| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Mitogen-activated protein kinase kinase kinase 20 |
|---|
| Ligand | BDBM50513876 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1854813 (CHEMBL4355542) |
|---|
| IC50 | 983±n/a nM |
|---|
| Citation |  Yang, J; Shibu, MA; Kong, L; Luo, J; BadrealamKhan, F; Huang, Y; Tu, ZC; Yun, CH; Huang, CY; Ding, K; Lu, X Design, Synthesis, and Structure-Activity Relationships of 1,2,3-Triazole Benzenesulfonamides as New Selective Leucine-Zipper and Sterile-? Motif Kinase (ZAK) Inhibitors. J Med Chem63:2114-2130 (2020) [PubMed] Article Yang, J; Shibu, MA; Kong, L; Luo, J; BadrealamKhan, F; Huang, Y; Tu, ZC; Yun, CH; Huang, CY; Ding, K; Lu, X Design, Synthesis, and Structure-Activity Relationships of 1,2,3-Triazole Benzenesulfonamides as New Selective Leucine-Zipper and Sterile-? Motif Kinase (ZAK) Inhibitors. J Med Chem63:2114-2130 (2020) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Mitogen-activated protein kinase kinase kinase 20 |
|---|
| Name: | Mitogen-activated protein kinase kinase kinase 20 |
|---|
| Synonyms: | M3K20_HUMAN | MAP3K20 | MLK7 | MLTK | Mixed lineage kinase 7 | ZAK |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 91168.58 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_1495629 |
|---|
| Residue: | 800 |
|---|
| Sequence: | MSSLGASFVQIKFDDLQFFENCGGGSFGSVYRAKWISQDKEVAVKKLLKIEKEAEILSVL
SHRNIIQFYGVILEPPNYGIVTEYASLGSLYDYINSNRSEEMDMDHIMTWATDVAKGMHY
LHMEAPVKVIHRDLKSRNVVIAADGVLKICDFGASRFHNHTTHMSLVGTFPWMAPEVIQS
LPVSETCDTYSYGVVLWEMLTREVPFKGLEGLQVAWLVVEKNERLTIPSSCPRSFAELLH
QCWEADAKKRPSFKQIISILESMSNDTSLPDKCNSFLHNKAEWRCEIEATLERLKKLERD
LSFKEQELKERERRLKMWEQKLTEQSNTPLLPSFEIGAWTEDDVYCWVQQLVRKGDSSAE
MSVYASLFKENNITGKRLLLLEEEDLKDMGIVSKGHIIHFKSAIEKLTHDYINLFHFPPL
IKDSGGEPEENEEKIVNLELVFGFHLKPGTGPQDCKWKMYMEMDGDEIAITYIKDVTFNT
NLPDAEILKMTKPPFVMEKWIVGIAKSQTVECTVTYESDVRTPKSTKHVHSIQWSRTKPQ
DEVKAVQLAIQTLFTNSDGNPGSRSDSSADCQWLDTLRMRQIASNTSLQRSQSNPILGSP
FFSHFDGQDSYAAAVRRPQVPIKYQQITPVNQSRSSSPTQYGLTKNFSSLHLNSRDSGFS
SGNTDTSSERGRYSDRSRNKYGRGSISLNSSPRGRYSGKSQHSTPSRGRYPGKFYRVSQS
ALNPHQSPDFKRSPRDLHQPNTIPGMPLHPETDSRASEEDSKVSEGGWTKVEYRKKPHRP
SPAKTNKERARGDHRGWRNF
|
|
|
|---|
| BDBM50513876 |
|---|
| n/a |
|---|
| Name | BDBM50513876 |
|---|
| Synonyms: | CHEMBL4459658 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H15ClF2N6O3S |
|---|
| Mol. Mass. | 516.908 |
|---|
| SMILES | COc1n[nH]c2ncc(cc12)-c1ncc([nH]1)-c1c(F)ccc(NS(=O)(=O)c2cccc(Cl)c2)c1F |
|---|
| Structure | 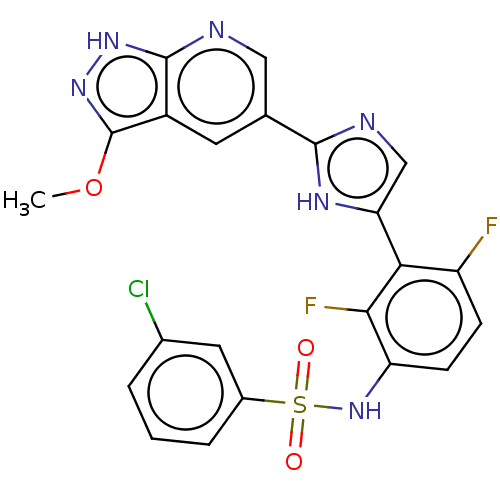
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Yang, J; Shibu, MA; Kong, L; Luo, J; BadrealamKhan, F; Huang, Y; Tu, ZC; Yun, CH; Huang, CY; Ding, K; Lu, X Design, Synthesis, and Structure-Activity Relationships of 1,2,3-Triazole Benzenesulfonamides as New Selective Leucine-Zipper and Sterile-? Motif Kinase (ZAK) Inhibitors. J Med Chem63:2114-2130 (2020) [PubMed] Article
Yang, J; Shibu, MA; Kong, L; Luo, J; BadrealamKhan, F; Huang, Y; Tu, ZC; Yun, CH; Huang, CY; Ding, K; Lu, X Design, Synthesis, and Structure-Activity Relationships of 1,2,3-Triazole Benzenesulfonamides as New Selective Leucine-Zipper and Sterile-? Motif Kinase (ZAK) Inhibitors. J Med Chem63:2114-2130 (2020) [PubMed] Article