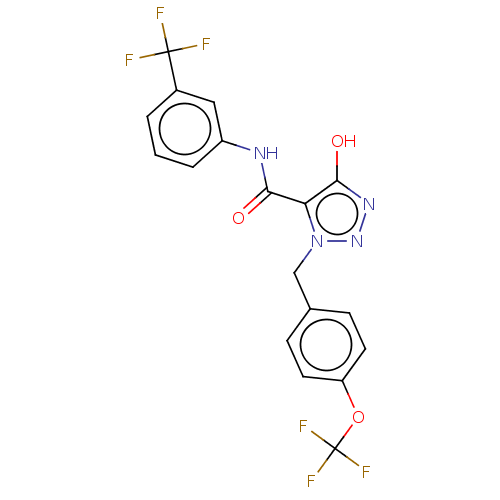
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Pippione, AC; Carnovale, IM; Bonanni, D; Sini, M; Goyal, P; Marini, E; Pors, K; Adinolfi, S; Zonari, D; Festuccia, C; Wahlgren, WY; Friemann, R; Bagnati, R; Boschi, D; Oliaro-Bosso, S; Lolli, ML Potent and selective aldo-keto reductase 1C3 (AKR1C3) inhibitors based on the benzoisoxazole moiety: application of a bioisosteric scaffold hopping approach to flufenamic acid. Eur J Med Chem150:930-945 (2018) [PubMed] Article
Pippione, AC; Carnovale, IM; Bonanni, D; Sini, M; Goyal, P; Marini, E; Pors, K; Adinolfi, S; Zonari, D; Festuccia, C; Wahlgren, WY; Friemann, R; Bagnati, R; Boschi, D; Oliaro-Bosso, S; Lolli, ML Potent and selective aldo-keto reductase 1C3 (AKR1C3) inhibitors based on the benzoisoxazole moiety: application of a bioisosteric scaffold hopping approach to flufenamic acid. Eur J Med Chem150:930-945 (2018) [PubMed] Article