| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 11B2, mitochondrial |
|---|
| Ligand | BDBM50500186 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1545187 (CHEMBL3750668) |
|---|
| IC50 | 13±n/a nM |
|---|
| Citation |  Papillon, JP; Lou, C; Singh, AK; Adams, CM; Ksander, GM; Beil, ME; Chen, W; Leung-Chu, J; Fu, F; Gan, L; Hu, CW; Jeng, AY; LaSala, D; Liang, G; Rigel, DF; Russell, KS; Vest, JA; Watson, C Discovery of N-[5-(6-Chloro-3-cyano-1-methyl-1H-indol-2-yl)-pyridin-3-ylmethyl]-ethanesulfonamide, a Cortisol-Sparing CYP11B2 Inhibitor that Lowers Aldosterone in Human Subjects. J Med Chem58:9382-94 (2015) [PubMed] Article Papillon, JP; Lou, C; Singh, AK; Adams, CM; Ksander, GM; Beil, ME; Chen, W; Leung-Chu, J; Fu, F; Gan, L; Hu, CW; Jeng, AY; LaSala, D; Liang, G; Rigel, DF; Russell, KS; Vest, JA; Watson, C Discovery of N-[5-(6-Chloro-3-cyano-1-methyl-1H-indol-2-yl)-pyridin-3-ylmethyl]-ethanesulfonamide, a Cortisol-Sparing CYP11B2 Inhibitor that Lowers Aldosterone in Human Subjects. J Med Chem58:9382-94 (2015) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 11B2, mitochondrial |
|---|
| Name: | Cytochrome P450 11B2, mitochondrial |
|---|
| Synonyms: | Aldosterone Synthase (CYP11B2) | Aldosterone synthase | Aldosterone-synthesizing enzyme | C11B2_HUMAN | CYP11B2 | CYPXIB2 | Cytochrome P450 11B2 | Cytochrome P450 11B2 (CYP11B2) | Cytochrome P450 11B2, mitochondrial | P-450Aldo | P-450C18 | Steroid 18-hydroxylase |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 57582.15 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P19099 |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALRAKAEVCVAAPWLSLQRARALGTRAARAPRTVLPFEAMPQHPGNRWLRLLQIWREQG
YEHLHLEMHQTFQELGPIFRYNLGGPRMVCVMLPEDVEKLQQVDSLHPCRMILEPWVAYR
QHRGHKCGVFLLNGPEWRFNRLRLNPDVLSPKAVQRFLPMVDAVARDFSQALKKKVLQNA
RGSLTLDVQPSIFHYTIEASNLALFGERLGLVGHSPSSASLNFLHALEVMFKSTVQLMFM
PRSLSRWISPKVWKEHFEAWDCIFQYGDNCIQKIYQELAFNRPQHYTGIVAELLLKAELS
LEAIKANSMELTAGSVDTTAFPLLMTLFELARNPDVQQILRQESLAAAASISEHPQKATT
ELPLLRAALKETLRLYPVGLFLERVVSSDLVLQNYHIPAGTLVQVFLYSLGRNAALFPRP
ERYNPQRWLDIRGSGRNFHHVPFGFGMRQCLGRRLAEAEMLLLLHHVLKHFLVETLTQED
IKMVYSFILRPGTSPLLTFRAIN
|
|
|
|---|
| BDBM50500186 |
|---|
| n/a |
|---|
| Name | BDBM50500186 |
|---|
| Synonyms: | CHEMBL3747069 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C17H15ClN4O2S |
|---|
| Mol. Mass. | 374.845 |
|---|
| SMILES | CCS(=O)(=O)Nc1cncc(c1)-c1c(C#N)c2ccc(Cl)cc2n1C |
|---|
| Structure | 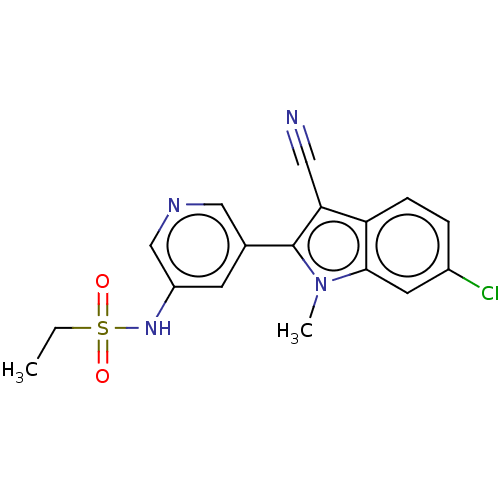
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Papillon, JP; Lou, C; Singh, AK; Adams, CM; Ksander, GM; Beil, ME; Chen, W; Leung-Chu, J; Fu, F; Gan, L; Hu, CW; Jeng, AY; LaSala, D; Liang, G; Rigel, DF; Russell, KS; Vest, JA; Watson, C Discovery of N-[5-(6-Chloro-3-cyano-1-methyl-1H-indol-2-yl)-pyridin-3-ylmethyl]-ethanesulfonamide, a Cortisol-Sparing CYP11B2 Inhibitor that Lowers Aldosterone in Human Subjects. J Med Chem58:9382-94 (2015) [PubMed] Article
Papillon, JP; Lou, C; Singh, AK; Adams, CM; Ksander, GM; Beil, ME; Chen, W; Leung-Chu, J; Fu, F; Gan, L; Hu, CW; Jeng, AY; LaSala, D; Liang, G; Rigel, DF; Russell, KS; Vest, JA; Watson, C Discovery of N-[5-(6-Chloro-3-cyano-1-methyl-1H-indol-2-yl)-pyridin-3-ylmethyl]-ethanesulfonamide, a Cortisol-Sparing CYP11B2 Inhibitor that Lowers Aldosterone in Human Subjects. J Med Chem58:9382-94 (2015) [PubMed] Article